 Found 43012 hits with Last Name = 'ai' and Initial = 'j'
Found 43012 hits with Last Name = 'ai' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Substance-P receptor
(Homo sapiens (Human)) | BDBM50295070
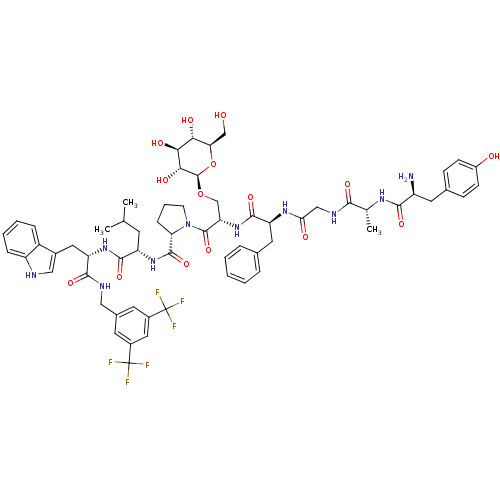
((S)-1-((2S,5S,11R,14S)-14-amino-5-benzyl-15-(4-hyd...)Show SMILES CC(C)C[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@H](CO[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O)NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C63H76F6N10O15/c1-32(2)20-44(57(89)76-46(25-37-28-71-43-13-8-7-12-41(37)43)56(88)72-27-36-21-38(62(64,65)66)26-39(22-36)63(67,68)69)77-59(91)48-14-9-19-79(48)60(92)47(31-93-61-53(85)52(84)51(83)49(30-80)94-61)78-58(90)45(24-34-10-5-4-6-11-34)75-50(82)29-73-54(86)33(3)74-55(87)42(70)23-35-15-17-40(81)18-16-35/h4-8,10-13,15-18,21-22,26,28,32-33,42,44-49,51-53,61,71,80-81,83-85H,9,14,19-20,23-25,27,29-31,70H2,1-3H3,(H,72,88)(H,73,86)(H,74,87)(H,75,82)(H,76,89)(H,77,91)(H,78,90)/t33-,42+,44+,45+,46+,47+,48+,49-,51-,52+,53-,61-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]substance P from human NK1 receptor expressed in CHO cell membrane |
J Med Chem 52: 5164-75 (2010)
Article DOI: 10.1021/jm900473p
BindingDB Entry DOI: 10.7270/Q2MC90ZZ |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50295070

((S)-1-((2S,5S,11R,14S)-14-amino-5-benzyl-15-(4-hyd...)Show SMILES CC(C)C[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@H](CO[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O)NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C63H76F6N10O15/c1-32(2)20-44(57(89)76-46(25-37-28-71-43-13-8-7-12-41(37)43)56(88)72-27-36-21-38(62(64,65)66)26-39(22-36)63(67,68)69)77-59(91)48-14-9-19-79(48)60(92)47(31-93-61-53(85)52(84)51(83)49(30-80)94-61)78-58(90)45(24-34-10-5-4-6-11-34)75-50(82)29-73-54(86)33(3)74-55(87)42(70)23-35-15-17-40(81)18-16-35/h4-8,10-13,15-18,21-22,26,28,32-33,42,44-49,51-53,61,71,80-81,83-85H,9,14,19-20,23-25,27,29-31,70H2,1-3H3,(H,72,88)(H,73,86)(H,74,87)(H,75,82)(H,76,89)(H,77,91)(H,78,90)/t33-,42+,44+,45+,46+,47+,48+,49-,51-,52+,53-,61-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]substance P from human NK1 receptor expressed in CHO cell membrane |
J Med Chem 52: 5164-75 (2010)
Article DOI: 10.1021/jm900473p
BindingDB Entry DOI: 10.7270/Q2MC90ZZ |
More data for this
Ligand-Target Pair | |
Caspase-3
(Homo sapiens (Human)) | BDBM50160957
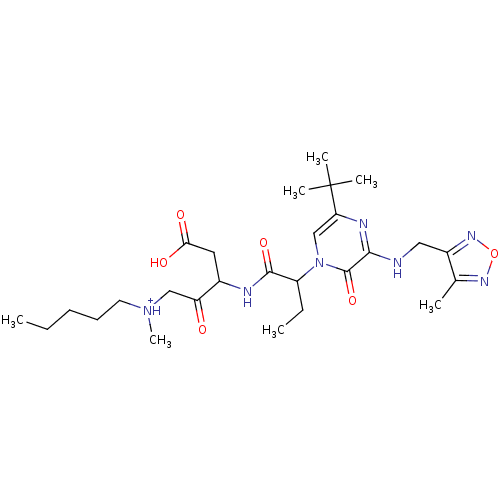
(CHEMBL179503 | [3-(2-{5-tert-Butyl-3-[(4-methyl-fu...)Show SMILES CCCCC[NH+](C)CC(=O)C(CC(O)=O)NC(=O)C(CC)n1cc(nc(NCc2nonc2C)c1=O)C(C)(C)C Show InChI InChI=1S/C27H43N7O6/c1-8-10-11-12-33(7)15-21(35)18(13-23(36)37)29-25(38)20(9-2)34-16-22(27(4,5)6)30-24(26(34)39)28-14-19-17(3)31-40-32-19/h16,18,20H,8-15H2,1-7H3,(H,28,30)(H,29,38)(H,36,37)/p+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory concentration against recombinant human caspase-3 in neuronal precursor (NT2) cells |
Bioorg Med Chem Lett 15: 1173-80 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.006
BindingDB Entry DOI: 10.7270/Q2D50MGS |
More data for this
Ligand-Target Pair | |
Trypsin
