 Found 57 hits with Last Name = 'dickman' and Initial = 'd'
Found 57 hits with Last Name = 'dickman' and Initial = 'd' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Tryptase delta
(Homo sapiens (Human)) | BDBM50475863
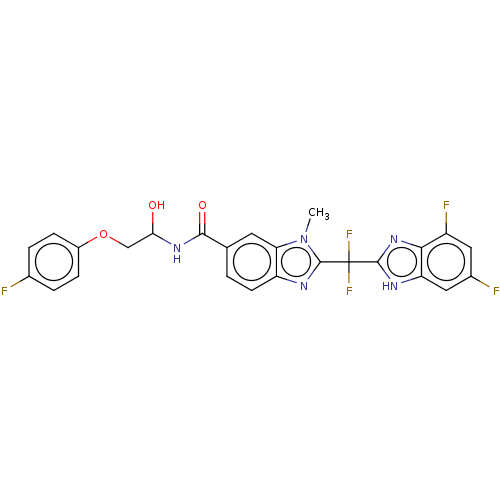
(CHEMBL209054)Show SMILES Cn1c(nc2ccc(cc12)C(=O)NC(O)COc1ccc(F)cc1)C(F)(F)c1nc2c(F)cc(F)cc2[nH]1 Show InChI InChI=1S/C25H18F5N5O3/c1-35-19-8-12(22(37)33-20(36)11-38-15-5-3-13(26)4-6-15)2-7-17(19)32-24(35)25(29,30)23-31-18-10-14(27)9-16(28)21(18)34-23/h2-10,20,36H,11H2,1H3,(H,31,34)(H,33,37) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Inhibition of mast cell tryptase |
Bioorg Med Chem Lett 16: 4053-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.003
BindingDB Entry DOI: 10.7270/Q26H4M69 |
More data for this
Ligand-Target Pair | |
Tryptase delta
(Homo sapiens (Human)) | BDBM50475860
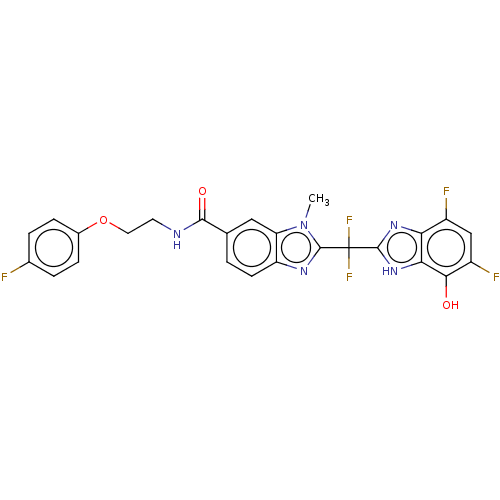
(CHEMBL424699)Show SMILES Cn1c(nc2ccc(cc12)C(=O)NCCOc1ccc(F)cc1)C(F)(F)c1nc2c(F)cc(F)c(O)c2[nH]1 Show InChI InChI=1S/C25H18F5N5O3/c1-35-18-10-12(22(37)31-8-9-38-14-5-3-13(26)4-6-14)2-7-17(18)32-24(35)25(29,30)23-33-19-15(27)11-16(28)21(36)20(19)34-23/h2-7,10-11,36H,8-9H2,1H3,(H,31,37)(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 34 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Inhibition of mast cell tryptase |
Bioorg Med Chem Lett 16: 4053-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.003
BindingDB Entry DOI: 10.7270/Q26H4M69 |
More data for this
Ligand-Target Pair | |
Tryptase delta
(Homo sapiens (Human)) | BDBM50475861
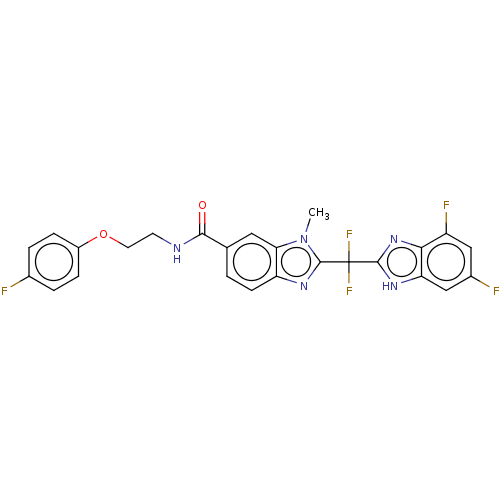
(CHEMBL211733)Show SMILES Cn1c(nc2ccc(cc12)C(=O)NCCOc1ccc(F)cc1)C(F)(F)c1nc2c(F)cc(F)cc2[nH]1 Show InChI InChI=1S/C25H18F5N5O2/c1-35-20-10-13(22(36)31-8-9-37-16-5-3-14(26)4-6-16)2-7-18(20)33-24(35)25(29,30)23-32-19-12-15(27)11-17(28)21(19)34-23/h2-7,10-12H,8-9H2,1H3,(H,31,36)(H,32,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Inhibition of mast cell tryptase |
Bioorg Med Chem Lett 16: 4053-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.003
BindingDB Entry DOI: 10.7270/Q26H4M69 |
More data for this
Ligand-Target Pair | |
Tryptase delta
(Homo sapiens (Human)) | BDBM50475865

(CHEMBL212765)Show SMILES Cn1c(nc2ccc(cc12)C(=O)NCCOc1ccc(F)cc1)C(F)(F)c1nc2c(F)c(O)c(F)cc2[nH]1 Show InChI InChI=1S/C25H18F5N5O3/c1-35-18-10-12(22(37)31-8-9-38-14-5-3-13(26)4-6-14)2-7-16(18)33-24(35)25(29,30)23-32-17-11-15(27)21(36)19(28)20(17)34-23/h2-7,10-11,36H,8-9H2,1H3,(H,31,37)(H,32,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Inhibition of mast cell tryptase |
Bioorg Med Chem Lett 16: 4053-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.003
BindingDB Entry DOI: 10.7270/Q26H4M69 |
More data for this
Ligand-Target Pair | |
Tryptase delta
(Homo sapiens (Human)) | BDBM50475864
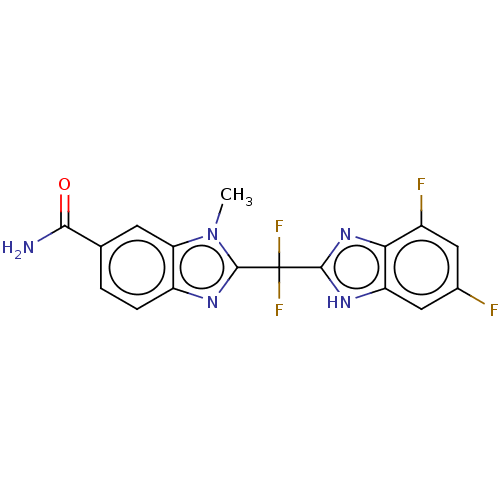
(CHEMBL378085)Show SMILES Cn1c(nc2ccc(cc12)C(N)=O)C(F)(F)c1nc2c(F)cc(F)cc2[nH]1 Show InChI InChI=1S/C17H11F4N5O/c1-26-12-4-7(14(22)27)2-3-10(12)24-16(26)17(20,21)15-23-11-6-8(18)5-9(19)13(11)25-15/h2-6H,1H3,(H2,22,27)(H,23,25) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Inhibition of mast cell tryptase |
Bioorg Med Chem Lett 16: 4053-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.003
BindingDB Entry DOI: 10.7270/Q26H4M69 |
More data for this
Ligand-Target Pair | |
Tryptase delta
(Homo sapiens (Human)) | BDBM50475862

