 Found 697 hits with Last Name = 'zheng' and Initial = 'f'
Found 697 hits with Last Name = 'zheng' and Initial = 'f' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Lanosterol synthase
(Rattus norvegicus) | BDBM50055641

(2,2-dimethyl-3-((3E,7E,11E)-3,7,12-trimethyl-14-(6...)Show SMILES [#6]-[#6](-[#6]-[#6]\[#6]=[#6](\[#6])-[#6])-[#16]-[#6]-[#6]\[#6](-[#6])=[#6]\[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]-[#6]1-[#8]C1([#6])[#6] Show InChI InChI=1S/C29H50OS/c1-23(2)13-11-18-27(6)31-22-21-26(5)15-10-9-14-24(3)16-12-17-25(4)19-20-28-29(7,8)30-28/h13-15,17,27-28H,9-12,16,18-22H2,1-8H3/b24-14+,25-17+,26-15+ | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Simon Fraser University
Curated by ChEMBL
| Assay Description
Compound was tested for kinetic inhibition constant against Oxidosqualene-lanosterol cyclase from rat liver |
J Med Chem 40: 201-9 (1997)
Article DOI: 10.1021/jm960483a
BindingDB Entry DOI: 10.7270/Q2P26X73 |
More data for this
Ligand-Target Pair | |
Lanosterol synthase
(Rattus norvegicus) | BDBM50055642

(2,2-Dimethyl-3-[(3E,7E,11E)-3,7,12-trimethyl-15-(4...)Show SMILES [#6]\[#6](-[#6])=[#6]\[#6]-[#6]-[#16]-[#6]-[#6]-[#6]\[#6](-[#6])=[#6]\[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]-[#6]1-[#8]C1([#6])[#6] Show InChI InChI=1S/C28H48OS/c1-23(2)13-11-21-30-22-12-18-25(4)15-9-8-14-24(3)16-10-17-26(5)19-20-27-28(6,7)29-27/h13-15,17,27H,8-12,16,18-22H2,1-7H3/b24-14+,25-15+,26-17+ | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Simon Fraser University
Curated by ChEMBL
| Assay Description
Compound was tested for kinetic inhibition constant against Oxidosqualene-lanosterol cyclase from rat liver |
J Med Chem 40: 201-9 (1997)
Article DOI: 10.1021/jm960483a
BindingDB Entry DOI: 10.7270/Q2P26X73 |
More data for this
Ligand-Target Pair | |
Lanosterol synthase
(Rattus norvegicus) | BDBM50055643

(2,2-Dimethyl-3-[(E)-3-methyl-7-((3E,7E)-4,8,12-tri...)Show SMILES [#6]-[#6](-[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]-[#6]1-[#8]C1([#6])[#6])-[#16]-[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]\[#6]=[#6](\[#6])-[#6] Show InChI InChI=1S/C29H50OS/c1-23(2)13-9-14-24(3)15-10-16-25(4)18-12-22-31-27(6)19-11-17-26(5)20-21-28-29(7,8)30-28/h13,15,17-18,27-28H,9-12,14,16,19-22H2,1-8H3/b24-15+,25-18+,26-17+ | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Simon Fraser University
Curated by ChEMBL
| Assay Description
Compound was tested for kinetic inhibition constant against Oxidosqualene-lanosterol cyclase from pig liver |
J Med Chem 40: 201-9 (1997)
Article DOI: 10.1021/jm960483a
BindingDB Entry DOI: 10.7270/Q2P26X73 |
More data for this
Ligand-Target Pair | |
Lanosterol synthase
(Rattus norvegicus) | BDBM50055642

(2,2-Dimethyl-3-[(3E,7E,11E)-3,7,12-trimethyl-15-(4...)Show SMILES [#6]\[#6](-[#6])=[#6]\[#6]-[#6]-[#16]-[#6]-[#6]-[#6]\[#6](-[#6])=[#6]\[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]-[#6]1-[#8]C1([#6])[#6] Show InChI InChI=1S/C28H48OS/c1-23(2)13-11-21-30-22-12-18-25(4)15-9-8-14-24(3)16-10-17-26(5)19-20-27-28(6,7)29-27/h13-15,17,27H,8-12,16,18-22H2,1-7H3/b24-14+,25-15+,26-17+ | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Simon Fraser University
Curated by ChEMBL
| Assay Description
Compound was tested for kinetic inhibition constant against Oxidosqualene-lanosterol cyclase from pig liver |
J Med Chem 40: 201-9 (1997)
Article DOI: 10.1021/jm960483a
BindingDB Entry DOI: 10.7270/Q2P26X73 |
More data for this
Ligand-Target Pair | |
Lanosterol synthase
(Rattus norvegicus) | BDBM50055641

(2,2-dimethyl-3-((3E,7E,11E)-3,7,12-trimethyl-14-(6...)Show SMILES [#6]-[#6](-[#6]-[#6]\[#6]=[#6](\[#6])-[#6])-[#16]-[#6]-[#6]\[#6](-[#6])=[#6]\[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]-[#6]1-[#8]C1([#6])[#6] Show InChI InChI=1S/C29H50OS/c1-23(2)13-11-18-27(6)31-22-21-26(5)15-10-9-14-24(3)16-12-17-25(4)19-20-28-29(7,8)30-28/h13-15,17,27-28H,9-12,16,18-22H2,1-8H3/b24-14+,25-17+,26-15+ | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Simon Fraser University
Curated by ChEMBL
| Assay Description
Compound was tested for kinetic inhibition constant against Oxidosqualene-lanosterol cyclase from pig liver |
J Med Chem 40: 201-9 (1997)
Article DOI: 10.1021/jm960483a
BindingDB Entry DOI: 10.7270/Q2P26X73 |
More data for this
Ligand-Target Pair | |
Lanosterol synthase
(Rattus norvegicus) | BDBM50055644

(2-Methyl-3-((7E,11E,15Z)-3,7,12,20-tetramethyl-16-...)Show SMILES [#6]-[#6](-[#6]-[#6]-[#6]\[#6](-[#6])=[#6]\[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]\[#6]=[#6](\[#6]-[#6]\[#6]=[#6](/[#6])-[#6])-[#6]=[#6])-[#6]-[#6]-[#6]-1-[#8]-[#6]-1-[#6] Show InChI InChI=1S/C30H50O/c1-8-29(20-11-14-24(2)3)21-13-19-26(5)16-10-9-15-25(4)17-12-18-27(6)22-23-30-28(7)31-30/h8,14-16,21,27-28,30H,1,9-13,17-20,22-23H2,2-7H3/b25-15+,26-16+,29-21+ | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Simon Fraser University
Curated by ChEMBL
| Assay Description
Compound was tested for kinetic inhibition constant against Oxidosqualene-lanosterol cyclase from rat liver |
J Med Chem 40: 201-9 (1997)
Article DOI: 10.1021/jm960483a
BindingDB Entry DOI: 10.7270/Q2P26X73 |
More data for this
Ligand-Target Pair | |
Lanosterol synthase
(Rattus norvegicus) | BDBM50055644

(2-Methyl-3-((7E,11E,15Z)-3,7,12,20-tetramethyl-16-...)Show SMILES [#6]-[#6](-[#6]-[#6]-[#6]\[#6](-[#6])=[#6]\[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]\[#6]=[#6](\[#6]-[#6]\[#6]=[#6](/[#6])-[#6])-[#6]=[#6])-[#6]-[#6]-[#6]-1-[#8]-[#6]-1-[#6] Show InChI InChI=1S/C30H50O/c1-8-29(20-11-14-24(2)3)21-13-19-26(5)16-10-9-15-25(4)17-12-18-27(6)22-23-30-28(7)31-30/h8,14-16,21,27-28,30H,1,9-13,17-20,22-23H2,2-7H3/b25-15+,26-16+,29-21+ | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Simon Fraser University
Curated by ChEMBL
| Assay Description
Compound was tested for kinetic inhibition constant against Oxidosqualene-lanosterol cyclase from pig liver |
J Med Chem 40: 201-9 (1997)
Article DOI: 10.1021/jm960483a
BindingDB Entry DOI: 10.7270/Q2P26X73 |
More data for this
Ligand-Target Pair | |
Lanosterol synthase
(Rattus norvegicus) | BDBM50055640