(Homo sapiens (Human)) | BDBM50507483
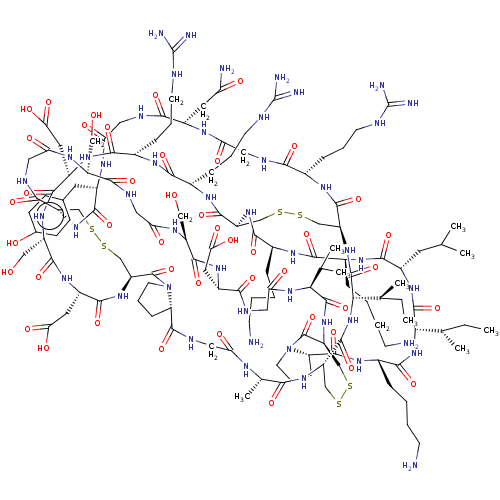
(CHEMBL4530379)Show SMILES CC[C@H](C)[C@@H]1NC(=O)[C@H](CCCCN)NC(=O)[C@@H]2CCCN2C(=O)[C@@H]2CSSC[C@@H]3NC(=O)[C@H](C)NC(=O)CNC(=O)[C@@H]4CCCN4C(=O)[C@@H]4CSSC[C@H](NC(=O)[C@H](Cc5ccc(O)cc5)NC(=O)CNC(=O)[C@H](CC(N)=O)NC(=O)CNC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CSSC[C@H](NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CC(C)C)NC1=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(=O)N4)NC(=O)[C@@H](NC3=O)[C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(=O)NCC(=O)N[C@@H](C(C)C)C(=O)N2 |r| Show InChI InChI=1S/C136H220N46O44S6/c1-10-66(7)105-130(223)170-77(43-64(3)4)118(211)164-72(23-12-15-35-137)113(206)162-73(24-13-16-36-138)116(209)174-87-59-228-229-60-88-125(218)161-71(26-18-38-147-134(141)142)108(201)150-50-97(190)158-79(45-94(140)187)109(202)151-51-96(189)157-78(44-69-31-33-70(186)34-32-69)119(212)173-86-58-227-231-62-90(176-121(214)82(48-103(198)199)169-123(216)85(57-185)171-120(213)81(47-102(196)197)168-115(208)76(28-20-40-149-136(145)146)163-114(207)75(165-124(87)217)27-19-39-148-135(143)144)132(225)181-41-21-29-92(181)127(220)155-49-95(188)156-68(9)107(200)172-89(126(219)180-106(67(8)11-2)131(224)175-88)61-230-232-63-91(133(226)182-42-22-30-93(182)128(221)166-74(117(210)179-105)25-14-17-37-139)177-129(222)104(65(5)6)178-100(193)54-152-110(203)80(46-101(194)195)167-122(215)84(56-184)160-99(192)52-153-111(204)83(55-183)159-98(191)53-154-112(86)205/h31-34,64-68,71-93,104-106,183-186H,10-30,35-63,137-139H2,1-9H3,(H2,140,187)(H,150,201)(H,151,202)(H,152,203)(H,153,204)(H,154,205)(H,155,220)(H,156,188)(H,157,189)(H,158,190)(H,159,191)(H,160,192)(H,161,218)(H,162,206)(H,163,207)(H,164,211)(H,165,217)(H,166,221)(H,167,215)(H,168,208)(H,169,216)(H,170,223)(H,171,213)(H,172,200)(H,173,212)(H,174,209)(H,175,224)(H,176,214)(H,177,222)(H,178,193)(H,179,210)(H,180,219)(H,194,195)(H,196,197)(H,198,199)(H4,141,142,147)(H4,143,144,148)(H4,145,146,149)/t66-,67-,68-,71-,72-,73-,74-,75-,76-,77-,78-,79-,80-,81-,82-,83-,84-,85-,86-,87-,88-,89-,90-,91-,92-,93-,104-,105-,106-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Inhibition of trypsin (unknown origin) |
ACS Med Chem Lett 9: 1258-1262 (2018)
Article DOI: 10.1021/acsmedchemlett.8b00422
BindingDB Entry DOI: 10.7270/Q2Q81HCD |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50408679

(CHEMBL5287792)Show SMILES COc1cc2nc(nc(N)c2cc1OC)N(C)CCCCCCN(C)C(=O)c1ccc(CN(C)C)o1 Show InChI InChI=1S/C26H38N6O4/c1-30(2)17-18-11-12-21(36-18)25(33)31(3)13-9-7-8-10-14-32(4)26-28-20-16-23(35-6)22(34-5)15-19(20)24(27)29-26/h11-12,15-16H,7-10,13-14,17H2,1-6H3,(H2,27,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| | 0.00260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Antagonist activity at cloned muscarinic M2 receptor-Gqi5 chimeric protein expressed in CHO cells assessed as acetylcholine-induced change in cytosol... |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50295069
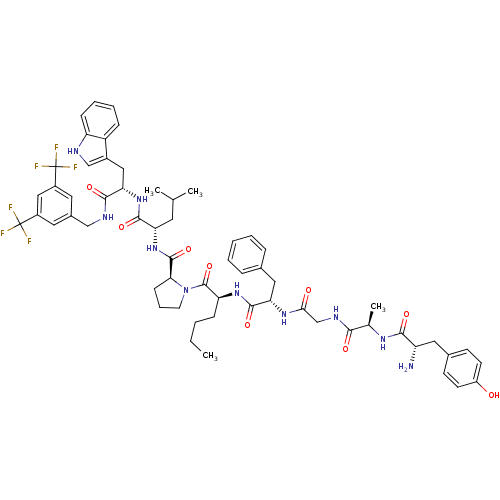