(CHEMBL379076)Show SMILES Cn1c(nc2ccc(cc12)C(=O)NCCO)C(F)(F)c1nc2c(F)cc(F)cc2[nH]1 Show InChI InChI=1S/C19H15F4N5O2/c1-28-14-6-9(16(30)24-4-5-29)2-3-12(14)26-18(28)19(22,23)17-25-13-8-10(20)7-11(21)15(13)27-17/h2-3,6-8,29H,4-5H2,1H3,(H,24,30)(H,25,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Inhibition of mast cell tryptase |
Bioorg Med Chem Lett 16: 4053-8 (2006)
Article DOI: 10.1016/j.bmcl.2006.05.003
BindingDB Entry DOI: 10.7270/Q26H4M69 |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50067563

((S)-2-{[2'-Methyl-5-(pyridin-3-yloxymethyl)-biphen...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COc2cccnc2)cc1-c1ccccc1C)C(O)=O Show InChI InChI=1S/C25H26N2O4S/c1-17-6-3-4-8-20(17)22-14-18(16-31-19-7-5-12-26-15-19)9-10-21(22)24(28)27-23(25(29)30)11-13-32-2/h3-10,12,14-15,23H,11,13,16H2,1-2H3,(H,27,28)(H,29,30)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50067584

((S)-2-{[5-((R)-2-Amino-3-mercapto-propylamino)-bip...)Show SMILES CSCC[C@H](NC(=O)c1ccc(NC[C@@H](N)CS)cc1-c1ccccc1)C(O)=O Show InChI InChI=1S/C21H27N3O3S2/c1-29-10-9-19(21(26)27)24-20(25)17-8-7-16(23-12-15(22)13-28)11-18(17)14-5-3-2-4-6-14/h2-8,11,15,19,23,28H,9-10,12-13,22H2,1H3,(H,24,25)(H,26,27)/t15-,19+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076811
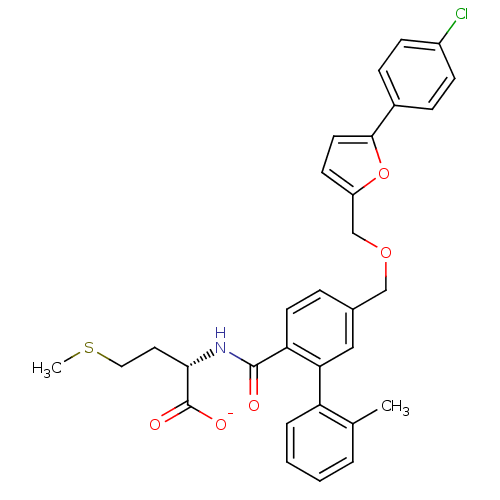
(CHEMBL10877 | Lithium; (S)-2-({5-[5-(4-chloro-phen...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(Cl)cc2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C31H30ClNO5S/c1-20-5-3-4-6-25(20)27-17-21(7-13-26(27)30(34)33-28(31(35)36)15-16-39-2)18-37-19-24-12-14-29(38-24)22-8-10-23(32)11-9-22/h3-14,17,28H,15-16,18-19H2,1-2H3,(H,33,34)(H,35,36)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076809
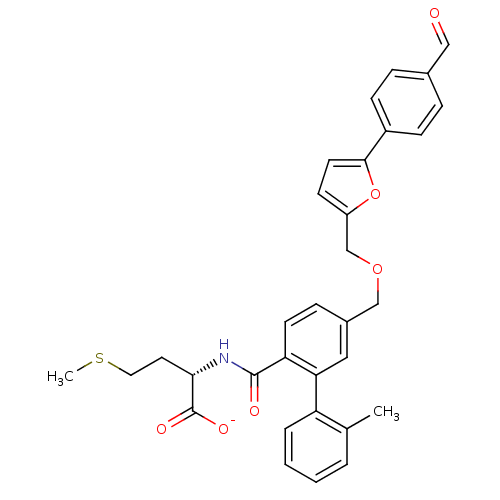
(CHEMBL417606 | Lithium; (S)-2-({5-[5-(4-formyl-phe...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(C=O)cc2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C32H31NO6S/c1-21-5-3-4-6-26(21)28-17-23(9-13-27(28)31(35)33-29(32(36)37)15-16-40-2)19-38-20-25-12-14-30(39-25)24-10-7-22(18-34)8-11-24/h3-14,17-18,29H,15-16,19-20H2,1-2H3,(H,33,35)(H,36,37)/p-1/t29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076798
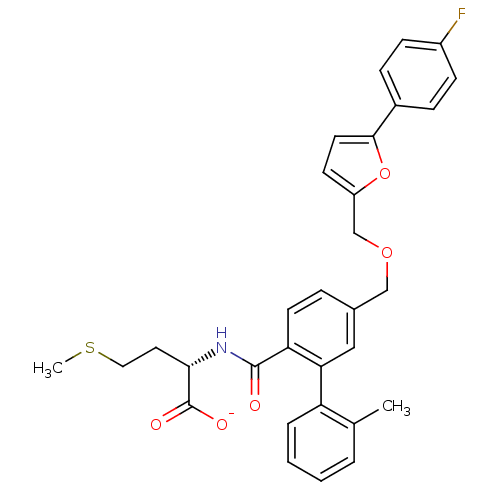
(CHEMBL273697 | Lithium; (S)-2-({5-[5-(4-fluoro-phe...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(F)cc2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C31H30FNO5S/c1-20-5-3-4-6-25(20)27-17-21(7-13-26(27)30(34)33-28(31(35)36)15-16-39-2)18-37-19-24-12-14-29(38-24)22-8-10-23(32)11-9-22/h3-14,17,28H,15-16,18-19H2,1-2H3,(H,33,34)(H,35,36)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068561
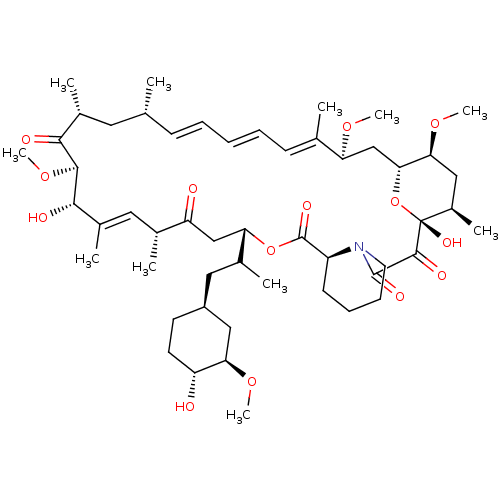
((16E,24E,26E,28E)-1,18-Dihydroxy-12-[2-(4-hydroxy-...)Show SMILES CO[C@@H]1C[C@H](CC(C)[C@@H]2CC(=O)[C@H](C)\C=C(C)\[C@H](O)[C@@H](OC)C(=O)[C@H](C)C[C@H](C)\C=C\C=C\C=C(C)\[C@H](C[C@H]3O[C@](O)([C@H](C)C[C@@H]3OC)C(=O)C(=O)N3CCCC[C@H]3C(=O)O2)OC)CC[C@H]1O |c:14,33,t:29,31| Show InChI InChI=1S/C52H81NO14/c1-30-17-13-12-14-18-31(2)41(62-8)29-45-44(64-10)26-36(7)52(61,67-45)49(58)50(59)53-22-16-15-19-38(53)51(60)66-42(33(4)25-37-20-21-39(54)43(27-37)63-9)28-40(55)32(3)24-35(6)47(57)48(65-11)46(56)34(5)23-30/h12-14,17-18,24,30,32-34,36-39,41-45,47-48,54,57,61H,15-16,19-23,25-29H2,1-11H3/b14-12+,17-13+,31-18+,35-24+/t30-,32-,33?,34-,36-,37+,38+,39-,41+,42+,43-,44+,45-,47+,48+,52-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
UniChem
Similars
| MMDB
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
The compound was tested for binding afifnity to FK506 binding protein 12 with an ascomycin conjugate of alkaline phosphatase in a competition binding... |
Bioorg Med Chem Lett 10: 1405-8 (2000)
BindingDB Entry DOI: 10.7270/Q2CV4GZH |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50089437