(3-[(3E,7E)-11-((E)-4,8-Dimethyl-nona-3,7-dienylsul...)Show SMILES [#6]\[#6](-[#6])=[#6]\[#6]-[#6]\[#6](-[#6])=[#6]\[#6]-[#6]-[#16]-[#6]-[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]-[#6]1-[#8]C1([#6])[#6] Show InChI InChI=1S/C28H48OS/c1-23(2)13-10-15-25(4)18-12-22-30-21-9-8-14-24(3)16-11-17-26(5)19-20-27-28(6,7)29-27/h13-14,17-18,27H,8-12,15-16,19-22H2,1-7H3/b24-14+,25-18+,26-17+ | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Simon Fraser University
Curated by ChEMBL
| Assay Description
Compound was tested for kinetic inhibition constant against Oxidosqualene-lanosterol cyclase from pig liver |
J Med Chem 40: 201-9 (1997)
Article DOI: 10.1021/jm960483a
BindingDB Entry DOI: 10.7270/Q2P26X73 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50553436
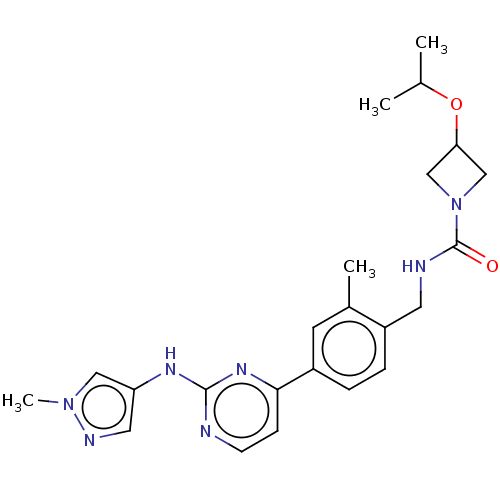
(CHEMBL4744041)Show SMILES CC(C)OC1CN(C1)C(=O)NCc1ccc(cc1C)-c1ccnc(Nc2cnn(C)c2)n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Time-dependent inhibition of CYP3A4 in human liver microsomes using testosterone in presence of NADPH by LC-MS/MS analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00702
BindingDB Entry DOI: 10.7270/Q2BK1H1M |
More data for this
Ligand-Target Pair | |
Lanosterol synthase
(Rattus norvegicus) | BDBM50055645
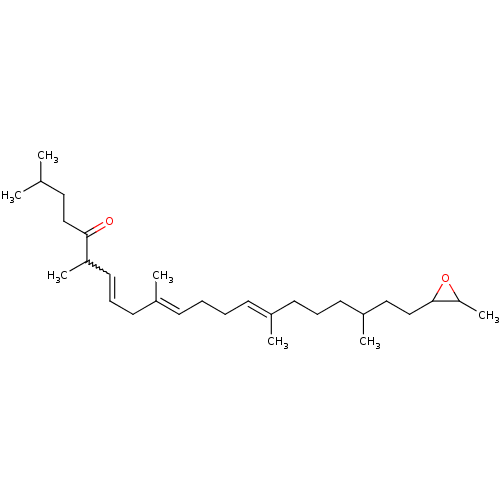
((6E,10E,14E)-2,6,10,15,19-Pentamethyl-21-(3-methyl...)Show SMILES CC(C)CCC(=O)C(C)C=CC\C(C)=C\CC\C=C(/C)CCCC(C)CCC1OC1C |w:9.8| Show InChI InChI=1S/C29H50O2/c1-22(2)18-20-28(30)26(6)17-11-16-24(4)13-9-8-12-23(3)14-10-15-25(5)19-21-29-27(7)31-29/h11-13,17,22,25-27,29H,8-10,14-16,18-21H2,1-7H3/b17-11?,23-12+,24-13+ | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| 6.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Simon Fraser University
Curated by ChEMBL
| Assay Description
Inhibition constant against rat liver Oxidosqualene-lanosterol cyclase |
J Med Chem 40: 201-9 (1997)
Article DOI: 10.1021/jm960483a
BindingDB Entry DOI: 10.7270/Q2P26X73 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50553434
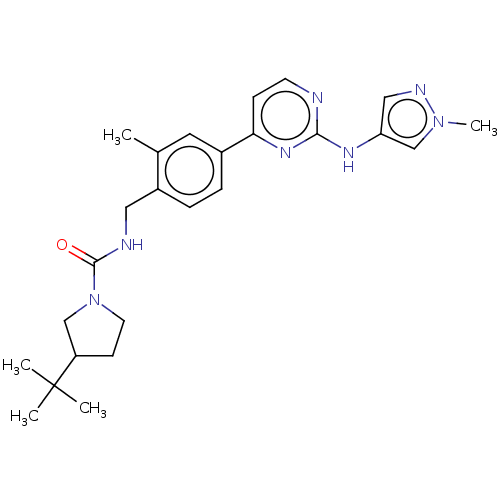
(CHEMBL4782313)Show SMILES Cc1cc(ccc1CNC(=O)N1CCC(C1)C(C)(C)C)-c1ccnc(Nc2cnn(C)c2)n1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length N-terminal His6-tagged BTK expressed in baculovirus infected Sf21 insect cells using fluorescein-labeled ... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00702
BindingDB Entry DOI: 10.7270/Q2BK1H1M |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50553431
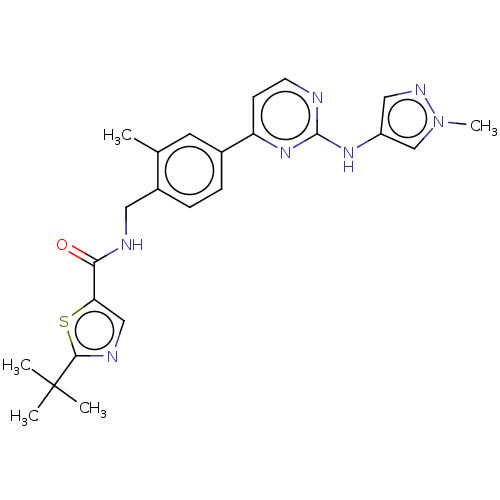
(CHEMBL4754065)Show SMILES Cc1cc(ccc1CNC(=O)c1cnc(s1)C(C)(C)C)-c1ccnc(Nc2cnn(C)c2)n1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length N-terminal His6-tagged BTK expressed in baculovirus infected Sf21 insect cells using fluorescein-labeled ... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00702
BindingDB Entry DOI: 10.7270/Q2BK1H1M |
More data for this
Ligand-Target Pair | |
Lanosterol synthase
(Homo sapiens (Human)) | BDBM50055641