((S)-1-((2S,5S,11R,14S)-14-amino-5-benzyl-2-butyl-1...)Show SMILES CCCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C60H72F6N10O9/c1-5-6-16-46(73-56(83)48(28-36-13-8-7-9-14-36)72-51(78)33-70-52(79)35(4)71-53(80)44(67)27-37-19-21-42(77)22-20-37)58(85)76-23-12-18-50(76)57(84)75-47(24-34(2)3)55(82)74-49(29-39-32-68-45-17-11-10-15-43(39)45)54(81)69-31-38-25-40(59(61,62)63)30-41(26-38)60(64,65)66/h7-11,13-15,17,19-22,25-26,30,32,34-35,44,46-50,68,77H,5-6,12,16,18,23-24,27-29,31,33,67H2,1-4H3,(H,69,81)(H,70,79)(H,71,80)(H,72,78)(H,73,83)(H,74,82)(H,75,84)/t35-,44+,46+,47+,48+,49+,50+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]substance P from human NK1 receptor expressed in CHO cell membrane |
J Med Chem 52: 5164-75 (2010)
Article DOI: 10.1021/jm900473p
BindingDB Entry DOI: 10.7270/Q2MC90ZZ |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50295069

((S)-1-((2S,5S,11R,14S)-14-amino-5-benzyl-2-butyl-1...)Show SMILES CCCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C60H72F6N10O9/c1-5-6-16-46(73-56(83)48(28-36-13-8-7-9-14-36)72-51(78)33-70-52(79)35(4)71-53(80)44(67)27-37-19-21-42(77)22-20-37)58(85)76-23-12-18-50(76)57(84)75-47(24-34(2)3)55(82)74-49(29-39-32-68-45-17-11-10-15-43(39)45)54(81)69-31-38-25-40(59(61,62)63)30-41(26-38)60(64,65)66/h7-11,13-15,17,19-22,25-26,30,32,34-35,44,46-50,68,77H,5-6,12,16,18,23-24,27-29,31,33,67H2,1-4H3,(H,69,81)(H,70,79)(H,71,80)(H,72,78)(H,73,83)(H,74,82)(H,75,84)/t35-,44+,46+,47+,48+,49+,50+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]substance P from human NK1 receptor expressed in CHO cell membrane |
J Med Chem 52: 5164-75 (2010)
Article DOI: 10.1021/jm900473p
BindingDB Entry DOI: 10.7270/Q2MC90ZZ |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50007522
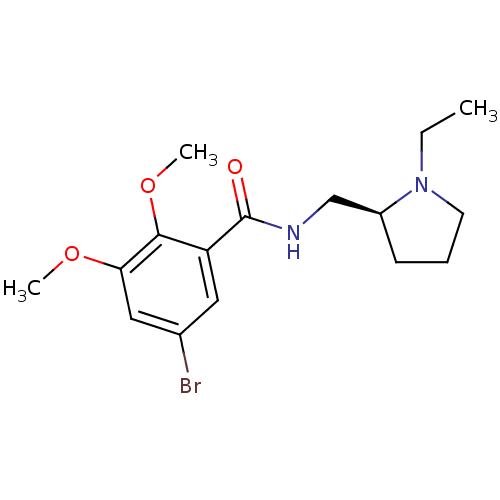
(5-Bromo-N-(1-ethyl-pyrrolidin-2-ylmethyl)-2,3-dime...)Show InChI InChI=1S/C16H23BrN2O3/c1-4-19-7-5-6-12(19)10-18-16(20)13-8-11(17)9-14(21-2)15(13)22-3/h8-9,12H,4-7,10H2,1-3H3,(H,18,20)/t12-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institutet
Curated by ChEMBL
| Assay Description
Binding affinity to dopamine D2 receptor (unknown origin) |
Bioorg Med Chem 16: 6467-73 (2008)
Article DOI: 10.1016/j.bmc.2008.05.039
BindingDB Entry DOI: 10.7270/Q2QN66JM |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50408509

(CHEMBL5277326)Show InChI InChI=1S/C20H24N2OS/c1-4-18(23)15-10-11-20-17(14-15)22(13-7-12-21(2)3)16-8-5-6-9-19(16)24-20/h5-6,8-11,14H,4,7,12-13H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| | 0.00340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Antagonist activity at AT1 receptor in rat aortic rings |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50498577
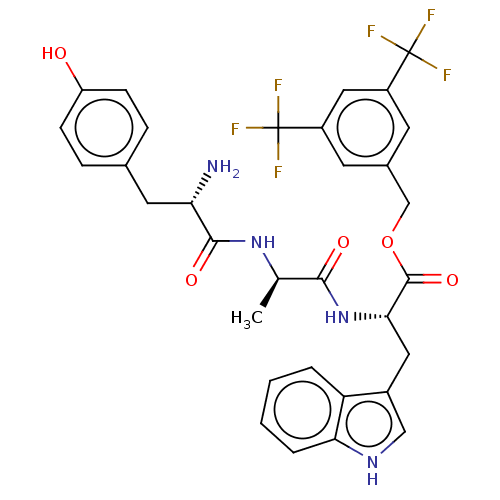
(CHEMBL3609619)Show SMILES C[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)OCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C32H30F6N4O5/c1-17(41-29(45)25(39)12-18-6-8-23(43)9-7-18)28(44)42-27(13-20-15-40-26-5-3-2-4-24(20)26)30(46)47-16-19-10-21(31(33,34)35)14-22(11-19)32(36,37)38/h2-11,14-15,17,25,27,40,43H,12-13,16,39H2,1H3,(H,41,45)(H,42,44)/t17-,25+,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]-substance P from human NK1 receptor transfected in CHO cells |