(CHEMBL437064 | Rapamycin analogue)Show SMILES CO[C@@H]1C[C@H](C[C@@H](C)[C@@H]2CC(=O)[C@H](C)\C=C(C)\[C@@H](O)[C@@H](OC)C(=O)[C@H](C)C[C@H](C)\C=C\C=C\C=C(C)\[C@H](C[C@@H]3CC[C@@H](C)[C@@](O)(O3)C(=O)C(=O)N3CCCC[C@H]3C(=O)O2)N(O)C(=O)Oc2ccccc2)CC[C@H]1O |c:14,33,t:29,31| Show InChI InChI=1S/C57H82N2O15/c1-34-18-12-10-13-19-35(2)45(59(69)56(67)72-42-20-14-11-15-21-42)32-43-25-23-40(7)57(68,74-43)53(64)54(65)58-27-17-16-22-44(58)55(66)73-48(37(4)30-41-24-26-46(60)49(31-41)70-8)33-47(61)36(3)29-39(6)51(63)52(71-9)50(62)38(5)28-34/h10-15,18-21,29,34,36-38,40-41,43-46,48-49,51-52,60,63,68-69H,16-17,22-28,30-33H2,1-9H3/b13-10+,18-12+,35-19+,39-29+/t34-,36-,37-,38-,40-,41+,43+,44+,45+,46-,48+,49-,51-,52+,57-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
The compound was tested for binding afifnity to FK506 binding protein 12 using Rapamycin as control, with an ascomycin conjugate of alkaline phosphat... |
Bioorg Med Chem Lett 10: 1405-8 (2000)
BindingDB Entry DOI: 10.7270/Q2CV4GZH |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076802
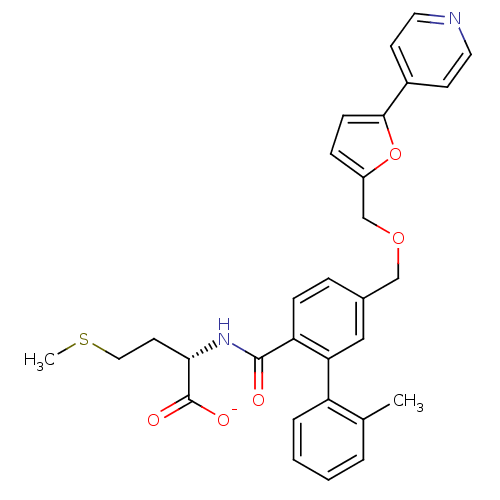
(CHEMBL10912 | Lithium; (S)-2-{[2'-methyl-5-(5-pyri...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccncc2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C30H30N2O5S/c1-20-5-3-4-6-24(20)26-17-21(7-9-25(26)29(33)32-27(30(34)35)13-16-38-2)18-36-19-23-8-10-28(37-23)22-11-14-31-15-12-22/h3-12,14-15,17,27H,13,16,18-19H2,1-2H3,(H,32,33)(H,34,35)/p-1/t27-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076795
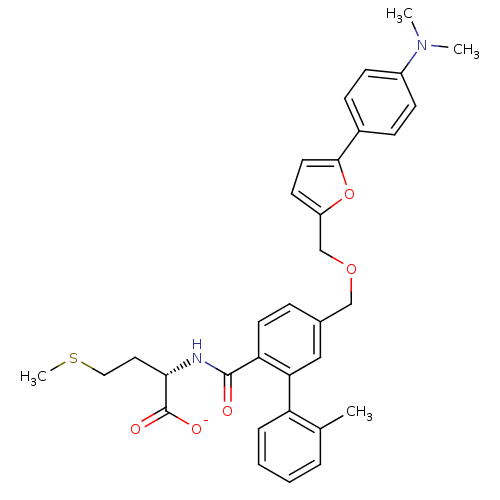
(CHEMBL11210 | Lithium; (S)-2-({5-[5-(4-dimethylami...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(cc2)N(C)C)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C33H36N2O5S/c1-22-7-5-6-8-27(22)29-19-23(9-15-28(29)32(36)34-30(33(37)38)17-18-41-4)20-39-21-26-14-16-31(40-26)24-10-12-25(13-11-24)35(2)3/h5-16,19,30H,17-18,20-21H2,1-4H3,(H,34,36)(H,37,38)/p-1/t30-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076791
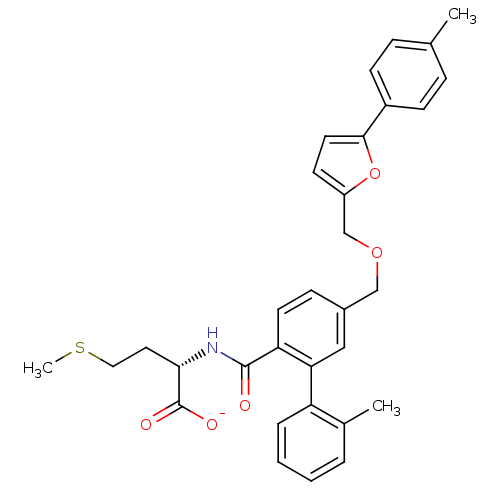
(CHEMBL10990 | Lithium; (S)-4-methylsulfanyl-2-{[2'...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(C)cc2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C32H33NO5S/c1-21-8-11-24(12-9-21)30-15-13-25(38-30)20-37-19-23-10-14-27(28(18-23)26-7-5-4-6-22(26)2)31(34)33-29(32(35)36)16-17-39-3/h4-15,18,29H,16-17,19-20H2,1-3H3,(H,33,34)(H,35,36)/p-1/t29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076794
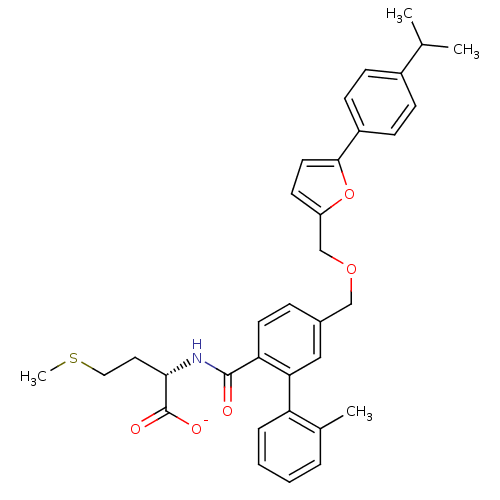
(CHEMBL276602 | Lithium; (S)-2-({5-[5-(4-isopropyl-...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(cc2)C(C)C)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C34H37NO5S/c1-22(2)25-10-12-26(13-11-25)32-16-14-27(40-32)21-39-20-24-9-15-29(30(19-24)28-8-6-5-7-23(28)3)33(36)35-31(34(37)38)17-18-41-4/h5-16,19,22,31H,17-18,20-21H2,1-4H3,(H,35,36)(H,37,38)/p-1/t31-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076810
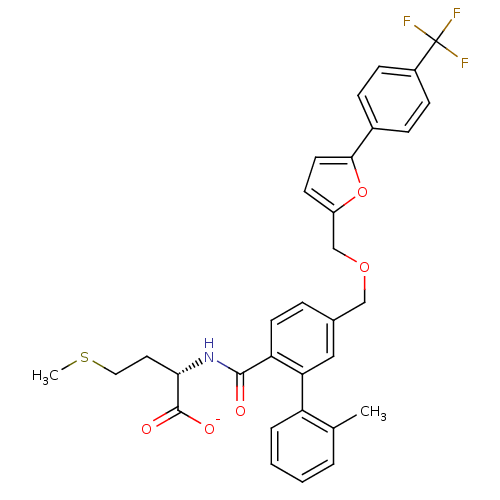
(CHEMBL428001 | Lithium; (S)-4-methylsulfanyl-2-({2...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(cc2)C(F)(F)F)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C32H30F3NO5S/c1-20-5-3-4-6-25(20)27-17-21(7-13-26(27)30(37)36-28(31(38)39)15-16-42-2)18-40-19-24-12-14-29(41-24)22-8-10-23(11-9-22)32(33,34)35/h3-14,17,28H,15-16,18-19H2,1-2H3,(H,36,37)(H,38,39)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
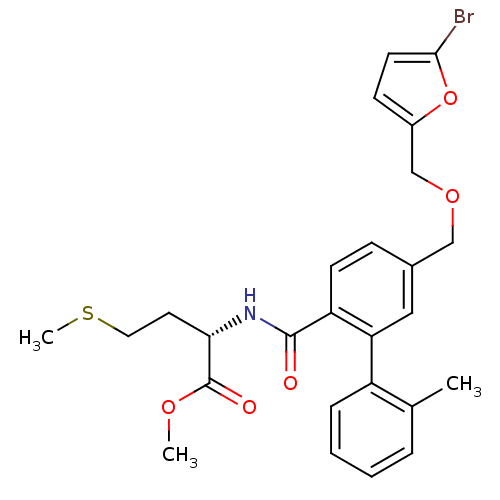
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076799
((S)-2-{[5-(5-Bromo-furan-2-ylmethoxymethyl)-2'-met...)Show SMILES COC(=O)[C@H](CCSC)NC(=O)c1ccc(COCc2ccc(Br)o2)cc1-c1ccccc1C Show InChI InChI=1S/C26H28BrNO5S/c1-17-6-4-5-7-20(17)22-14-18(15-32-16-19-9-11-24(27)33-19)8-10-21(22)25(29)28-23(12-13-34-3)26(30)31-2/h4-11,14,23H,12-13,15-16H2,1-3H3,(H,28,29)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