(2,2-dimethyl-3-((3E,7E,11E)-3,7,12-trimethyl-14-(6...)Show SMILES [#6]-[#6](-[#6]-[#6]\[#6]=[#6](\[#6])-[#6])-[#16]-[#6]-[#6]\[#6](-[#6])=[#6]\[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]\[#6]=[#6](/[#6])-[#6]-[#6]-[#6]1-[#8]C1([#6])[#6] Show InChI InChI=1S/C29H50OS/c1-23(2)13-11-18-27(6)31-22-21-26(5)15-10-9-14-24(3)16-12-17-25(4)19-20-28-29(7,8)30-28/h13-15,17,27-28H,9-12,16,18-22H2,1-8H3/b24-14+,25-17+,26-15+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.220 | n/a | n/a | n/a | n/a | n/a | n/a |
Simon Fraser University
Curated by ChEMBL
| Assay Description
Compound was tested for inhibition of purified Oxidosqualene-lanosterol cyclase from Candida albicans |
J Med Chem 40: 201-9 (1997)
Article DOI: 10.1021/jm960483a
BindingDB Entry DOI: 10.7270/Q2P26X73 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50553435
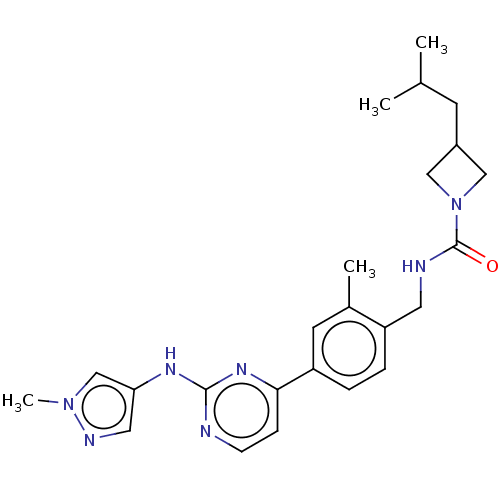
(CHEMBL4796367)Show SMILES CC(C)CC1CN(C1)C(=O)NCc1ccc(cc1C)-c1ccnc(Nc2cnn(C)c2)n1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length N-terminal His6-tagged BTK expressed in baculovirus infected Sf21 insect cells using fluorescein-labeled ... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00702
BindingDB Entry DOI: 10.7270/Q2BK1H1M |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50553426
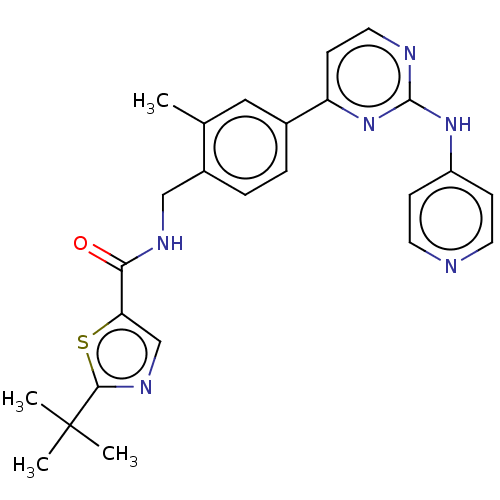
(CHEMBL4746275)Show SMILES Cc1cc(ccc1CNC(=O)c1cnc(s1)C(C)(C)C)-c1ccnc(Nc2ccncc2)n1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length N-terminal His6-tagged BTK expressed in baculovirus infected Sf21 insect cells using fluorescein-labeled ... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00702
BindingDB Entry DOI: 10.7270/Q2BK1H1M |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM368388
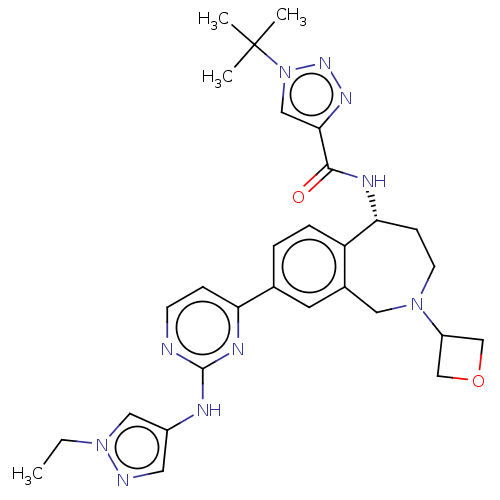
((R)-1-(tert-butyl)-N-(8-(2-((1-ethyl-1H-pyrazol-4-...)Show SMILES CCn1cc(Nc2nccc(n2)-c2ccc3[C@@H](CCN(Cc3c2)C2COC2)NC(=O)c2cn(nn2)C(C)(C)C)cn1 |r| Show InChI InChI=1S/C29H36N10O2/c1-5-38-15-21(13-31-38)32-28-30-10-8-24(34-28)19-6-7-23-20(12-19)14-37(22-17-41-18-22)11-9-25(23)33-27(40)26-16-39(36-35-26)29(2,3)4/h6-8,10,12-13,15-16,22,25H,5,9,11,14,17-18H2,1-4H3,(H,33,40)(H,30,32,34)/t25-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324281
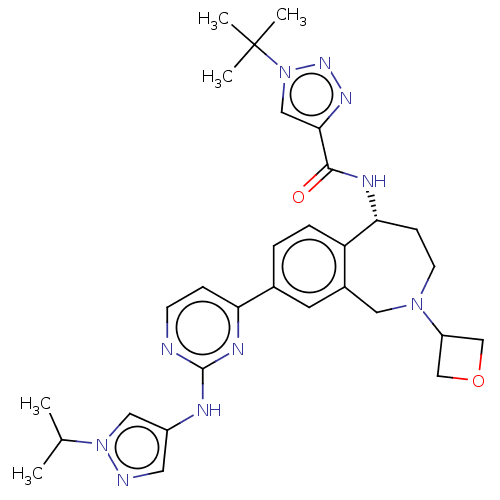
((R)-1-(tert-butyl)-N-(8-(2-((1-isopropyl-1H-pyrazo...)Show SMILES CC(C)n1cc(Nc2nccc(n2)-c2ccc3[C@@H](CCN(Cc3c2)C2COC2)NC(=O)c2cn(nn2)C(C)(C)C)cn1 |r| Show InChI InChI=1S/C30H38N10O2/c1-19(2)39-15-22(13-32-39)33-29-31-10-8-25(35-29)20-6-7-24-21(12-20)14-38(23-17-42-18-23)11-9-26(24)34-28(41)27-16-40(37-36-27)30(3,4)5/h6-8,10,12-13,15-16,19,23,26H,9,11,14,17-18H2,1-5H3,(H,34,41)(H,31,33,35)/t26-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324282

(1-(tert-butyl)-N�((R)-8-(2-((1-methyl-1H-pyrazol-4...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3[C@@H](CCN(Cc3c2)[C@@H]2CCOC2)NC(=O)c2cn(nn2)C(C)(C)C)cn1 |r| Show InChI InChI=1S/C29H36N10O2/c1-29(2,3)39-17-26(35-36-39)27(40)33-25-8-11-38(22-9-12-41-18-22)15-20-13-19(5-6-23(20)25)24-7-10-30-28(34-24)32-21-14-31-37(4)16-21/h5-7,10,13-14,16-17,22,25H,8-9,11-12,15,18H2,1-4H3,(H,33,40)(H,30,32,34)/t22-,25-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324284
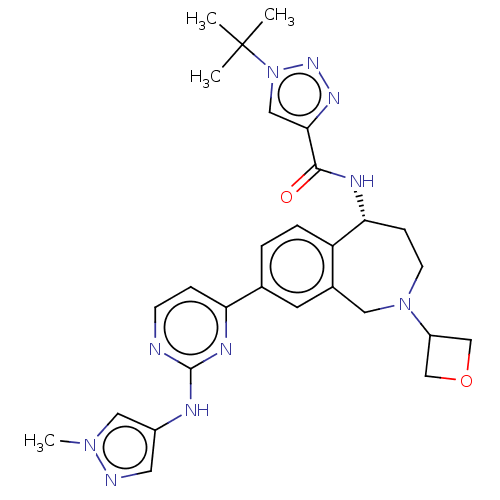
((R)-1-(tert-butyl)-N-(8-(2-((1-methyl-1H-pyrazol-4...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3[C@@H](CCN(Cc3c2)C2COC2)NC(=O)c2cn(nn2)C(C)(C)C)cn1 |r| Show InChI InChI=1S/C28H34N10O2/c1-28(2,3)38-15-25(34-35-38)26(39)32-24-8-10-37(21-16-40-17-21)13-19-11-18(5-6-22(19)24)23-7-9-29-27(33-23)31-20-12-30-36(4)14-20/h5-7,9,11-12,14-15,21,24H,8,10,13,16-17H2,1-4H3,(H,32,39)(H,29,31,33)/t24-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324285
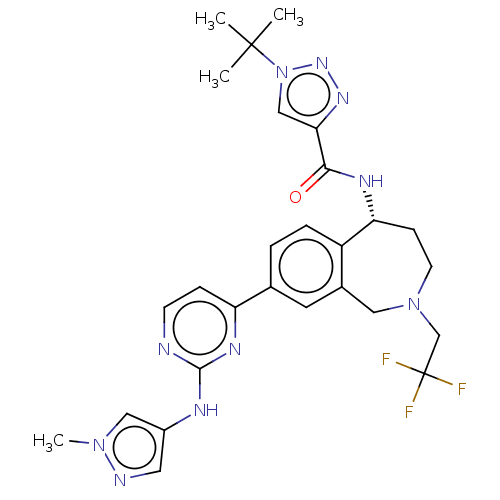
((R)-1-(tert-butyl)-N-(8-(2-((1-methyl-1H-pyrazol-4...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3[C@@H](CCN(CC(F)(F)F)Cc3c2)NC(=O)c2cn(nn2)C(C)(C)C)cn1 |r| Show InChI InChI=1S/C27H31F3N10O/c1-26(2,3)40-15-23(36-37-40)24(41)34-22-8-10-39(16-27(28,29)30)13-18-11-17(5-6-20(18)22)21-7-9-31-25(35-21)33-19-12-32-38(4)14-19/h5-7,9,11-12,14-15,22H,8,10,13,16H2,1-4H3,(H,34,41)(H,31,33,35)/t22-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324287