Bioorg Med Chem Lett 25: 3716-20 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.030
BindingDB Entry DOI: 10.7270/Q2R78J7N |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50498575
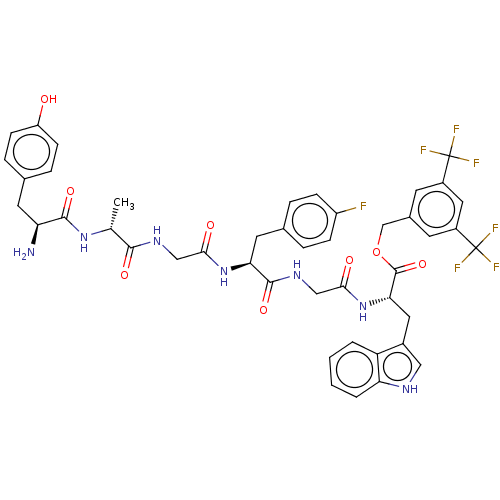
(CHEMBL3608939)Show SMILES C[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)NCC(=O)N[C@@H](Cc1ccc(F)cc1)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)OCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C45H44F7N7O8/c1-24(57-41(64)34(53)16-25-8-12-32(60)13-9-25)40(63)55-21-38(61)58-36(17-26-6-10-31(46)11-7-26)42(65)56-22-39(62)59-37(18-28-20-54-35-5-3-2-4-33(28)35)43(66)67-23-27-14-29(44(47,48)49)19-30(15-27)45(50,51)52/h2-15,19-20,24,34,36-37,54,60H,16-18,21-23,53H2,1H3,(H,55,63)(H,56,65)(H,57,64)(H,58,61)(H,59,62)/t24-,34+,36+,37+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]-substance P from human NK1 receptor transfected in CHO cells |
Bioorg Med Chem Lett 25: 3716-20 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.030
BindingDB Entry DOI: 10.7270/Q2R78J7N |
More data for this
Ligand-Target Pair | |
Caspase-3
(Homo sapiens (Human)) | BDBM50160974
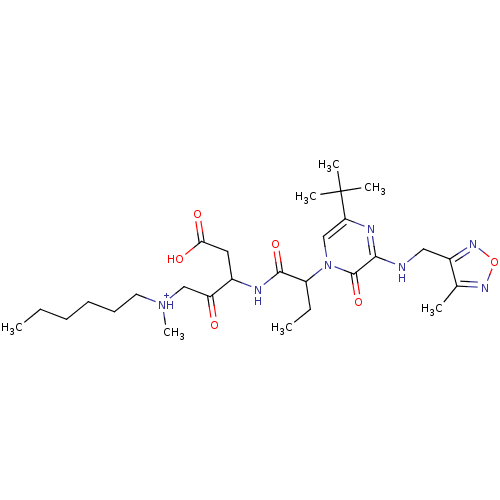
(CHEMBL366927 | [3-(2-{5-tert-Butyl-3-[(4-methyl-fu...)Show SMILES CCCCCC[NH+](C)CC(=O)C(CC(O)=O)NC(=O)C(CC)n1cc(nc(NCc2nonc2C)c1=O)C(C)(C)C Show InChI InChI=1S/C28H45N7O6/c1-8-10-11-12-13-34(7)16-22(36)19(14-24(37)38)30-26(39)21(9-2)35-17-23(28(4,5)6)31-25(27(35)40)29-15-20-18(3)32-41-33-20/h17,19,21H,8-16H2,1-7H3,(H,29,31)(H,30,39)(H,37,38)/p+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Frosst Centre for Therapeutic Research
Curated by ChEMBL
| Assay Description
Inhibitory concentration against recombinant human caspase-3 in neuronal precursor (NT2) cells |
Bioorg Med Chem Lett 15: 1173-80 (2005)
Article DOI: 10.1016/j.bmcl.2004.12.006
BindingDB Entry DOI: 10.7270/Q2D50MGS |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50323869
((S)-1-((4S,7S,13R)-13-((S)-2-amino-3-(4-hydroxyphe...)Show SMILES CC(C)C[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)CNC(=O)[C@@H](NC(=O)[C@@H](N)Cc2ccc(O)cc2)C(C)(C)SSC1(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C61H72F6N10O9S2/c1-33(2)23-44(53(82)73-46(28-37-31-69-43-16-11-10-15-41(37)43)52(81)70-30-36-24-38(60(62,63)64)29-39(25-36)61(65,66)67)74-55(84)47-17-12-22-77(47)57(86)50-59(5,6)88-87-58(3,4)49(75-51(80)42(68)26-35-18-20-40(78)21-19-35)56(85)71-32-48(79)72-45(54(83)76-50)27-34-13-8-7-9-14-34/h7-11,13-16,18-21,24-25,29,31,33,42,44-47,49-50,69,78H,12,17,22-23,26-28,30,32,68H2,1-6H3,(H,70,81)(H,71,85)(H,72,79)(H,73,82)(H,74,84)(H,75,80)(H,76,83)/t42-,44-,45-,46-,47-,49+,50-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]substance P frome human NK1 receptor |
J Med Chem 53: 5491-501 (2010)
Article DOI: 10.1021/jm100157m
BindingDB Entry DOI: 10.7270/Q21N81BD |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50323869