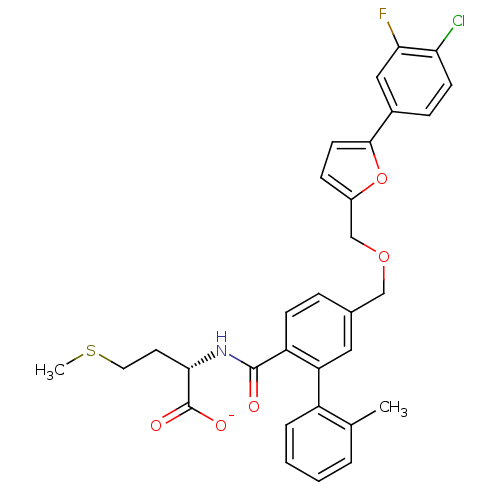
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076792
(CHEMBL10812 | Lithium; (S)-2-({5-[5-(4-chloro-3-fl...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(Cl)c(F)c2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C31H29ClFNO5S/c1-19-5-3-4-6-23(19)25-15-20(7-10-24(25)30(35)34-28(31(36)37)13-14-40-2)17-38-18-22-9-12-29(39-22)21-8-11-26(32)27(33)16-21/h3-12,15-16,28H,13-14,17-18H2,1-2H3,(H,34,35)(H,36,37)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076801
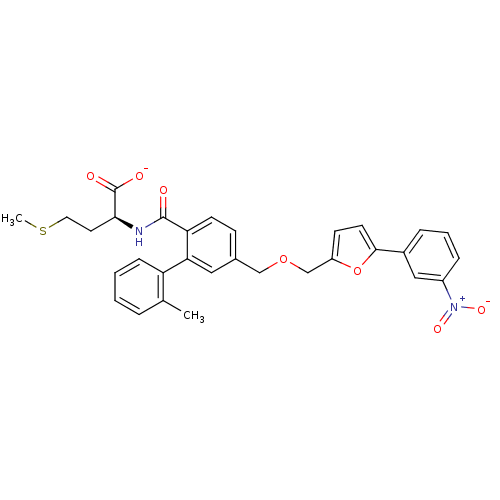
(CHEMBL430096 | Lithium; (S)-2-({2'-methyl-5-[5-(3-...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2cccc(c2)[N+]([O-])=O)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C31H30N2O7S/c1-20-6-3-4-9-25(20)27-16-21(10-12-26(27)30(34)32-28(31(35)36)14-15-41-2)18-39-19-24-11-13-29(40-24)22-7-5-8-23(17-22)33(37)38/h3-13,16-17,28H,14-15,18-19H2,1-2H3,(H,32,34)(H,35,36)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076797
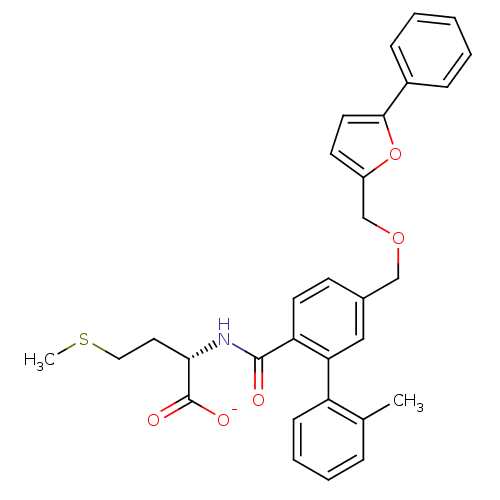
(CHEMBL10955 | Lithium; (S)-2-{[2'-methyl-5-(5-phen...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccccc2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C31H31NO5S/c1-21-8-6-7-11-25(21)27-18-22(12-14-26(27)30(33)32-28(31(34)35)16-17-38-2)19-36-20-24-13-15-29(37-24)23-9-4-3-5-10-23/h3-15,18,28H,16-17,19-20H2,1-2H3,(H,32,33)(H,34,35)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076793
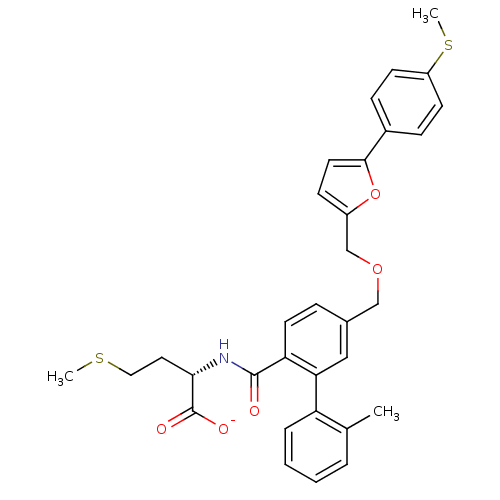
(CHEMBL11003 | Lithium; (S)-2-({2'-methyl-5-[5-(4-m...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(SC)cc2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C32H33NO5S2/c1-21-6-4-5-7-26(21)28-18-22(8-14-27(28)31(34)33-29(32(35)36)16-17-39-2)19-37-20-24-11-15-30(38-24)23-9-12-25(40-3)13-10-23/h4-15,18,29H,16-17,19-20H2,1-3H3,(H,33,34)(H,35,36)/p-1/t29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 6.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076803
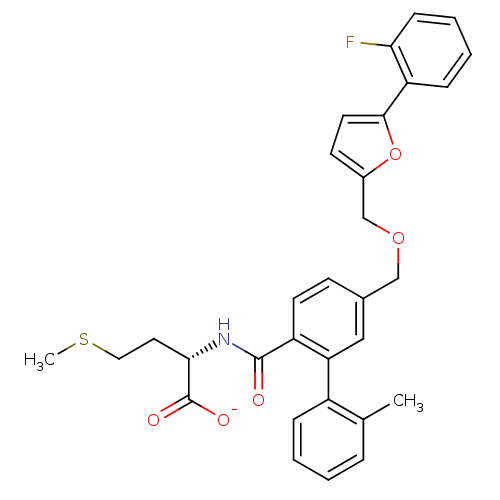
(CHEMBL10970 | Lithium; (S)-2-({5-[5-(2-fluoro-phen...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccccc2F)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C31H30FNO5S/c1-20-7-3-4-8-23(20)26-17-21(11-13-24(26)30(34)33-28(31(35)36)15-16-39-2)18-37-19-22-12-14-29(38-22)25-9-5-6-10-27(25)32/h3-14,17,28H,15-16,18-19H2,1-2H3,(H,33,34)(H,35,36)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076790
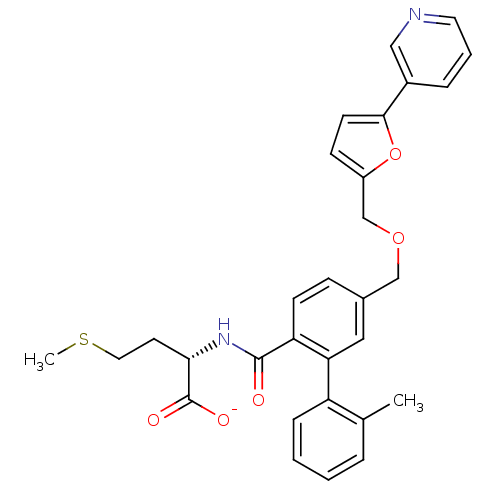
(CHEMBL10902 | Lithium; (S)-2-{[2'-methyl-5-(5-pyri...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2cccnc2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C30H30N2O5S/c1-20-6-3-4-8-24(20)26-16-21(9-11-25(26)29(33)32-27(30(34)35)13-15-38-2)18-36-19-23-10-12-28(37-23)22-7-5-14-31-17-22/h3-12,14,16-17,27H,13,15,18-19H2,1-2H3,(H,32,33)(H,34,35)/p-1/t27-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 6.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076800
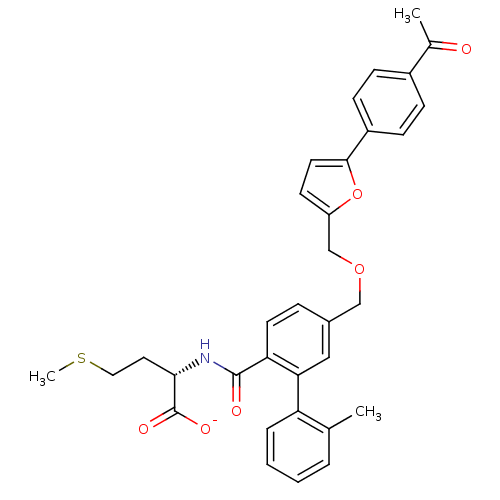
(CHEMBL430307 | Lithium; (S)-2-({5-[5-(4-acetyl-phe...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(cc2)C(C)=O)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C33H33NO6S/c1-21-6-4-5-7-27(21)29-18-23(8-14-28(29)32(36)34-30(33(37)38)16-17-41-3)19-39-20-26-13-15-31(40-26)25-11-9-24(10-12-25)22(2)35/h4-15,18,30H,16-17,19-20H2,1-3H3,(H,34,36)(H,37,38)/p-1/t30-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 6.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076788
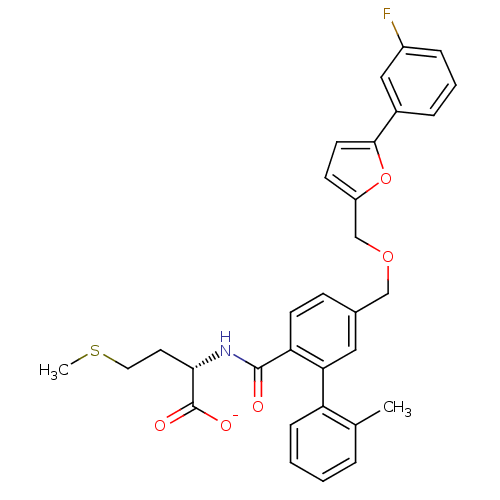