(1-(tert-butyl)-N—((R)-2-((S)-2-hydroxypropyl)...)Show SMILES C[C@H](O)CN1CC[C@@H](NC(=O)c2cn(nn2)C(C)(C)C)c2ccc(cc2C1)-c1ccnc(Nc2cnn(C)c2)n1 |r| Show InChI InChI=1S/C28H36N10O2/c1-18(39)14-37-11-9-24(32-26(40)25-17-38(35-34-25)28(2,3)4)22-7-6-19(12-20(22)15-37)23-8-10-29-27(33-23)31-21-13-30-36(5)16-21/h6-8,10,12-13,16-18,24,39H,9,11,14-15H2,1-5H3,(H,32,40)(H,29,31,33)/t18-,24+/m0/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324288
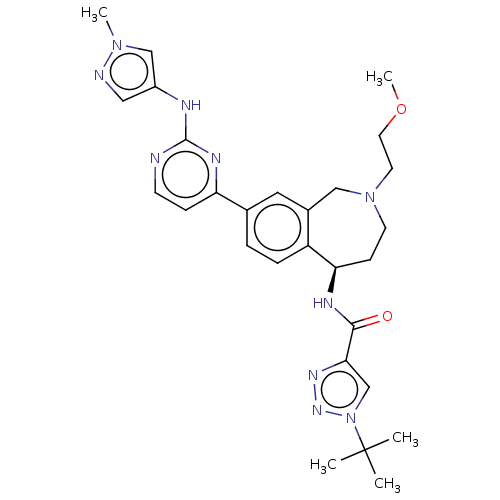
((R)-1-(tert-butyl)-N-(2-(2-methoxyethyl)-8-(2-((1-...)Show SMILES COCCN1CC[C@@H](NC(=O)c2cn(nn2)C(C)(C)C)c2ccc(cc2C1)-c1ccnc(Nc2cnn(C)c2)n1 |r| Show InChI InChI=1S/C28H36N10O2/c1-28(2,3)38-18-25(34-35-38)26(39)32-24-9-11-37(12-13-40-5)16-20-14-19(6-7-22(20)24)23-8-10-29-27(33-23)31-21-15-30-36(4)17-21/h6-8,10,14-15,17-18,24H,9,11-13,16H2,1-5H3,(H,32,39)(H,29,31,33)/t24-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324289
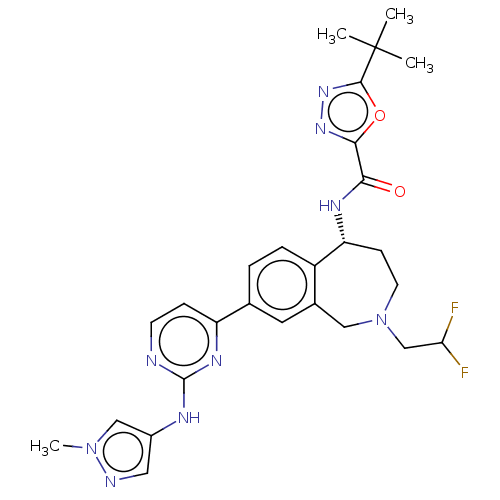
((R)-5-(tert-butyl)-N-(2-(2,2-difluoroethyl)-8-(2-(...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3[C@@H](CCN(CC(F)F)Cc3c2)NC(=O)c2nnc(o2)C(C)(C)C)cn1 |r| Show InChI InChI=1S/C27H31F2N9O2/c1-27(2,3)25-36-35-24(40-25)23(39)33-21-8-10-38(15-22(28)29)13-17-11-16(5-6-19(17)21)20-7-9-30-26(34-20)32-18-12-31-37(4)14-18/h5-7,9,11-12,14,21-22H,8,10,13,15H2,1-4H3,(H,33,39)(H,30,32,34)/t21-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324291