((S)-1-((4S,7S,13R)-13-((S)-2-amino-3-(4-hydroxyphe...)Show SMILES CC(C)C[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)CNC(=O)[C@@H](NC(=O)[C@@H](N)Cc2ccc(O)cc2)C(C)(C)SSC1(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C61H72F6N10O9S2/c1-33(2)23-44(53(82)73-46(28-37-31-69-43-16-11-10-15-41(37)43)52(81)70-30-36-24-38(60(62,63)64)29-39(25-36)61(65,66)67)74-55(84)47-17-12-22-77(47)57(86)50-59(5,6)88-87-58(3,4)49(75-51(80)42(68)26-35-18-20-40(78)21-19-35)56(85)71-32-48(79)72-45(54(83)76-50)27-34-13-8-7-9-14-34/h7-11,13-16,18-21,24-25,29,31,33,42,44-47,49-50,69,78H,12,17,22-23,26-28,30,32,68H2,1-6H3,(H,70,81)(H,71,85)(H,72,79)(H,73,82)(H,74,84)(H,75,80)(H,76,83)/t42-,44-,45-,46-,47-,49+,50-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]substance P frome human NK1 receptor |
J Med Chem 53: 5491-501 (2010)
Article DOI: 10.1021/jm100157m
BindingDB Entry DOI: 10.7270/Q21N81BD |
More data for this
Ligand-Target Pair | |
Melanocyte-stimulating hormone receptor
(Homo sapiens (Human)) | BDBM50590042
(CHEMBL5175981)Show SMILES [H][C@]12CSc3nnc(SC[C@]([H])(NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc4c[nH]c5ccccc45)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc4ccccc4)NC(=O)[C@H](Cc4cnc[nH]4)NC(=O)CNC(=O)[C@H](CCCC)NC1=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c4ccccc14)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N2)c1CCC2C(COC(=O)NCCN)C2CCc31 |r| | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >0.00549 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00793
BindingDB Entry DOI: 10.7270/Q20C50R5 |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50439327
(CHEMBL2419544)Show SMILES C[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)NCC(=O)N[C@@H](Cc1ccccc1)C(=O)NCC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)OCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C45H45F6N7O8/c1-25(56-41(63)34(52)17-27-11-13-32(59)14-12-27)40(62)54-22-38(60)57-36(18-26-7-3-2-4-8-26)42(64)55-23-39(61)58-37(19-29-21-53-35-10-6-5-9-33(29)35)43(65)66-24-28-15-30(44(46,47)48)20-31(16-28)45(49,50)51/h2-16,20-21,25,34,36-37,53,59H,17-19,22-24,52H2,1H3,(H,54,62)(H,55,64)(H,56,63)(H,57,60)(H,58,61)/t25-,34+,36+,37+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00570 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]substance P from human NK1 receptor expressed in CHO cells |
Bioorg Med Chem Lett 23: 4975-8 (2013)
Article DOI: 10.1016/j.bmcl.2013.06.065
BindingDB Entry DOI: 10.7270/Q2222W64 |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50264406
((S)-1-((2S,5S,11R,14S)-14-amino-5-benzyl-15-(4-hyd...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C59H70F6N10O9S/c1-33(2)23-46(54(81)73-48(28-38-31-67-44-14-9-8-13-42(38)44)53(80)68-30-37-24-39(58(60,61)62)29-40(25-37)59(63,64)65)74-56(83)49-15-10-21-75(49)57(84)45(20-22-85-4)72-55(82)47(27-35-11-6-5-7-12-35)71-50(77)32-69-51(78)34(3)70-52(79)43(66)26-36-16-18-41(76)19-17-36/h5-9,11-14,16-19,24-25,29,31,33-34,43,45-49,67,76H,10,15,20-23,26-28,30,32,66H2,1-4H3,(H,68,80)(H,69,78)(H,70,79)(H,71,77)(H,72,82)(H,73,81)(H,74,83)/t34-,43+,45+,46+,47+,48+,49+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]substance P from human NK1 receptor expressed in CHO cell membrane by liquid scintillation counting |
J Med Chem 51: 6334-47 (2008)
Article DOI: 10.1021/jm800389v
