 Found 15 hits with Last Name = 'grindey' and Initial = 'gb'
Found 15 hits with Last Name = 'grindey' and Initial = 'gb' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Trifunctional purine biosynthetic protein adenosine-3
(Mus musculus) | BDBM50280415
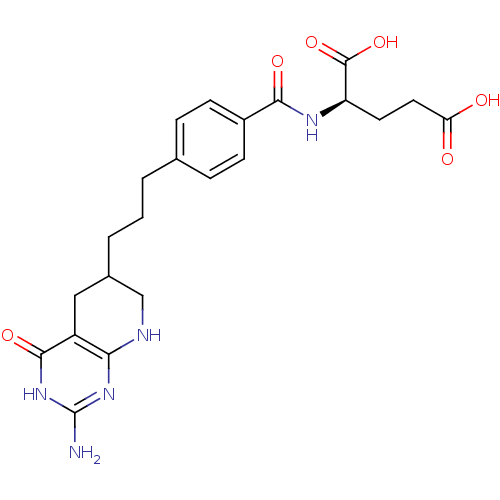
((R)-2-{4-[3-(2-Amino-4-oxo-3,4,5,6,7,8-hexahydro-p...)Show SMILES Nc1nc2NCC(CCCc3ccc(cc3)C(=O)N[C@H](CCC(O)=O)C(O)=O)Cc2c(=O)[nH]1 Show InChI InChI=1S/C22H27N5O6/c23-22-26-18-15(20(31)27-22)10-13(11-24-18)3-1-2-12-4-6-14(7-5-12)19(30)25-16(21(32)33)8-9-17(28)29/h4-7,13,16H,1-3,8-11H2,(H,25,30)(H,28,29)(H,32,33)(H4,23,24,26,27,31) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
| 0.0000190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against glycinamide ribonucleotide formyltransferase (GARFT) from L1210 murine leukemic cells |
Bioorg Med Chem Lett 2: 339-342 (1992)
Article DOI: 10.1016/S0960-894X(01)80214-6
BindingDB Entry DOI: 10.7270/Q2RF5TXV |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Mus musculus) | BDBM22590
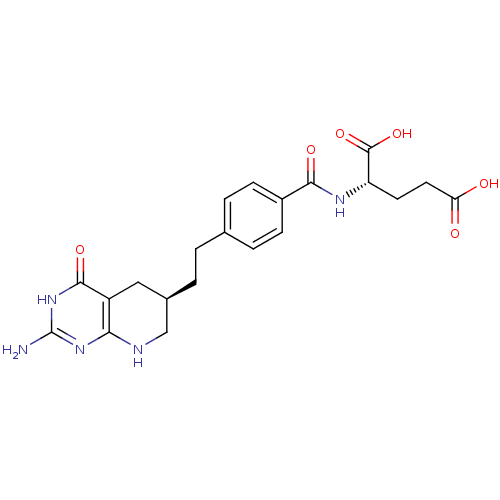
((2S)-2-[(4-{2-[(6R)-2-amino-4-oxo-1H,4H,5H,6H,7H,8...)Show SMILES Nc1nc2NC[C@H](CCc3ccc(cc3)C(=O)N[C@@H](CCC(O)=O)C(O)=O)Cc2c(=O)[nH]1 Show InChI InChI=1S/C21H25N5O6/c22-21-25-17-14(19(30)26-21)9-12(10-23-17)2-1-11-3-5-13(6-4-11)18(29)24-15(20(31)32)7-8-16(27)28/h3-6,12,15H,1-2,7-10H2,(H,24,29)(H,27,28)(H,31,32)(H4,22,23,25,26,30)/t12-,15+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
| 0.000120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against glycinamide ribonucleotide formyltransferase (GARFT) from L1210 murine leukemic cells |
Bioorg Med Chem Lett 2: 339-342 (1992)
Article DOI: 10.1016/S0960-894X(01)80214-6
BindingDB Entry DOI: 10.7270/Q2RF5TXV |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Mus musculus) | BDBM50280414
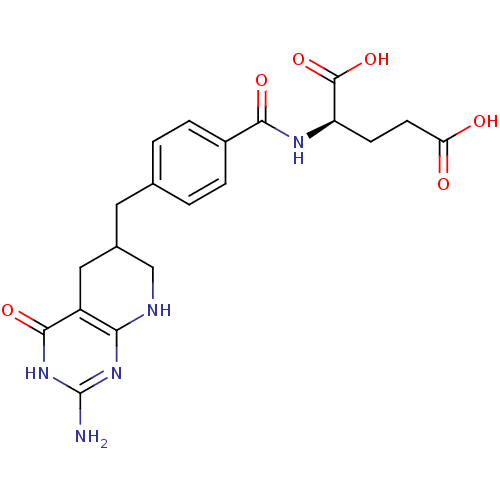
((R)-2-[4-(2-Amino-4-oxo-3,4,5,6,7,8-hexahydro-pyri...)Show SMILES Nc1nc2NCC(Cc3ccc(cc3)C(=O)N[C@H](CCC(O)=O)C(O)=O)Cc2c(=O)[nH]1 Show InChI InChI=1S/C20H23N5O6/c21-20-24-16-13(18(29)25-20)8-11(9-22-16)7-10-1-3-12(4-2-10)17(28)23-14(19(30)31)5-6-15(26)27/h1-4,11,14H,5-9H2,(H,23,28)(H,26,27)(H,30,31)(H4,21,22,24,25,29) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