((R)—N-(2-(2-((1-methyl-1H-pyrazol-4-yl)amino)...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3[C@@H](CCCCc3c2)NC(=O)c2noc(n2)C2(C)CC2)cn1 |r| Show InChI InChI=1S/C26H28N8O2/c1-26(10-11-26)24-32-22(33-36-24)23(35)30-21-6-4-3-5-16-13-17(7-8-19(16)21)20-9-12-27-25(31-20)29-18-14-28-34(2)15-18/h7-9,12-15,21H,3-6,10-11H2,1-2H3,(H,30,35)(H,27,29,31)/t21-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324292
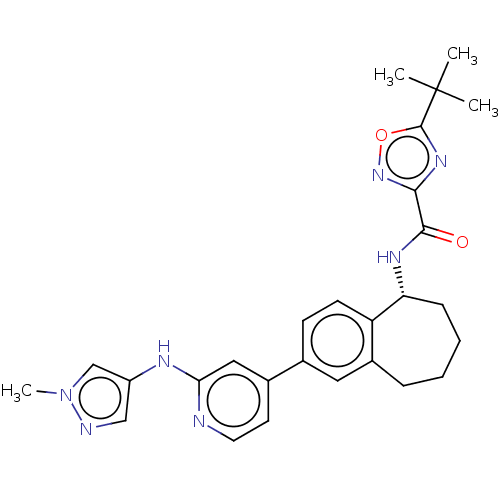
((R)-5-(tert-butyl)-N-(2-(2-((1-methyl-1H-pyrazol-4...)Show SMILES Cn1cc(Nc2cc(ccn2)-c2ccc3[C@@H](CCCCc3c2)NC(=O)c2noc(n2)C(C)(C)C)cn1 |r| Show InChI InChI=1S/C27H31N7O2/c1-27(2,3)26-32-24(33-36-26)25(35)31-22-8-6-5-7-19-13-17(9-10-21(19)22)18-11-12-28-23(14-18)30-20-15-29-34(4)16-20/h9-16,22H,5-8H2,1-4H3,(H,28,30)(H,31,35)/t22-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324294
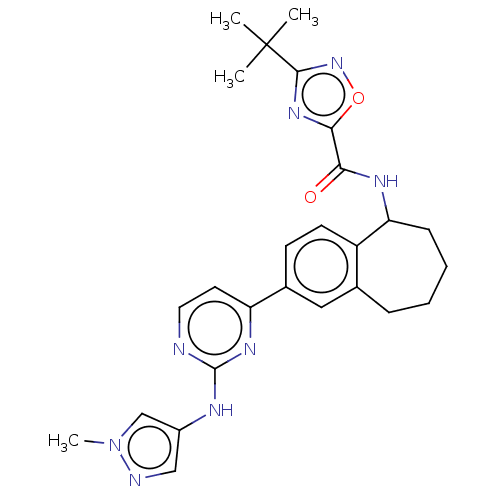
(3-(tert-butyl)-N-(2-(2-((1-methyl-1H-pyrazol-4-yl)...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3C(CCCCc3c2)NC(=O)c2nc(no2)C(C)(C)C)cn1 Show InChI InChI=1S/C26H30N8O2/c1-26(2,3)24-32-23(36-33-24)22(35)30-21-8-6-5-7-16-13-17(9-10-19(16)21)20-11-12-27-25(31-20)29-18-14-28-34(4)15-18/h9-15,21H,5-8H2,1-4H3,(H,30,35)(H,27,29,31) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324295
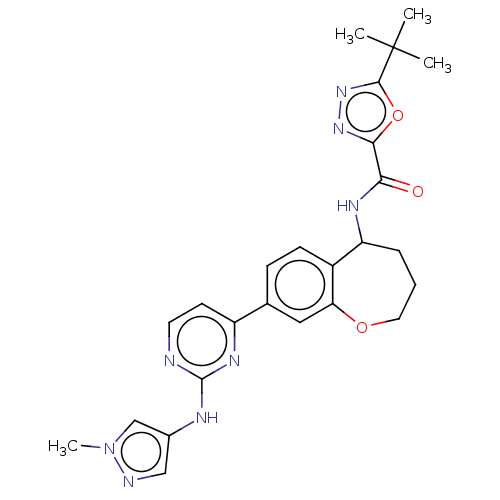
(5-(tert-butyl)-N-(8-(2-((1-methyl-1H-pyrazol-4-yl)...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3C(CCCOc3c2)NC(=O)c2nnc(o2)C(C)(C)C)cn1 Show InChI InChI=1S/C25H28N8O3/c1-25(2,3)23-32-31-22(36-23)21(34)29-19-6-5-11-35-20-12-15(7-8-17(19)20)18-9-10-26-24(30-18)28-16-13-27-33(4)14-16/h7-10,12-14,19H,5-6,11H2,1-4H3,(H,29,34)(H,26,28,30) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324296
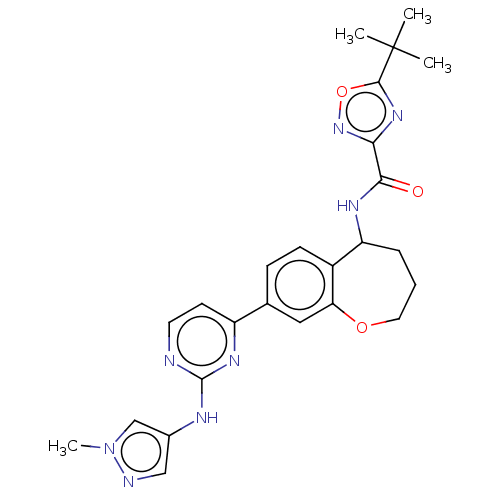
(5-(tert-butyl)-N-(8-(2-((1-methyl-1H-pyrazol-4-yl)...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3C(CCCOc3c2)NC(=O)c2noc(n2)C(C)(C)C)cn1 Show InChI InChI=1S/C25H28N8O3/c1-25(2,3)23-31-21(32-36-23)22(34)29-19-6-5-11-35-20-12-15(7-8-17(19)20)18-9-10-26-24(30-18)28-16-13-27-33(4)14-16/h7-10,12-14,19H,5-6,11H2,1-4H3,(H,29,34)(H,26,28,30) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM368406
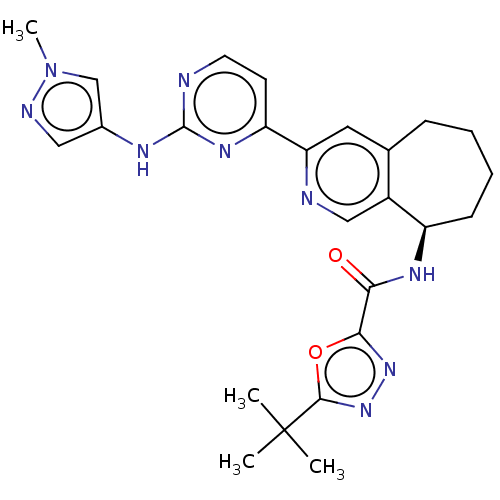
((R)-5-(tert-butyl)-N-(3-(2-((1-methyl-1H-pyrazol-4...)Show SMILES Cn1cc(Nc2nccc(n2)-c2cc3CCCC[C@@H](NC(=O)c4nnc(o4)C(C)(C)C)c3cn2)cn1 |r| Show InChI InChI=1S/C25H29N9O2/c1-25(2,3)23-33-32-22(36-23)21(35)30-18-8-6-5-7-15-11-20(27-13-17(15)18)19-9-10-26-24(31-19)29-16-12-28-34(4)14-16/h9-14,18H,5-8H2,1-4H3,(H,30,35)(H,26,29,31)/t18-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324299
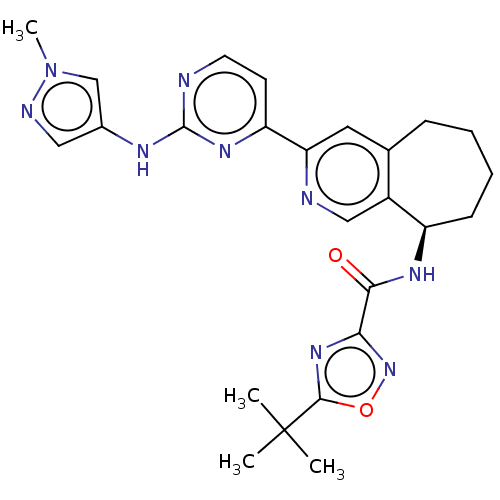
((R)-5-(tert-butyl)-N-(3-(2-((1-methyl-1H-pyrazol-4...)Show SMILES Cn1cc(Nc2nccc(n2)-c2cc3CCCC[C@@H](NC(=O)c4noc(n4)C(C)(C)C)c3cn2)cn1 |r| Show InChI InChI=1S/C25H29N9O2/c1-25(2,3)23-32-21(33-36-23)22(35)30-18-8-6-5-7-15-11-20(27-13-17(15)18)19-9-10-26-24(31-19)29-16-12-28-34(4)14-16/h9-14,18H,5-8H2,1-4H3,(H,30,35)(H,26,29,31)/t18-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324300
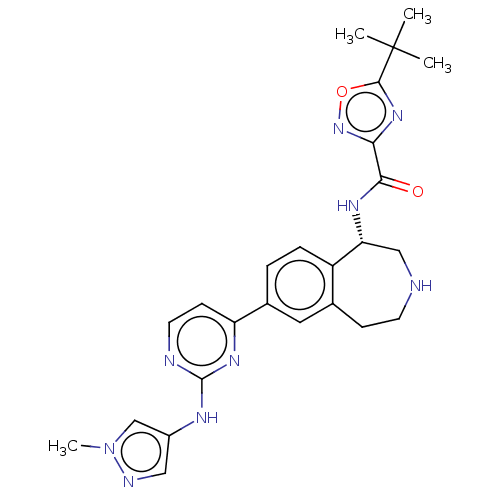
((S)-5-(tert-butyl)-N-(7- (2-((1-methyl-1H- pyrazol...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3[C@@H](CNCCc3c2)NC(=O)c2noc(n2)C(C)(C)C)cn1 |r| Show InChI InChI=1S/C25H29N9O2/c1-25(2,3)23-32-21(33-36-23)22(35)30-20-13-26-9-7-15-11-16(5-6-18(15)20)19-8-10-27-24(31-19)29-17-12-28-34(4)14-17/h5-6,8,10-12,14,20,26H,7,9,13H2,1-4H3,(H,30,35)(H,27,29,31)/t20-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324302
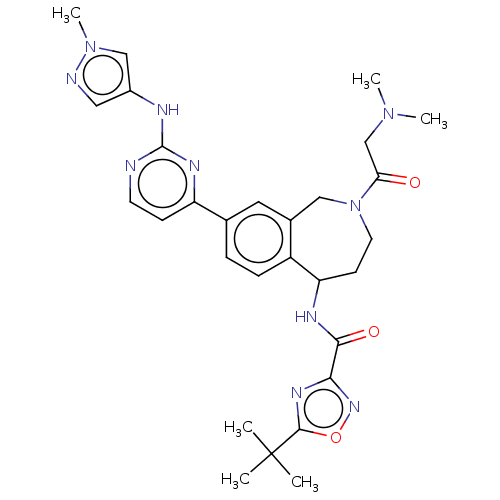
(5-(tert-butyl)-N-(2-(2- (dimethylamino)acetyl)- 8-...)Show SMILES CN(C)CC(=O)N1CCC(NC(=O)c2noc(n2)C(C)(C)C)c2ccc(cc2C1)-c1ccnc(Nc2cnn(C)c2)n1 Show InChI InChI=1S/C29H36N10O3/c1-29(2,3)27-35-25(36-42-27)26(41)33-23-10-12-39(24(40)17-37(4)5)15-19-13-18(7-8-21(19)23)22-9-11-30-28(34-22)32-20-14-31-38(6)16-20/h7-9,11,13-14,16,23H,10,12,15,17H2,1-6H3,(H,33,41)(H,30,32,34) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324303
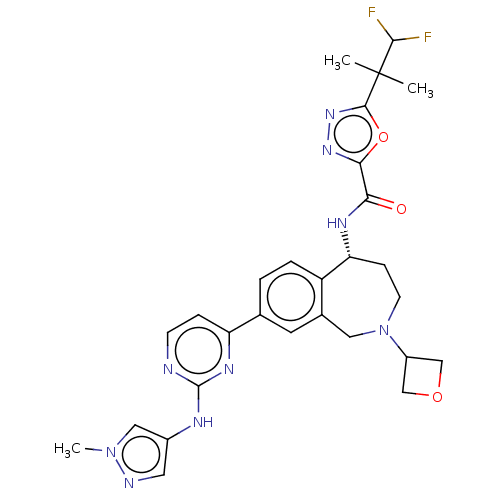
((R)-5-(1,1-difluoro-2- methylpropan-2-yl)- N-(8- (...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3[C@@H](CCN(Cc3c2)C2COC2)NC(=O)c2nnc(o2)C(C)(C)C(F)F)cn1 |r| Show InChI InChI=1S/C28H31F2N9O3/c1-28(2,25(29)30)26-37-36-24(42-26)23(40)34-22-7-9-39(19-14-41-15-19)12-17-10-16(4-5-20(17)22)21-6-8-31-27(35-21)33-18-11-32-38(3)13-18/h4-6,8,10-11,13,19,22,25H,7,9,12,14-15H2,1-3H3,(H,34,40)(H,31,33,35)/t22-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324305
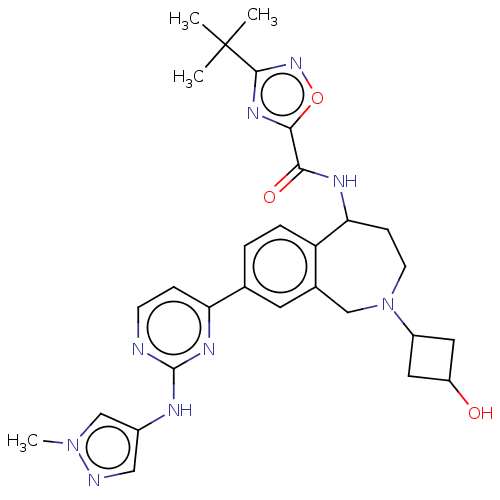
(3-(tert-butyl)-N-(2-(3- hydroxycyclobutyl)-8-(2- (...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3C(CCN(Cc3c2)C2CC(O)C2)NC(=O)c2nc(no2)C(C)(C)C)cn1 |(7.73,-4.39,;6.19,-4.39,;5.28,-5.64,;3.82,-5.16,;2.48,-5.93,;1.15,-5.16,;-.18,-5.93,;-1.52,-5.16,;-1.52,-3.62,;-.18,-2.85,;1.15,-3.62,;-.18,-1.31,;1.15,-.54,;1.15,1,;-.18,1.77,;-.18,3.31,;-1.38,4.26,;-2.88,3.91,;-3.54,2.52,;-2.88,1.13,;-1.52,1,;-1.52,-.54,;-5.02,2.92,;-5.79,4.25,;-7.13,3.48,;-8.62,3.88,;-6.36,2.15,;1.15,4.08,;2.48,3.31,;2.48,1.77,;3.94,3.83,;5.4,3.35,;6.31,4.6,;5.4,5.84,;3.94,5.37,;7.85,4.6,;8.62,5.93,;8.62,3.26,;7.08,5.93,;3.82,-3.62,;5.28,-3.14,)| Show InChI InChI=1S/C29H35N9O3/c1-29(2,3)27-35-26(41-36-27)25(40)33-24-8-10-38(20-12-21(39)13-20)15-18-11-17(5-6-22(18)24)23-7-9-30-28(34-23)32-19-14-31-37(4)16-19/h5-7,9,11,14,16,20-21,24,39H,8,10,12-13,15H2,1-4H3,(H,33,40)(H,30,32,34) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324306
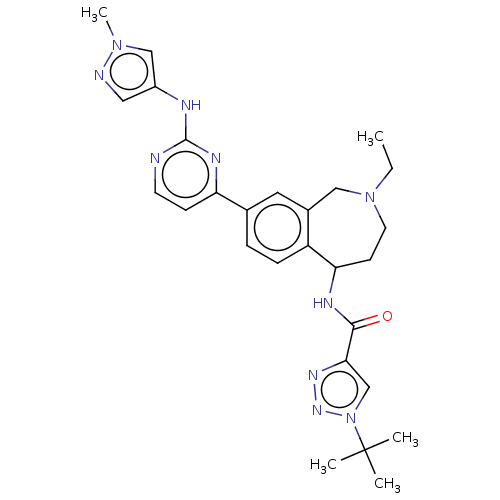
(1-(tert-butyl)-N-(2-ethyl- 8-(2-((1-methyl-1H- pyr...)Show SMILES CCN1CCC(NC(=O)c2cn(nn2)C(C)(C)C)c2ccc(cc2C1)-c1ccnc(Nc2cnn(C)c2)n1 Show InChI InChI=1S/C27H34N10O/c1-6-36-12-10-23(31-25(38)24-17-37(34-33-24)27(2,3)4)21-8-7-18(13-19(21)15-36)22-9-11-28-26(32-22)30-20-14-29-35(5)16-20/h7-9,11,13-14,16-17,23H,6,10,12,15H2,1-5H3,(H,31,38)(H,28,30,32) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
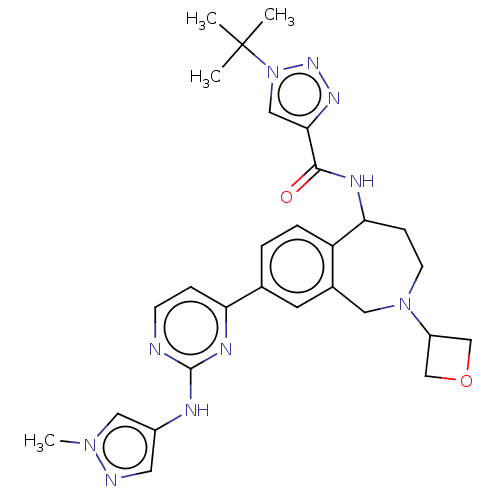
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324307
(1-(tert-butyl)-N-(8-(2- ((1-methyl-1H-pyrazol- 4-y...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3C(CCN(Cc3c2)C2COC2)NC(=O)c2cn(nn2)C(C)(C)C)cn1 Show InChI InChI=1S/C28H34N10O2/c1-28(2,3)38-15-25(34-35-38)26(39)32-24-8-10-37(21-16-40-17-21)13-19-11-18(5-6-22(19)24)23-7-9-29-27(33-23)31-20-12-30-36(4)14-20/h5-7,9,11-12,14-15,21,24H,8,10,13,16-17H2,1-4H3,(H,32,39)(H,29,31,33) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324309
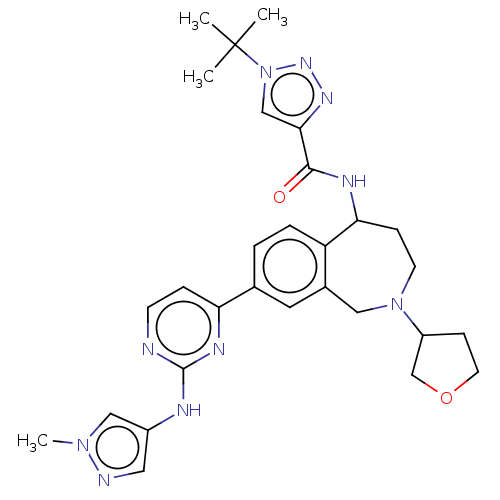
(1-(tert-butyl)-N-(8-(2- ((1-methyl-1H- pyrazol-4- ...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3C(CCN(Cc3c2)C2CCOC2)NC(=O)c2cn(nn2)C(C)(C)C)cn1 Show InChI InChI=1S/C29H36N10O2/c1-29(2,3)39-17-26(35-36-39)27(40)33-25-8-11-38(22-9-12-41-18-22)15-20-13-19(5-6-23(20)25)24-7-10-30-28(34-24)32-21-14-31-37(4)16-21/h5-7,10,13-14,16-17,22,25H,8-9,11-12,15,18H2,1-4H3,(H,33,40)(H,30,32,34) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324310
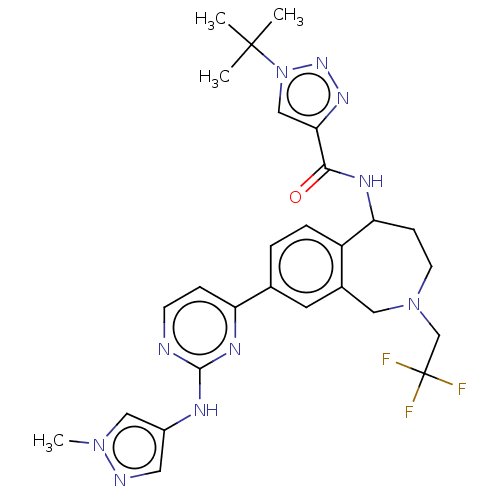
(1-(tert-butyl)-N-(8- (2-((1- methyl-1H-pyrazol-4- ...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3C(CCN(CC(F)(F)F)Cc3c2)NC(=O)c2cn(nn2)C(C)(C)C)cn1 Show InChI InChI=1S/C27H31F3N10O/c1-26(2,3)40-15-23(36-37-40)24(41)34-22-8-10-39(16-27(28,29)30)13-18-11-17(5-6-20(18)22)21-7-9-31-25(35-21)33-19-12-32-38(4)14-19/h5-7,9,11-12,14-15,22H,8,10,13,16H2,1-4H3,(H,34,41)(H,31,33,35) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324311
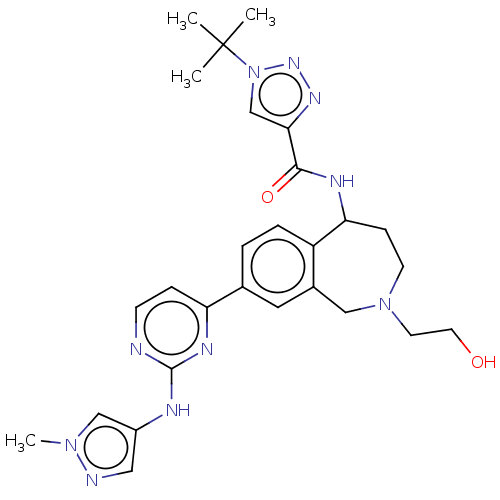
(1-(tert-butyl)-N-(2-(2- hydroxyethyl)-8-(2-((1- me...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3C(CCN(CCO)Cc3c2)NC(=O)c2cn(nn2)C(C)(C)C)cn1 Show InChI InChI=1S/C27H34N10O2/c1-27(2,3)37-17-24(33-34-37)25(39)31-23-8-10-36(11-12-38)15-19-13-18(5-6-21(19)23)22-7-9-28-26(32-22)30-20-14-29-35(4)16-20/h5-7,9,13-14,16-17,23,38H,8,10-12,15H2,1-4H3,(H,31,39)(H,28,30,32) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324313