BindingDB Entry DOI: 10.7270/Q26T0MF1 |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50264406

((S)-1-((2S,5S,11R,14S)-14-amino-5-benzyl-15-(4-hyd...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C59H70F6N10O9S/c1-33(2)23-46(54(81)73-48(28-38-31-67-44-14-9-8-13-42(38)44)53(80)68-30-37-24-39(58(60,61)62)29-40(25-37)59(63,64)65)74-56(83)49-15-10-21-75(49)57(84)45(20-22-85-4)72-55(82)47(27-35-11-6-5-7-12-35)71-50(77)32-69-51(78)34(3)70-52(79)43(66)26-36-16-18-41(76)19-17-36/h5-9,11-14,16-19,24-25,29,31,33-34,43,45-49,67,76H,10,15,20-23,26-28,30,32,66H2,1-4H3,(H,68,80)(H,69,78)(H,70,79)(H,71,77)(H,72,82)(H,73,81)(H,74,83)/t34-,43+,45+,46+,47+,48+,49+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]substance P frome human NK1 receptor |
J Med Chem 53: 5491-501 (2010)
Article DOI: 10.1021/jm100157m
BindingDB Entry DOI: 10.7270/Q21N81BD |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50264406

((S)-1-((2S,5S,11R,14S)-14-amino-5-benzyl-15-(4-hyd...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C59H70F6N10O9S/c1-33(2)23-46(54(81)73-48(28-38-31-67-44-14-9-8-13-42(38)44)53(80)68-30-37-24-39(58(60,61)62)29-40(25-37)59(63,64)65)74-56(83)49-15-10-21-75(49)57(84)45(20-22-85-4)72-55(82)47(27-35-11-6-5-7-12-35)71-50(77)32-69-51(78)34(3)70-52(79)43(66)26-36-16-18-41(76)19-17-36/h5-9,11-14,16-19,24-25,29,31,33-34,43,45-49,67,76H,10,15,20-23,26-28,30,32,66H2,1-4H3,(H,68,80)(H,69,78)(H,70,79)(H,71,77)(H,72,82)(H,73,81)(H,74,83)/t34-,43+,45+,46+,47+,48+,49+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]substance P from human NK1 receptor expressed in CHO cell membranes |
Bioorg Med Chem 17: 7337-43 (2009)
Article DOI: 10.1016/j.bmc.2009.08.035
BindingDB Entry DOI: 10.7270/Q2CJ8DK5 |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50264406

((S)-1-((2S,5S,11R,14S)-14-amino-5-benzyl-15-(4-hyd...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C59H70F6N10O9S/c1-33(2)23-46(54(81)73-48(28-38-31-67-44-14-9-8-13-42(38)44)53(80)68-30-37-24-39(58(60,61)62)29-40(25-37)59(63,64)65)74-56(83)49-15-10-21-75(49)57(84)45(20-22-85-4)72-55(82)47(27-35-11-6-5-7-12-35)71-50(77)32-69-51(78)34(3)70-52(79)43(66)26-36-16-18-41(76)19-17-36/h5-9,11-14,16-19,24-25,29,31,33-34,43,45-49,67,76H,10,15,20-23,26-28,30,32,66H2,1-4H3,(H,68,80)(H,69,78)(H,70,79)(H,71,77)(H,72,82)(H,73,81)(H,74,83)/t34-,43+,45+,46+,47+,48+,49+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]substance P from human NK1 receptor expressed in CHO cell membranes |
Bioorg Med Chem 17: 7337-43 (2009)
Article DOI: 10.1016/j.bmc.2009.08.035
BindingDB Entry DOI: 10.7270/Q2CJ8DK5 |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50341318
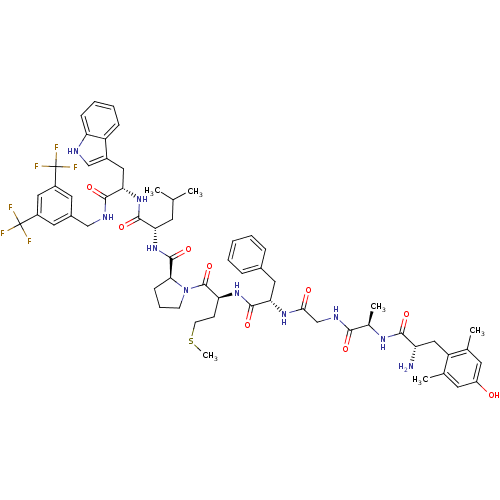
((S)-1-((2S,5S,11R,14S)-14-amino-5-benzyl-15-(4-hyd...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C61H74F6N10O9S/c1-33(2)21-48(56(83)75-50(27-39-31-69-46-16-11-10-15-43(39)46)55(82)70-30-38-24-40(60(62,63)64)28-41(25-38)61(65,66)67)76-58(85)51-17-12-19-77(51)59(86)47(18-20-87-6)74-57(84)49(26-37-13-8-7-9-14-37)73-52(79)32-71-53(80)36(5)72-54(81)45(68)29-44-34(3)22-42(78)23-35(44)4/h7-11,13-16,22-25,28,31,33,36,45,47-51,69,78H,12,17-21,26-27,29-30,32,68H2,1-6H3,(H,70,82)(H,71,80)(H,72,81)(H,73,79)(H,74,84)(H,75,83)(H,76,85)/t36-,45+,47+,48+,49+,50+,51+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00790 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]substance P from human NK1 receptor expressed in CHO cells |