(CHEMBL11195 | Lithium; (S)-2-({5-[5-(3-fluoro-phen...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2cccc(F)c2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C31H30FNO5S/c1-20-6-3-4-9-25(20)27-16-21(10-12-26(27)30(34)33-28(31(35)36)14-15-39-2)18-37-19-24-11-13-29(38-24)22-7-5-8-23(32)17-22/h3-13,16-17,28H,14-15,18-19H2,1-2H3,(H,33,34)(H,35,36)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 7.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076806
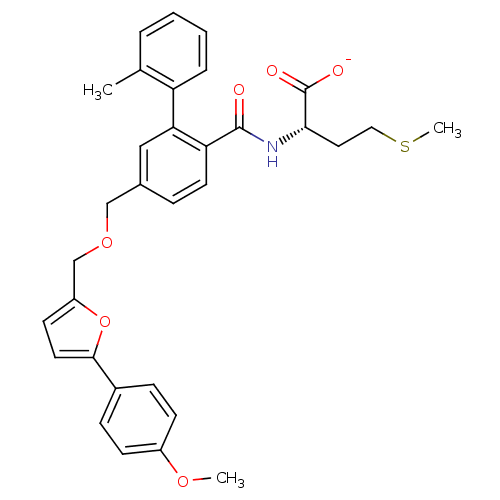
(CHEMBL273265 | Lithium; (S)-2-({5-[5-(4-methoxy-ph...)Show SMILES COc1ccc(cc1)-c1ccc(COCc2ccc(C(=O)N[C@@H](CCSC)C([O-])=O)c(c2)-c2ccccc2C)o1 Show InChI InChI=1S/C32H33NO6S/c1-21-6-4-5-7-26(21)28-18-22(8-14-27(28)31(34)33-29(32(35)36)16-17-40-3)19-38-20-25-13-15-30(39-25)23-9-11-24(37-2)12-10-23/h4-15,18,29H,16-17,19-20H2,1-3H3,(H,33,34)(H,35,36)/p-1/t29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 8.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076804
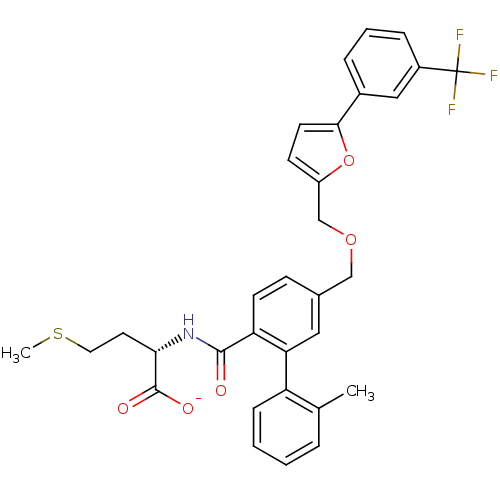
(CHEMBL273507 | Lithium; (S)-4-methylsulfanyl-2-({2...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2cccc(c2)C(F)(F)F)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C32H30F3NO5S/c1-20-6-3-4-9-25(20)27-16-21(10-12-26(27)30(37)36-28(31(38)39)14-15-42-2)18-40-19-24-11-13-29(41-24)22-7-5-8-23(17-22)32(33,34)35/h3-13,16-17,28H,14-15,18-19H2,1-2H3,(H,36,37)(H,38,39)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 8.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076789
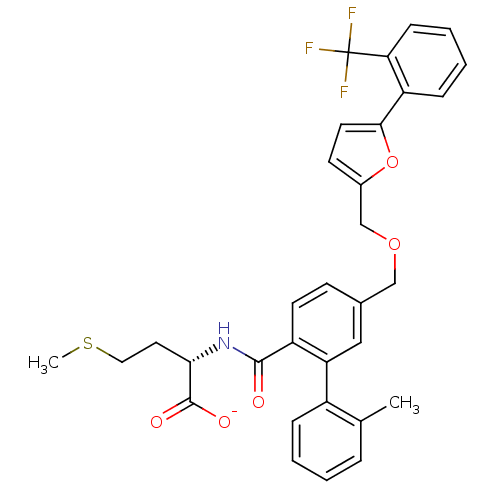
(CHEMBL274672 | Lithium; (S)-4-methylsulfanyl-2-({2...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccccc2C(F)(F)F)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C32H30F3NO5S/c1-20-7-3-4-8-23(20)26-17-21(11-13-24(26)30(37)36-28(31(38)39)15-16-42-2)18-40-19-22-12-14-29(41-22)25-9-5-6-10-27(25)32(33,34)35/h3-14,17,28H,15-16,18-19H2,1-2H3,(H,36,37)(H,38,39)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 9.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076807
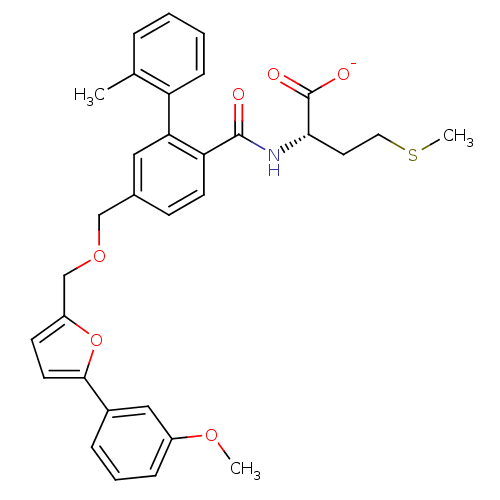
(CHEMBL273721 | Lithium; (S)-2-({5-[5-(3-methoxy-ph...)Show SMILES COc1cccc(c1)-c1ccc(COCc2ccc(C(=O)N[C@@H](CCSC)C([O-])=O)c(c2)-c2ccccc2C)o1 Show InChI InChI=1S/C32H33NO6S/c1-21-7-4-5-10-26(21)28-17-22(11-13-27(28)31(34)33-29(32(35)36)15-16-40-3)19-38-20-25-12-14-30(39-25)23-8-6-9-24(18-23)37-2/h4-14,17-18,29H,15-16,19-20H2,1-3H3,(H,33,34)(H,35,36)/p-1/t29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50089447
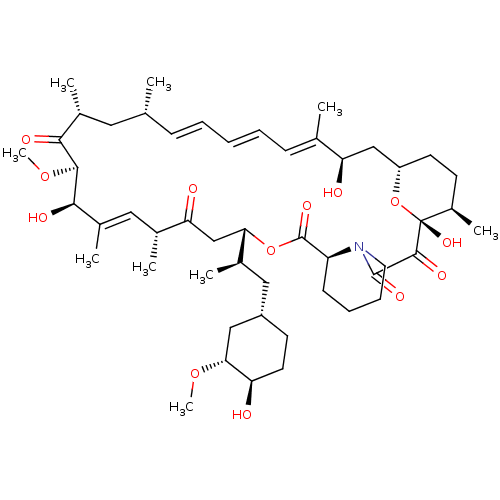
(CHEMBL281297 | Rapamycin analogue)Show SMILES CO[C@@H]1C[C@H](C[C@@H](C)[C@@H]2CC(=O)[C@H](C)\C=C(C)\[C@@H](O)[C@@H](OC)C(=O)[C@H](C)C[C@H](C)\C=C\C=C\C=C(C)\[C@H](O)C[C@@H]3CC[C@@H](C)[C@@](O)(O3)C(=O)C(=O)N3CCCC[C@H]3C(=O)O2)CC[C@H]1O |c:14,33,t:29,31| Show InChI InChI=1S/C50H77NO13/c1-29-15-11-10-12-16-30(2)40(53)27-37-20-18-35(7)50(60,64-37)47(57)48(58)51-22-14-13-17-38(51)49(59)63-42(32(4)25-36-19-21-39(52)43(26-36)61-8)28-41(54)31(3)24-34(6)45(56)46(62-9)44(55)33(5)23-29/h10-12,15-16,24,29,31-33,35-40,42-43,45-46,52-53,56,60H,13-14,17-23,25-28H2,1-9H3/b12-10+,15-11+,30-16+,34-24+/t29-,31-,32-,33-,35-,36+,37+,38+,39-,40-,42+,43-,45-,46+,50-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
The compound was tested for binding afifnity to FK506 binding protein 12 using Rapamycin as control, with an ascomycin conjugate of alkaline phosphat... |
Bioorg Med Chem Lett 10: 1405-8 (2000)
BindingDB Entry DOI: 10.7270/Q2CV4GZH |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076808
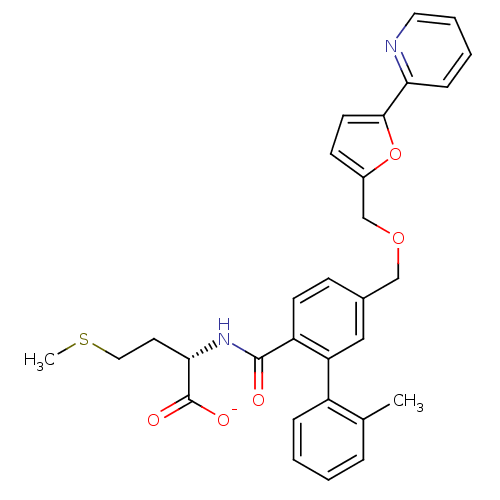
(CHEMBL10762 | Lithium; (S)-2-{[2'-methyl-5-(5-pyri...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccccn2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C30H30N2O5S/c1-20-7-3-4-8-23(20)25-17-21(10-12-24(25)29(33)32-27(30(34)35)14-16-38-2)18-36-19-22-11-13-28(37-22)26-9-5-6-15-31-26/h3-13,15,17,27H,14,16,18-19H2,1-2H3,(H,32,33)(H,34,35)/p-1/t27-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076813