| 0.000630 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against glycinamide ribonucleotide formyltransferase (GARFT) from L1210 murine leukemic cells |
Bioorg Med Chem Lett 2: 339-342 (1992)
Article DOI: 10.1016/S0960-894X(01)80214-6
BindingDB Entry DOI: 10.7270/Q2RF5TXV |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Homo sapiens (Human)) | BDBM50288820
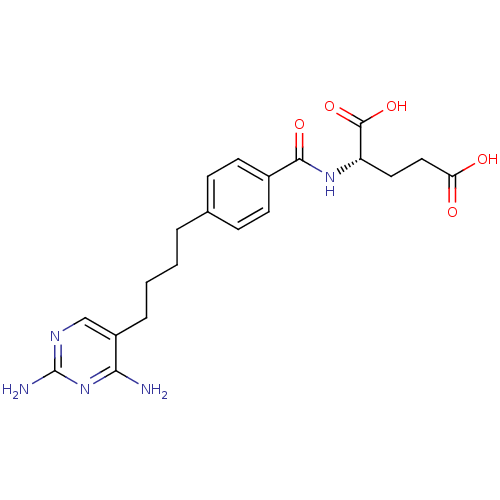
((S)-2-{(S)-4-[4-(2,4-Diamino-pyrimidin-5-yl)-butyl...)Show SMILES Nc1ncc(CCCCc2ccc(cc2)C(=O)N[C@@H](CCC(O)=O)C(O)=O)c(N)n1 Show InChI InChI=1S/C20H25N5O5/c21-17-14(11-23-20(22)25-17)4-2-1-3-12-5-7-13(8-6-12)18(28)24-15(19(29)30)9-10-16(26)27/h5-8,11,15H,1-4,9-10H2,(H,24,28)(H,26,27)(H,29,30)(H4,21,22,23,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Binding affinity of the compound against Recombinant human dihydrofolate reductase (DHFR) |
Bioorg Med Chem Lett 6: 473-476 (1996)
Article DOI: 10.1016/0960-894X(96)00053-4
BindingDB Entry DOI: 10.7270/Q2GQ6XQ2 |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Homo sapiens (Human)) | BDBM50288821

((S)-2-{(S)-4-[3-(2,4-Diamino-pyrimidin-5-yl)-propy...)Show SMILES Nc1ncc(CCCc2ccc(cc2)C(=O)N[C@@H](CCC(O)=O)C(O)=O)c(N)n1 Show InChI InChI=1S/C19H23N5O5/c20-16-13(10-22-19(21)24-16)3-1-2-11-4-6-12(7-5-11)17(27)23-14(18(28)29)8-9-15(25)26/h4-7,10,14H,1-3,8-9H2,(H,23,27)(H,25,26)(H,28,29)(H4,20,21,22,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Binding affinity of the compound against Recombinant human dihydrofolate reductase (DHFR) |
Bioorg Med Chem Lett 6: 473-476 (1996)
Article DOI: 10.1016/0960-894X(96)00053-4
BindingDB Entry DOI: 10.7270/Q2GQ6XQ2 |
More data for this
Ligand-Target Pair | |
Thymidylate synthase
(Mus musculus) | BDBM50003896
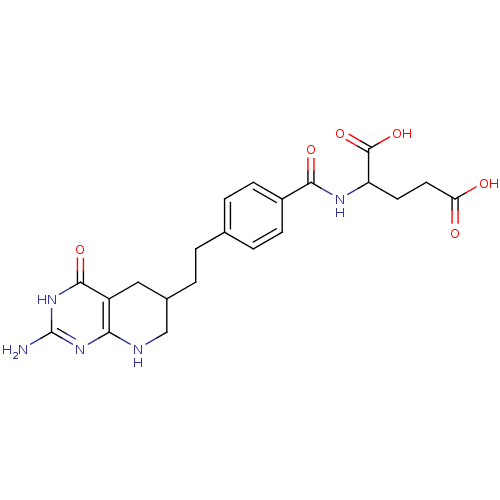
((DDATHF) 5,10-Dideazatetrahydrofolic acid2-{4-[2-(...)Show SMILES Nc1nc2NCC(CCc3ccc(cc3)C(=O)NC(CCC(O)=O)C(O)=O)Cc2c(=O)[nH]1 Show InChI InChI=1S/C21H25N5O6/c22-21-25-17-14(19(30)26-21)9-12(10-23-17)2-1-11-3-5-13(6-4-11)18(29)24-15(20(31)32)7-8-16(27)28/h3-6,12,15H,1-2,7-10H2,(H,24,29)(H,27,28)(H,31,32)(H4,22,23,25,26,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Princeton University
Curated by ChEMBL
| Assay Description
Compound was evaluated for competitive inhibition of recombinant mouse thymidylate synthase |