J Med Chem 54: 2029-38 (2011)
Article DOI: 10.1021/jm101023r
BindingDB Entry DOI: 10.7270/Q2862GRK |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50341318

((S)-1-((2S,5S,11R,14S)-14-amino-5-benzyl-15-(4-hyd...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C61H74F6N10O9S/c1-33(2)21-48(56(83)75-50(27-39-31-69-46-16-11-10-15-43(39)46)55(82)70-30-38-24-40(60(62,63)64)28-41(25-38)61(65,66)67)76-58(85)51-17-12-19-77(51)59(86)47(18-20-87-6)74-57(84)49(26-37-13-8-7-9-14-37)73-52(79)32-71-53(80)36(5)72-54(81)45(68)29-44-34(3)22-42(78)23-35(44)4/h7-11,13-16,22-25,28,31,33,36,45,47-51,69,78H,12,17-21,26-27,29-30,32,68H2,1-6H3,(H,70,82)(H,71,80)(H,72,81)(H,73,79)(H,74,84)(H,75,83)(H,76,85)/t36-,45+,47+,48+,49+,50+,51+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00790 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]substance P from human NK1 receptor expressed in CHO cells |
J Med Chem 54: 2029-38 (2011)
Article DOI: 10.1021/jm101023r
BindingDB Entry DOI: 10.7270/Q2862GRK |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Bos taurus (bovine)) | BDBM50124947
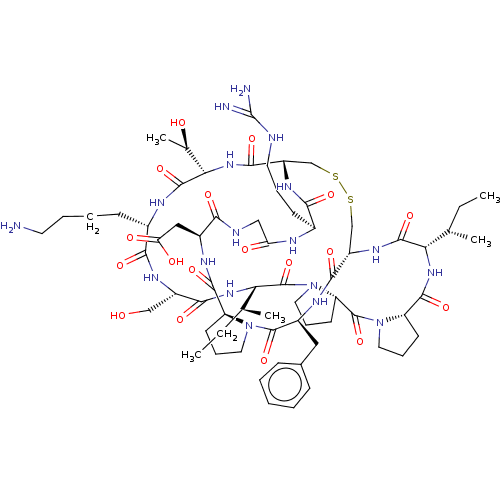
(CHEMBL453539)Show SMILES [H][C@@]12CCCN1C(=O)[C@H](Cc1ccccc1)NC(=O)[C@]1([H])CSSC[C@]([H])(NC(=O)[C@H](CCCNC(N)=N)NC(=O)CNC(=O)[C@H](CC(O)=O)NC2=O)C(=O)N[C@@]([H])([C@@H](C)O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@]([H])([C@@H](C)CC)C(=O)N2CCC[C@@]2([H])C(=O)N2CCC[C@@]2([H])C(=O)N[C@@]([H])([C@@H](C)CC)C(=O)N1 |r| Show InChI InChI=1S/C67H104N18O18S2/c1-6-35(3)51-62(99)79-44-33-104-105-34-45(78-55(92)39(20-13-25-71-67(69)70)73-49(88)31-72-54(91)41(30-50(89)90)75-60(97)46-21-14-26-83(46)64(101)42(76-58(44)95)29-38-17-9-8-10-18-38)59(96)82-53(37(5)87)63(100)74-40(19-11-12-24-68)56(93)77-43(32-86)57(94)81-52(36(4)7-2)66(103)85-28-16-23-48(85)65(102)84-27-15-22-47(84)61(98)80-51/h8-10,17-18,35-37,39-48,51-53,86-87H,6-7,11-16,19-34,68H2,1-5H3,(H,72,91)(H,73,88)(H,74,100)(H,75,97)(H,76,95)(H,77,93)(H,78,92)(H,79,99)(H,80,98)(H,81,94)(H,82,96)(H,89,90)(H4,69,70,71)/t35-,36-,37+,39-,40-,41-,42-,43-,44-,45-,46-,47-,48-,51-,52-,53-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Inhibition of bovine beta-trypsin using Boc-QAR-MCA as substrate measured every 30 secs for 10 mins |
Eur J Med Chem 155: 695-704 (2018)
Article DOI: 10.1016/j.ejmech.2018.06.029
BindingDB Entry DOI: 10.7270/Q29026CK |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM209097
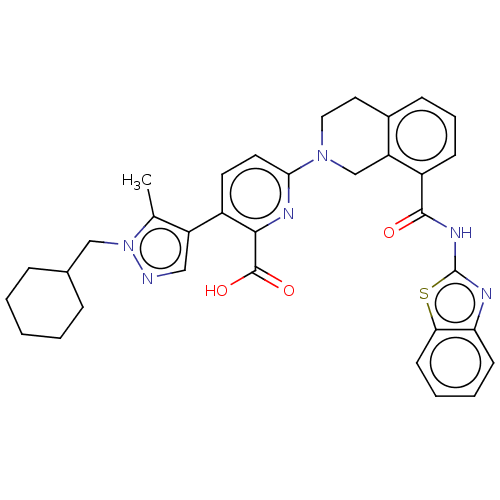
(US9266877, 43)Show SMILES Cc1c(cnn1CC1CCCCC1)-c1ccc(nc1C(O)=O)N1CCc2cccc(C(=O)Nc3nc4ccccc4s3)c2C1 Show InChI InChI=1S/C34H34N6O3S/c1-21-26(18-35-40(21)19-22-8-3-2-4-9-22)24-14-15-30(37-31(24)33(42)43)39-17-16-23-10-7-11-25(27(23)20-39)32(41)38-34-36-28-12-5-6-13-29(28)44-34/h5-7,10-15,18,22H,2-4,8-9,16-17,19-20H2,1H3,(H,42,43)(H,36,38,41) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of F-Bak (GQVGRQLAIIGDK(6-FAM)INR-amide probe binding to BCL-xl (unknown origin) incubated for 1 hr by TR-FRET assay |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.9b00568