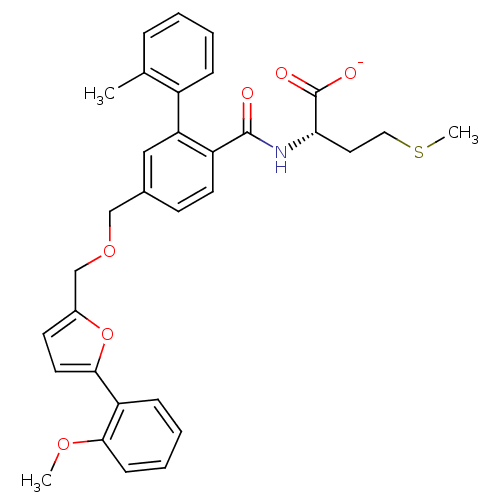
(CHEMBL10687 | Lithium; (S)-2-({5-[5-(2-methoxy-phe...)Show SMILES COc1ccccc1-c1ccc(COCc2ccc(C(=O)N[C@@H](CCSC)C([O-])=O)c(c2)-c2ccccc2C)o1 Show InChI InChI=1S/C32H33NO6S/c1-21-8-4-5-9-24(21)27-18-22(12-14-25(27)31(34)33-28(32(35)36)16-17-40-3)19-38-20-23-13-15-30(39-23)26-10-6-7-11-29(26)37-2/h4-15,18,28H,16-17,19-20H2,1-3H3,(H,33,34)(H,35,36)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076812
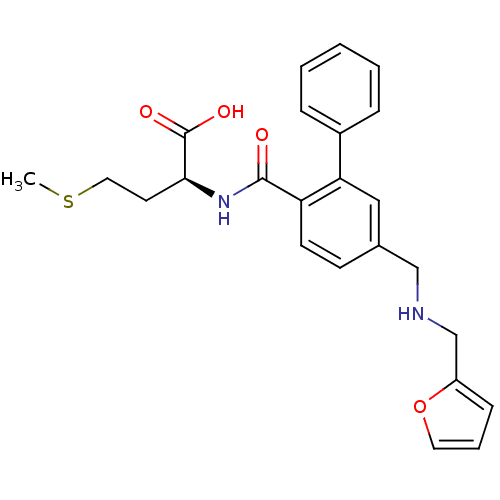
((S)-2-[(5-{[(Furan-2-ylmethyl)-amino]-methyl}-biph...)Show SMILES CSCC[C@H](NC(=O)c1ccc(CNCc2ccco2)cc1-c1ccccc1)C(O)=O Show InChI InChI=1S/C24H26N2O4S/c1-31-13-11-22(24(28)29)26-23(27)20-10-9-17(15-25-16-19-8-5-12-30-19)14-21(20)18-6-3-2-4-7-18/h2-10,12,14,22,25H,11,13,15-16H2,1H3,(H,26,27)(H,28,29)/t22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 560 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50089429
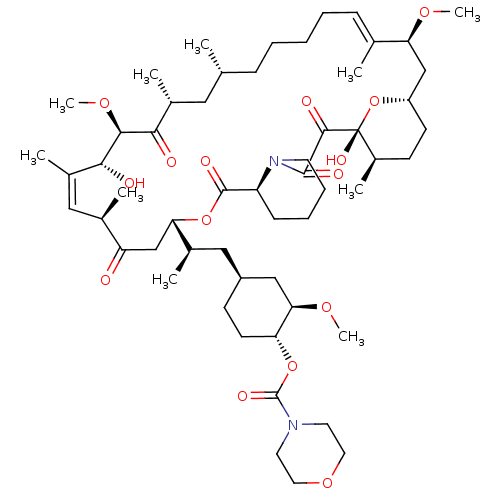
(4-[2-(1,18-dihydroxy-19,30-dimethoxy-15,17,21,23,2...)Show SMILES CO[C@@H]1C[C@H](C[C@@H](C)[C@@H]2CC(=O)[C@H](C)\C=C(C)\[C@@H](O)[C@@H](OC)C(=O)[C@H](C)C[C@H](C)CCCC\C=C(C)\[C@H](C[C@@H]3CC[C@@H](C)[C@@](O)(O3)C(=O)C(=O)N3CCCC[C@H]3C(=O)O2)OC)CC[C@H]1OC(=O)N1CCOCC1 |c:14,33| Show InChI InChI=1S/C56H90N2O15/c1-34-16-12-11-13-17-35(2)46(67-8)32-42-21-19-40(7)56(66,73-42)52(62)53(63)58-23-15-14-18-43(58)54(64)71-47(33-44(59)36(3)29-39(6)50(61)51(69-10)49(60)38(5)28-34)37(4)30-41-20-22-45(48(31-41)68-9)72-55(65)57-24-26-70-27-25-57/h17,29,34,36-38,40-43,45-48,50-51,61,66H,11-16,18-28,30-33H2,1-10H3/b35-17+,39-29+/t34-,36-,37-,38-,40-,41+,42+,43+,45-,46+,47+,48-,50-,51+,56-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
The compound was tested for binding afifnity to FK506 binding protein 12 using Rapamycin as control, with an ascomycin conjugate of alkaline phosphat... |
Bioorg Med Chem Lett 10: 1405-8 (2000)
BindingDB Entry DOI: 10.7270/Q2CV4GZH |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50089432
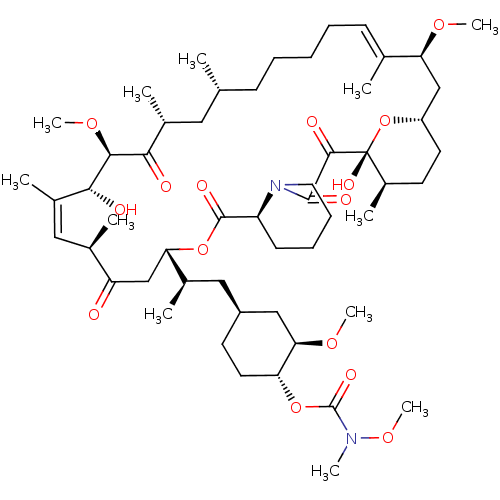
(1,18-dihydroxy-19,30-dimethoxy-12-{2-[3-methoxy-4-...)Show SMILES CO[C@@H]1C[C@H](C[C@@H](C)[C@@H]2CC(=O)[C@H](C)\C=C(C)\[C@@H](O)[C@@H](OC)C(=O)[C@H](C)C[C@H](C)CCCC\C=C(C)\[C@H](C[C@@H]3CC[C@@H](C)[C@@](O)(O3)C(=O)C(=O)N3CCCC[C@H]3C(=O)O2)OC)CC[C@H]1OC(=O)N(C)OC |c:14,33| Show InChI InChI=1S/C54H88N2O15/c1-32-18-14-13-15-19-33(2)44(65-9)30-40-23-21-38(7)54(64,71-40)50(60)51(61)56-25-17-16-20-41(56)52(62)69-45(31-42(57)34(3)27-37(6)48(59)49(67-11)47(58)36(5)26-32)35(4)28-39-22-24-43(46(29-39)66-10)70-53(63)55(8)68-12/h19,27,32,34-36,38-41,43-46,48-49,59,64H,13-18,20-26,28-31H2,1-12H3/b33-19+,37-27+/t32-,34-,35-,36-,38-,39+,40+,41+,43-,44+,45+,46-,48-,49+,54-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
The compound was tested for binding afifnity to FK506 binding protein 12 using Rapamycin as control, with an ascomycin conjugate of alkaline phosphat... |
Bioorg Med Chem Lett 10: 1405-8 (2000)
BindingDB Entry DOI: 10.7270/Q2CV4GZH |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076805
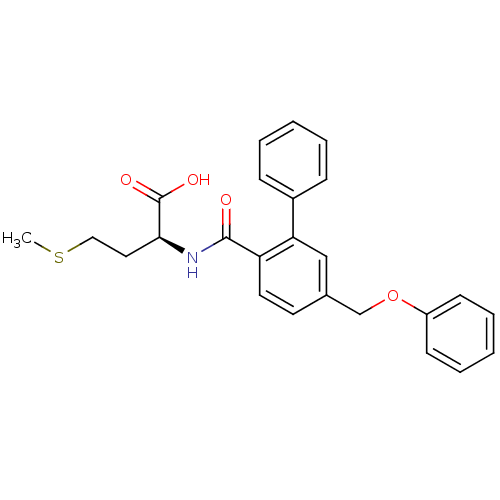
((S)-4-Methylsulfanyl-2-[(5-phenoxymethyl-biphenyl-...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COc2ccccc2)cc1-c1ccccc1)C(O)=O Show InChI InChI=1S/C25H25NO4S/c1-31-15-14-23(25(28)29)26-24(27)21-13-12-18(17-30-20-10-6-3-7-11-20)16-22(21)19-8-4-2-5-9-19/h2-13,16,23H,14-15,17H2,1H3,(H,26,27)(H,28,29)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity of the compound was evaluated against farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076793