J Med Chem 35: 4450-4 (1992)
BindingDB Entry DOI: 10.7270/Q2RR1ZV6 |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Mus musculus) | BDBM22590

((2S)-2-[(4-{2-[(6R)-2-amino-4-oxo-1H,4H,5H,6H,7H,8...)Show SMILES Nc1nc2NC[C@H](CCc3ccc(cc3)C(=O)N[C@@H](CCC(O)=O)C(O)=O)Cc2c(=O)[nH]1 Show InChI InChI=1S/C21H25N5O6/c22-21-25-17-14(19(30)26-21)9-12(10-23-17)2-1-11-3-5-13(6-4-11)18(29)24-15(20(31)32)7-8-16(27)28/h3-6,12,15H,1-2,7-10H2,(H,24,29)(H,27,28)(H,31,32)(H4,22,23,25,26,30)/t12-,15+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
| 120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Tested for the inhibition of trifunctional Glycinamide ribonucleotide formyltransferase isolated from murine L1210 cells. |
Bioorg Med Chem Lett 3: 2657-2660 (1993)
Article DOI: 10.1016/S0960-894X(01)80736-8
BindingDB Entry DOI: 10.7270/Q2R49QPW |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM22590

((2S)-2-[(4-{2-[(6R)-2-amino-4-oxo-1H,4H,5H,6H,7H,8...)Show SMILES Nc1nc2NC[C@H](CCc3ccc(cc3)C(=O)N[C@@H](CCC(O)=O)C(O)=O)Cc2c(=O)[nH]1 Show InChI InChI=1S/C21H25N5O6/c22-21-25-17-14(19(30)26-21)9-12(10-23-17)2-1-11-3-5-13(6-4-11)18(29)24-15(20(31)32)7-8-16(27)28/h3-6,12,15H,1-2,7-10H2,(H,24,29)(H,27,28)(H,31,32)(H4,22,23,25,26,30)/t12-,15+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
| 130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Tested for the inhibition of recombinant human monofunctional Glycinamide ribonucleotide formyltransferase |
Bioorg Med Chem Lett 3: 2657-2660 (1993)
Article DOI: 10.1016/S0960-894X(01)80736-8
BindingDB Entry DOI: 10.7270/Q2R49QPW |
More data for this
Ligand-Target Pair | |
Thymidylate synthase
(Mus musculus) | BDBM18796
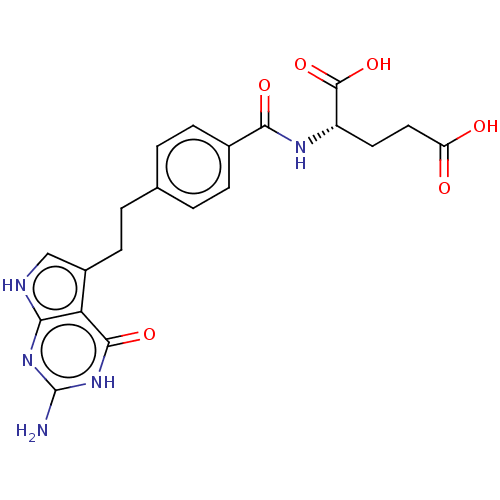
((2S)-2-{[4-(2-{2-amino-4-oxo-1H,4H,7H-pyrrolo[2,3-...)Show SMILES Nc1nc2[nH]cc(CCc3ccc(cc3)C(=O)N[C@@H](CCC(O)=O)C(O)=O)c2c(=O)[nH]1 |r| Show InChI InChI=1S/C20H21N5O6/c21-20-24-16-15(18(29)25-20)12(9-22-16)6-3-10-1-4-11(5-2-10)17(28)23-13(19(30)31)7-8-14(26)27/h1-2,4-5,9,13H,3,6-8H2,(H,23,28)(H,26,27)(H,30,31)(H4,21,22,24,25,29)/t13-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Princeton University
Curated by ChEMBL
| Assay Description
Compound was evaluated for competitive inhibition of recombinant mouse thymidylate synthase |
J Med Chem 35: 4450-4 (1992)