(5-(tert-butyl)-N-(2- ((R)-2-hydroxypropyl)- 8-(2-(...)Show SMILES C[C@@H](O)CN1CCC(NC(=O)c2noc(n2)C(C)(C)C)c2ccc(cc2C1)-c1ccnc(Nc2cnn(C)c2)n1 |r| Show InChI InChI=1S/C28H35N9O3/c1-17(38)14-37-11-9-23(32-25(39)24-34-26(40-35-24)28(2,3)4)21-7-6-18(12-19(21)15-37)22-8-10-29-27(33-22)31-20-13-30-36(5)16-20/h6-8,10,12-13,16-17,23,38H,9,11,14-15H2,1-5H3,(H,32,39)(H,29,31,33)/t17-,23?/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324314
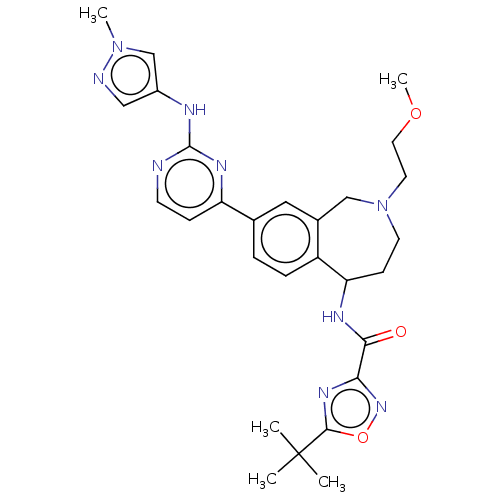
(5-(tert-butyl)-N-(2-(2- methoxyethyl)-8-(2-((1- me...)Show SMILES COCCN1CCC(NC(=O)c2noc(n2)C(C)(C)C)c2ccc(cc2C1)-c1ccnc(Nc2cnn(C)c2)n1 Show InChI InChI=1S/C28H35N9O3/c1-28(2,3)26-34-24(35-40-26)25(38)32-23-9-11-37(12-13-39-5)16-19-14-18(6-7-21(19)23)22-8-10-29-27(33-22)31-20-15-30-36(4)17-20/h6-8,10,14-15,17,23H,9,11-13,16H2,1-5H3,(H,32,38)(H,29,31,33) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324316
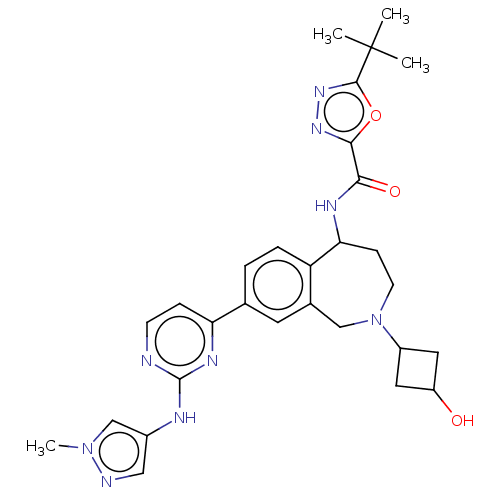
(5-(tert-butyl)-N-(2-(3- hydroxycyclobutyl)-8-(2- (...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3C(CCN(Cc3c2)C2CC(O)C2)NC(=O)c2nnc(o2)C(C)(C)C)cn1 |(7.77,-4.52,;6.23,-4.52,;5.32,-5.76,;3.86,-5.29,;2.52,-6.06,;1.19,-5.29,;-.14,-6.06,;-1.48,-5.29,;-1.48,-3.75,;-.14,-2.98,;1.19,-3.75,;-.14,-1.44,;1.19,-.67,;1.19,.87,;-.14,1.64,;-.14,3.18,;-.96,4.29,;-2.5,4.18,;-3.36,2.91,;-2.91,1.44,;-1.48,.87,;-1.48,-.67,;-4.89,3.14,;-5.75,4.41,;-7.03,3.55,;-8.54,3.85,;-6.16,2.28,;1.19,3.95,;2.52,3.18,;2.52,1.64,;3.86,3.95,;3.86,5.49,;5.32,5.97,;6.23,4.72,;5.32,3.48,;7.77,4.72,;7,6.06,;8.54,3.39,;8.54,6.06,;3.86,-3.75,;5.32,-3.27,)| Show InChI InChI=1S/C29H35N9O3/c1-29(2,3)27-36-35-26(41-27)25(40)33-24-8-10-38(20-12-21(39)13-20)15-18-11-17(5-6-22(18)24)23-7-9-30-28(34-23)32-19-14-31-37(4)16-19/h5-7,9,11,14,16,20-21,24,39H,8,10,12-13,15H2,1-4H3,(H,33,40)(H,30,32,34) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324317