(CHEMBL11003 | Lithium; (S)-2-({2'-methyl-5-[5-(4-m...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(SC)cc2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C32H33NO5S2/c1-21-6-4-5-7-26(21)28-18-22(8-14-27(28)31(34)33-29(32(35)36)16-17-39-2)19-37-20-24-11-15-30(38-24)23-9-12-25(40-3)13-10-23/h4-15,18,29H,16-17,19-20H2,1-3H3,(H,33,34)(H,35,36)/p-1/t29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 10 | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
EC50 is measured as inhibition of ras processing in a whole cell assay for farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076794

(CHEMBL276602 | Lithium; (S)-2-({5-[5-(4-isopropyl-...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(cc2)C(C)C)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C34H37NO5S/c1-22(2)25-10-12-26(13-11-25)32-16-14-27(40-32)21-39-20-24-9-15-29(30(19-24)28-8-6-5-7-23(28)3)33(36)35-31(34(37)38)17-18-41-4/h5-16,19,22,31H,17-18,20-21H2,1-4H3,(H,35,36)(H,37,38)/p-1/t31-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 0.780 | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
EC50 is measured as inhibition of ras processing in a whole cell assay for farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076788

(CHEMBL11195 | Lithium; (S)-2-({5-[5-(3-fluoro-phen...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2cccc(F)c2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C31H30FNO5S/c1-20-6-3-4-9-25(20)27-16-21(10-12-26(27)30(34)33-28(31(35)36)14-15-39-2)18-37-19-24-11-13-29(38-24)22-7-5-8-23(32)17-22/h3-13,16-17,28H,14-15,18-19H2,1-2H3,(H,33,34)(H,35,36)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 10 | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
EC50 is measured as inhibition of ras processing in a whole cell assay for farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076798