BindingDB Entry DOI: 10.7270/Q2RR1ZV6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Thymidylate synthase
(Homo sapiens (Human)) | BDBM50288820

((S)-2-{(S)-4-[4-(2,4-Diamino-pyrimidin-5-yl)-butyl...)Show SMILES Nc1ncc(CCCCc2ccc(cc2)C(=O)N[C@@H](CCC(O)=O)C(O)=O)c(N)n1 Show InChI InChI=1S/C20H25N5O5/c21-17-14(11-23-20(22)25-17)4-2-1-3-12-5-7-13(8-6-12)18(28)24-15(19(29)30)9-10-16(26)27/h5-8,11,15H,1-4,9-10H2,(H,24,28)(H,26,27)(H,29,30)(H4,21,22,23,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| >5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Binding affinity of the compound against Recombinant human thymidylate synthase (TS). |
Bioorg Med Chem Lett 6: 473-476 (1996)
Article DOI: 10.1016/0960-894X(96)00053-4
BindingDB Entry DOI: 10.7270/Q2GQ6XQ2 |
More data for this
Ligand-Target Pair | |
Thymidylate synthase
(Homo sapiens (Human)) | BDBM50288821

((S)-2-{(S)-4-[3-(2,4-Diamino-pyrimidin-5-yl)-propy...)Show SMILES Nc1ncc(CCCc2ccc(cc2)C(=O)N[C@@H](CCC(O)=O)C(O)=O)c(N)n1 Show InChI InChI=1S/C19H23N5O5/c20-16-13(10-22-19(21)24-16)3-1-2-11-4-6-12(7-5-11)17(27)23-14(18(28)29)8-9-15(25)26/h4-7,10,14H,1-3,8-9H2,(H,23,27)(H,25,26)(H,28,29)(H4,20,21,22,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| >5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Binding affinity of the compound against Recombinant human thymidylate synthase (TS) |
Bioorg Med Chem Lett 6: 473-476 (1996)
Article DOI: 10.1016/0960-894X(96)00053-4
BindingDB Entry DOI: 10.7270/Q2GQ6XQ2 |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Mus musculus) | BDBM50281128
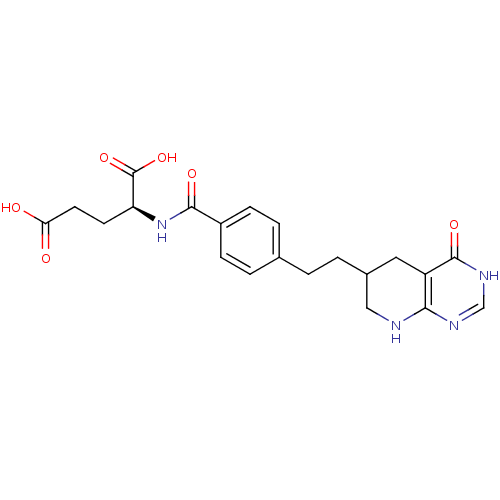
((S)-2-{4-[2-(4-Oxo-3,4,5,6,7,8-hexahydro-pyrido[2,...)Show SMILES OC(=O)CC[C@H](NC(=O)c1ccc(CCC2CNc3nc[nH]c(=O)c3C2)cc1)C(O)=O Show InChI InChI=1S/C21H24N4O6/c26-17(27)8-7-16(21(30)31)25-19(28)14-5-3-12(4-6-14)1-2-13-9-15-18(22-10-13)23-11-24-20(15)29/h3-6,11,13,16H,1-2,7-10H2,(H,25,28)(H,26,27)(H,30,31)(H2,22,23,24,29) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 2.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Tested for the inhibition of trifunctional Glycinamide ribonucleotide formyltransferase isolated from murine L1210 cells. |
Bioorg Med Chem Lett 3: 2657-2660 (1993)
Article DOI: 10.1016/S0960-894X(01)80736-8