((R)-5-(tert-butyl)-N-(2- (3-hydroxycyclobutyl)-8- ...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3[C@@H](CCN(Cc3c2)C2CC(O)C2)NC(=O)c2nnc(o2)C(C)(C)C)cn1 |r,wU:15.30,(7.77,-4.52,;6.23,-4.52,;5.32,-5.76,;3.86,-5.29,;2.52,-6.06,;1.19,-5.29,;-.14,-6.06,;-1.48,-5.29,;-1.48,-3.75,;-.14,-2.98,;1.19,-3.75,;-.14,-1.44,;1.19,-.67,;1.19,.87,;-.14,1.64,;-.14,3.18,;-.96,4.29,;-2.5,4.18,;-3.36,2.91,;-2.91,1.44,;-1.48,.87,;-1.48,-.67,;-4.89,3.14,;-5.75,4.41,;-7.03,3.55,;-8.54,3.85,;-6.16,2.28,;1.19,3.95,;2.52,3.18,;2.52,1.64,;3.86,3.95,;3.86,5.49,;5.32,5.97,;6.23,4.72,;5.32,3.48,;7.77,4.72,;7,6.06,;8.54,3.39,;8.54,6.06,;3.86,-3.75,;5.32,-3.27,)| Show InChI InChI=1S/C29H35N9O3/c1-29(2,3)27-36-35-26(41-27)25(40)33-24-8-10-38(20-12-21(39)13-20)15-18-11-17(5-6-22(18)24)23-7-9-30-28(34-23)32-19-14-31-37(4)16-19/h5-7,9,11,14,16,20-21,24,39H,8,10,12-13,15H2,1-4H3,(H,33,40)(H,30,32,34)/t20?,21?,24-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324318