(CHEMBL273697 | Lithium; (S)-2-({5-[5-(4-fluoro-phe...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(F)cc2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C31H30FNO5S/c1-20-5-3-4-6-25(20)27-17-21(7-13-26(27)30(34)33-28(31(35)36)15-16-39-2)18-37-19-24-12-14-29(38-24)22-8-10-23(32)11-9-22/h3-14,17,28H,15-16,18-19H2,1-2H3,(H,33,34)(H,35,36)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
EC50 is measured as inhibition of ras processing in a whole cell assay for farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076809

(CHEMBL417606 | Lithium; (S)-2-({5-[5-(4-formyl-phe...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(C=O)cc2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C32H31NO6S/c1-21-5-3-4-6-26(21)28-17-23(9-13-27(28)31(35)33-29(32(36)37)15-16-40-2)19-38-20-25-12-14-30(39-25)24-10-7-22(18-34)8-11-24/h3-14,17-18,29H,15-16,19-20H2,1-2H3,(H,33,35)(H,36,37)/p-1/t29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | >1 | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
EC50 is measured as inhibition of ras processing in a whole cell assay for farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076803

(CHEMBL10970 | Lithium; (S)-2-({5-[5-(2-fluoro-phen...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccccc2F)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C31H30FNO5S/c1-20-7-3-4-8-23(20)26-17-21(11-13-24(26)30(34)33-28(31(35)36)15-16-39-2)18-37-19-22-12-14-29(38-22)25-9-5-6-10-27(25)32/h3-14,17,28H,15-16,18-19H2,1-2H3,(H,33,34)(H,35,36)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | >1 | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
EC50 is measured as inhibition of ras processing in a whole cell assay for farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076791

(CHEMBL10990 | Lithium; (S)-4-methylsulfanyl-2-{[2'...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(C)cc2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C32H33NO5S/c1-21-8-11-24(12-9-21)30-15-13-25(38-30)20-37-19-23-10-14-27(28(18-23)26-7-5-4-6-22(26)2)31(34)33-29(32(35)36)16-17-39-3/h4-15,18,29H,16-17,19-20H2,1-3H3,(H,33,34)(H,35,36)/p-1/t29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 1.80 | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
EC50 is measured as inhibition of ras processing in a whole cell assay for farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076813

(CHEMBL10687 | Lithium; (S)-2-({5-[5-(2-methoxy-phe...)Show SMILES COc1ccccc1-c1ccc(COCc2ccc(C(=O)N[C@@H](CCSC)C([O-])=O)c(c2)-c2ccccc2C)o1 Show InChI InChI=1S/C32H33NO6S/c1-21-8-4-5-9-24(21)27-18-22(12-14-25(27)31(34)33-28(32(35)36)16-17-40-3)19-38-20-23-13-15-30(39-23)26-10-6-7-11-29(26)37-2/h4-15,18,28H,16-17,19-20H2,1-3H3,(H,33,34)(H,35,36)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | >1 | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
EC50 is measured as inhibition of ras processing in a whole cell assay for farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076807

(CHEMBL273721 | Lithium; (S)-2-({5-[5-(3-methoxy-ph...)Show SMILES COc1cccc(c1)-c1ccc(COCc2ccc(C(=O)N[C@@H](CCSC)C([O-])=O)c(c2)-c2ccccc2C)o1 Show InChI InChI=1S/C32H33NO6S/c1-21-7-4-5-10-26(21)28-17-22(11-13-27(28)31(34)33-29(32(35)36)15-16-40-3)19-38-20-25-12-14-30(39-25)23-8-6-9-24(18-23)37-2/h4-14,17-18,29H,15-16,19-20H2,1-3H3,(H,33,34)(H,35,36)/p-1/t29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | >1 | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
EC50 is measured as inhibition of ras processing in a whole cell assay for farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076808

(CHEMBL10762 | Lithium; (S)-2-{[2'-methyl-5-(5-pyri...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccccn2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C30H30N2O5S/c1-20-7-3-4-8-23(20)25-17-21(10-12-24(25)29(33)32-27(30(34)35)14-16-38-2)18-36-19-22-11-13-28(37-22)26-9-5-6-15-31-26/h3-13,15,17,27H,14,16,18-19H2,1-2H3,(H,32,33)(H,34,35)/p-1/t27-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | >0.300 | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
EC50 is measured as inhibition of ras processing in a whole cell assay for farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076810

(CHEMBL428001 | Lithium; (S)-4-methylsulfanyl-2-({2...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccc(cc2)C(F)(F)F)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C32H30F3NO5S/c1-20-5-3-4-6-25(20)27-17-21(7-13-26(27)30(37)36-28(31(38)39)15-16-42-2)18-40-19-24-12-14-29(41-24)22-8-10-23(11-9-22)32(33,34)35/h3-14,17,28H,15-16,18-19H2,1-2H3,(H,36,37)(H,38,39)/p-1/t28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
EC50 is measured as inhibition of ras processing in a whole cell assay for farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |
Protein farnesyltransferase subunit beta/geranylgeranyltransferase type-1 subunit alpha
(Homo sapiens (Human)) | BDBM50076802

(CHEMBL10912 | Lithium; (S)-2-{[2'-methyl-5-(5-pyri...)Show SMILES CSCC[C@H](NC(=O)c1ccc(COCc2ccc(o2)-c2ccncc2)cc1-c1ccccc1C)C([O-])=O Show InChI InChI=1S/C30H30N2O5S/c1-20-5-3-4-6-24(20)26-17-21(7-9-25(26)29(33)32-27(30(34)35)13-16-38-2)18-36-19-23-8-10-28(37-23)22-11-14-31-15-12-22/h3-12,14-15,17,27H,13,16,18-19H2,1-2H3,(H,32,33)(H,34,35)/p-1/t27-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | >0.300 | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
EC50 is measured as inhibition of ras processing in a whole cell assay for farnesyltransferase (FTase) |
Bioorg Med Chem Lett 9: 1069-74 (1999)
BindingDB Entry DOI: 10.7270/Q2X34WNT |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data