BindingDB Entry DOI: 10.7270/Q2R49QPW |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM50281128

((S)-2-{4-[2-(4-Oxo-3,4,5,6,7,8-hexahydro-pyrido[2,...)Show SMILES OC(=O)CC[C@H](NC(=O)c1ccc(CCC2CNc3nc[nH]c(=O)c3C2)cc1)C(O)=O Show InChI InChI=1S/C21H24N4O6/c26-17(27)8-7-16(21(30)31)25-19(28)14-5-3-12(4-6-14)1-2-13-9-15-18(22-10-13)23-11-24-20(15)29/h3-6,11,13,16H,1-2,7-10H2,(H,25,28)(H,26,27)(H,30,31)(H2,22,23,24,29) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 8.21E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Tested for the inhibition of recombinant human monofunctional Glycinamide ribonucleotide formyltransferase |
Bioorg Med Chem Lett 3: 2657-2660 (1993)
Article DOI: 10.1016/S0960-894X(01)80736-8
BindingDB Entry DOI: 10.7270/Q2R49QPW |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM50281129
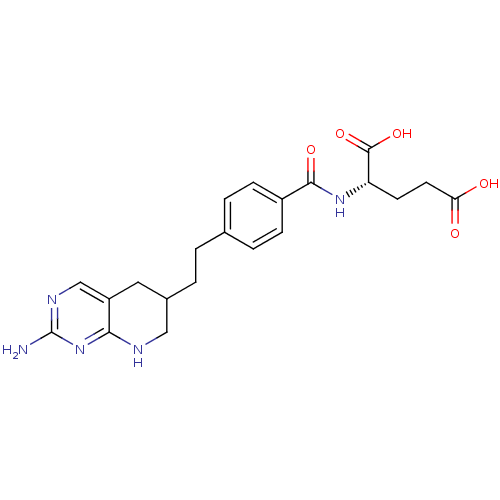
((S)-2-{4-[2-(2-Amino-5,6,7,8-tetrahydro-pyrido[2,3...)Show SMILES Nc1ncc2CC(CCc3ccc(cc3)C(=O)N[C@@H](CCC(O)=O)C(O)=O)CNc2n1 Show InChI InChI=1S/C21H25N5O5/c22-21-24-11-15-9-13(10-23-18(15)26-21)2-1-12-3-5-14(6-4-12)19(29)25-16(20(30)31)7-8-17(27)28/h3-6,11,13,16H,1-2,7-10H2,(H,25,29)(H,27,28)(H,30,31)(H3,22,23,24,26) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 1.86E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Tested for the inhibition of recombinant human monofunctional Glycinamide ribonucleotide formyltransferase |
Bioorg Med Chem Lett 3: 2657-2660 (1993)
Article DOI: 10.1016/S0960-894X(01)80736-8
BindingDB Entry DOI: 10.7270/Q2R49QPW |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Mus musculus) | BDBM50281129

((S)-2-{4-[2-(2-Amino-5,6,7,8-tetrahydro-pyrido[2,3...)Show SMILES Nc1ncc2CC(CCc3ccc(cc3)C(=O)N[C@@H](CCC(O)=O)C(O)=O)CNc2n1 Show InChI InChI=1S/C21H25N5O5/c22-21-24-11-15-9-13(10-23-18(15)26-21)2-1-12-3-5-14(6-4-12)19(29)25-16(20(30)31)7-8-17(27)28/h3-6,11,13,16H,1-2,7-10H2,(H,25,29)(H,27,28)(H,30,31)(H3,22,23,24,26) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 65 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Tested for the inhibition against the human dihydrofolate reductase |
Bioorg Med Chem Lett 3: 2657-2660 (1993)
Article DOI: 10.1016/S0960-894X(01)80736-8
BindingDB Entry DOI: 10.7270/Q2R49QPW |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data