(5-(tert-butyl)-N-(2-ethyl- 8-(2-((1-methyl-1H- pyr...)Show SMILES CCN1CCC(NC(=O)c2nnc(o2)C(C)(C)C)c2ccc(cc2C1)-c1ccnc(Nc2cnn(C)c2)n1 Show InChI InChI=1S/C27H33N9O2/c1-6-36-12-10-22(31-23(37)24-33-34-25(38-24)27(2,3)4)20-8-7-17(13-18(20)15-36)21-9-11-28-26(32-21)30-19-14-29-35(5)16-19/h7-9,11,13-14,16,22H,6,10,12,15H2,1-5H3,(H,31,37)(H,28,30,32) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324320
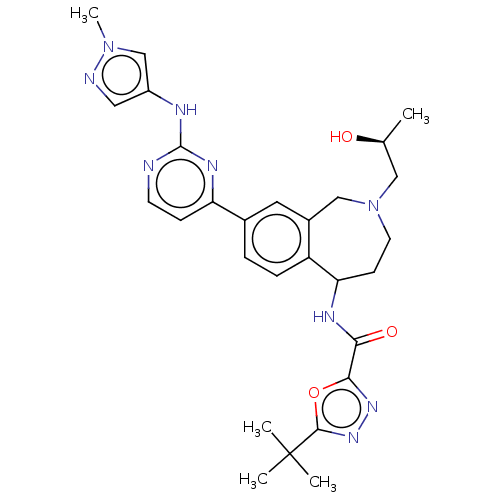
(5-(tert-butyl)-N-(2- ((S)-2-hydroxypropyl)- 8-(2-(...)Show SMILES C[C@H](O)CN1CCC(NC(=O)c2nnc(o2)C(C)(C)C)c2ccc(cc2C1)-c1ccnc(Nc2cnn(C)c2)n1 |r| Show InChI InChI=1S/C28H35N9O3/c1-17(38)14-37-11-9-23(32-24(39)25-34-35-26(40-25)28(2,3)4)21-7-6-18(12-19(21)15-37)22-8-10-29-27(33-22)31-20-13-30-36(5)16-20/h6-8,10,12-13,16-17,23,38H,9,11,14-15H2,1-5H3,(H,32,39)(H,29,31,33)/t17-,23?/m0/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
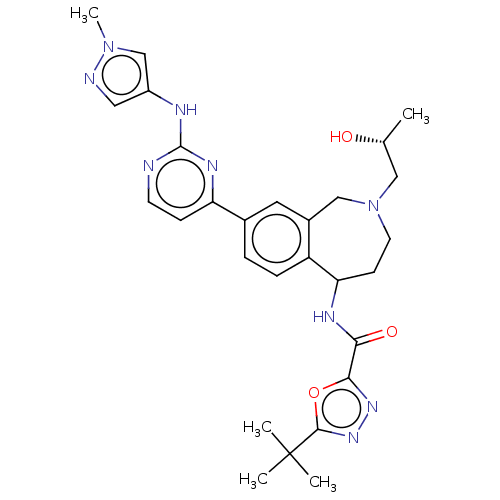
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324321
(5-(tert-butyl)-N-(2-((R)- 2-hydroxypropyl)-8-(2- (...)Show SMILES C[C@@H](O)CN1CCC(NC(=O)c2nnc(o2)C(C)(C)C)c2ccc(cc2C1)-c1ccnc(Nc2cnn(C)c2)n1 |r| Show InChI InChI=1S/C28H35N9O3/c1-17(38)14-37-11-9-23(32-24(39)25-34-35-26(40-25)28(2,3)4)21-7-6-18(12-19(21)15-37)22-8-10-29-27(33-22)31-20-13-30-36(5)16-20/h6-8,10,12-13,16-17,23,38H,9,11,14-15H2,1-5H3,(H,32,39)(H,29,31,33)/t17-,23?/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324322

(5-(tert-butyl)-N-(2-(2- hydroxy-2- methylpropyl)-8...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3C(CCN(CC(C)(C)O)Cc3c2)NC(=O)c2nnc(o2)C(C)(C)C)cn1 Show InChI InChI=1S/C29H37N9O3/c1-28(2,3)26-36-35-25(41-26)24(39)33-23-10-12-38(17-29(4,5)40)15-19-13-18(7-8-21(19)23)22-9-11-30-27(34-22)32-20-14-31-37(6)16-20/h7-9,11,13-14,16,23,40H,10,12,15,17H2,1-6H3,(H,33,39)(H,30,32,34) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324324
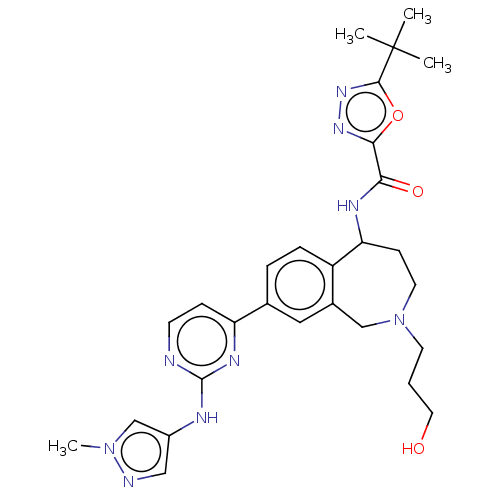
(5-(tert-butyl)-N-(2-(3- hydroxypropyl)- 8-(2-((1- ...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3C(CCN(CCCO)Cc3c2)NC(=O)c2nnc(o2)C(C)(C)C)cn1 Show InChI InChI=1S/C28H35N9O3/c1-28(2,3)26-35-34-25(40-26)24(39)32-23-9-12-37(11-5-13-38)16-19-14-18(6-7-21(19)23)22-8-10-29-27(33-22)31-20-15-30-36(4)17-20/h6-8,10,14-15,17,23,38H,5,9,11-13,16H2,1-4H3,(H,32,39)(H,29,31,33) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324325

(5-(tert-butyl)-N- (8-(2-((1- methyl-1H-pyrazol-4- ...)Show SMILES Cn1cc(Nc2nccc(n2)-c2ccc3C(CCN(CC(F)(F)F)Cc3c2)NC(=O)c2nnc(o2)C(C)(C)C)cn1 Show InChI InChI=1S/C27H30F3N9O2/c1-26(2,3)24-37-36-23(41-24)22(40)34-21-8-10-39(15-27(28,29)30)13-17-11-16(5-6-19(17)21)20-7-9-31-25(35-20)33-18-12-32-38(4)14-18/h5-7,9,11-12,14,21H,8,10,13,15H2,1-4H3,(H,34,40)(H,31,33,35) | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK [14-552]
(Homo sapiens (Human)) | BDBM324327

((R)-5-(tert-butyl)-N-(8- (2-((5,6-dihydro-4H- pyrr...)Show SMILES CC(C)(C)c1nnc(o1)C(=O)N[C@@H]1CCN(CCO)Cc2cc(ccc12)-c1ccnc(Nc2cnn3CCCc23)n1 |r| Show InChI InChI=1S/C29H35N9O3/c1-29(2,3)27-36-35-26(41-27)25(40)32-22-9-12-37(13-14-39)17-19-15-18(6-7-20(19)22)21-8-10-30-28(33-21)34-23-16-31-38-11-4-5-24(23)38/h6-8,10,15-16,22,39H,4-5,9,11-14,17H2,1-3H3,(H,32,40)(H,30,33,34)/t22-/m1/s1 | PDB
MMDB
Reactome pathway
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data