BindingDB Entry DOI: 10.7270/Q2542S8D |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50439329
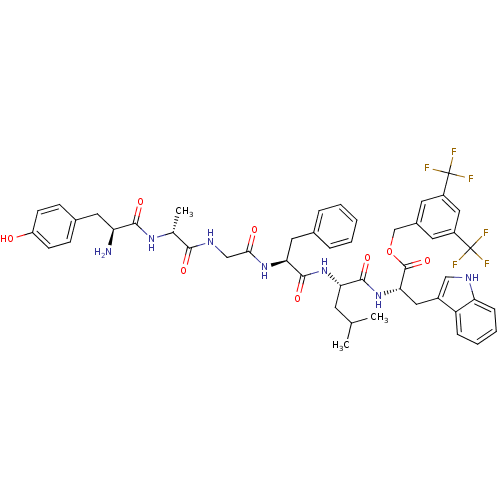
(CHEMBL2419542)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)OCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C49H53F6N7O8/c1-27(2)17-39(45(67)62-41(22-32-24-57-38-12-8-7-11-36(32)38)47(69)70-26-31-18-33(48(50,51)52)23-34(19-31)49(53,54)55)61-46(68)40(21-29-9-5-4-6-10-29)60-42(64)25-58-43(65)28(3)59-44(66)37(56)20-30-13-15-35(63)16-14-30/h4-16,18-19,23-24,27-28,37,39-41,57,63H,17,20-22,25-26,56H2,1-3H3,(H,58,65)(H,59,66)(H,60,64)(H,61,68)(H,62,67)/t28-,37+,39+,40+,41+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]substance P from human NK1 receptor expressed in CHO cells |
Bioorg Med Chem Lett 23: 4975-8 (2013)
Article DOI: 10.1016/j.bmcl.2013.06.065
BindingDB Entry DOI: 10.7270/Q2222W64 |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50162797
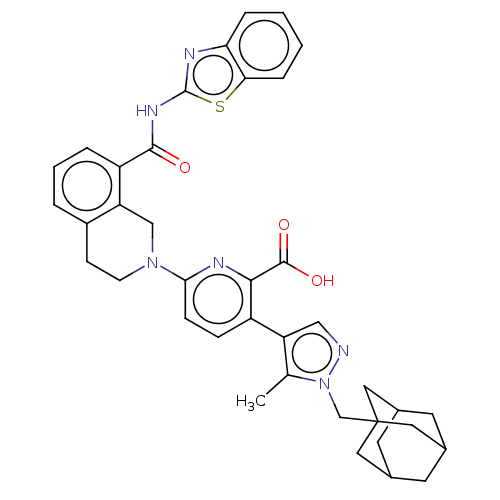
(CHEMBL3793424)Show SMILES Cc1c(cnn1CC12CC3CC(CC(C3)C1)C2)-c1ccc(nc1C(O)=O)N1CCc2cccc(C(=O)Nc3nc4ccccc4s3)c2C1 |TLB:14:9:16:12.13.15,14:13:16:10.9.8,THB:12:11:8:14.13.15,12:13:10.11.16:8| Show InChI InChI=1S/C38H38N6O3S/c1-22-29(19-39-44(22)21-38-16-23-13-24(17-38)15-25(14-23)18-38)27-9-10-33(41-34(27)36(46)47)43-12-11-26-5-4-6-28(30(26)20-43)35(45)42-37-40-31-7-2-3-8-32(31)48-37/h2-10,19,23-25H,11-18,20-21H2,1H3,(H,46,47)(H,40,42,45) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of F-Bak (GQVGRQLAIIGDK(6-FAM)INR-amide probe binding to BCL-xl (unknown origin) incubated for 1 hr by TR-FRET assay |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.9b00568
BindingDB Entry DOI: 10.7270/Q2542S8D |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50030754
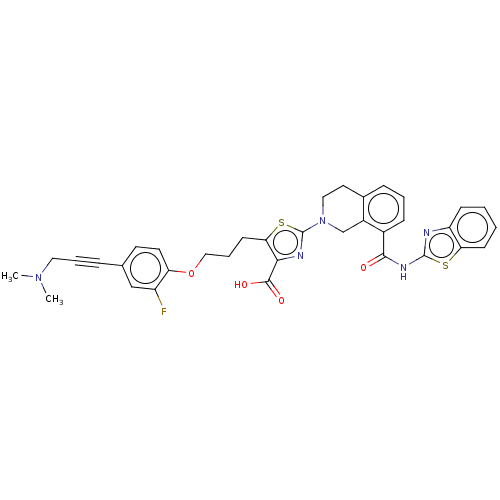
(CHEMBL3342332)Show SMILES CN(C)CC#Cc1ccc(OCCCc2sc(nc2C(O)=O)N2CCc3cccc(C(=O)Nc4nc5ccccc5s4)c3C2)c(F)c1 Show InChI InChI=1S/C35H32FN5O4S2/c1-40(2)17-6-8-22-14-15-28(26(36)20-22)45-19-7-13-30-31(33(43)44)38-35(47-30)41-18-16-23-9-5-10-24(25(23)21-41)32(42)39-34-37-27-11-3-4-12-29(27)46-34/h3-5,9-12,14-15,20H,7,13,16-19,21H2,1-2H3,(H,43,44)(H,37,39,42) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of BCL-XL (unknown origin) incubated for 1 hr by TR-FRET assay |
ACS Med Chem Lett 5: 1088-93 (2014)
Article DOI: 10.1021/ml5001867
BindingDB Entry DOI: 10.7270/Q2VX0J43 |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50030757
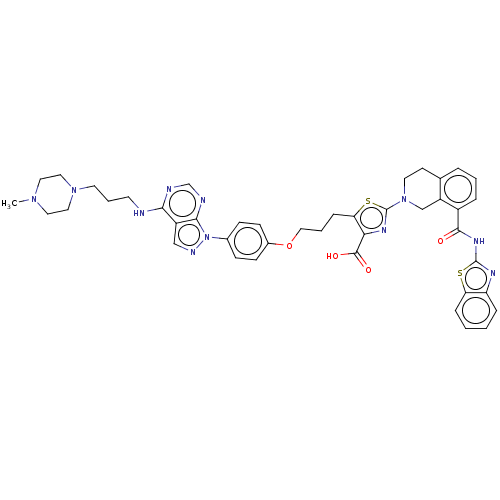
(CHEMBL3342196)Show SMILES CN1CCN(CCCNc2ncnc3n(ncc23)-c2ccc(OCCCc3sc(nc3C(O)=O)N3CCc4cccc(C(=O)Nc5nc6ccccc6s5)c4C3)cc2)CC1 Show InChI InChI=1S/C43H45N11O4S2/c1-51-20-22-52(23-21-51)18-6-17-44-38-32-25-47-54(39(32)46-27-45-38)29-12-14-30(15-13-29)58-24-5-11-36-37(41(56)57)49-43(60-36)53-19-16-28-7-4-8-31(33(28)26-53)40(55)50-42-48-34-9-2-3-10-35(34)59-42/h2-4,7-10,12-15,25,27H,5-6,11,16-24,26H2,1H3,(H,56,57)(H,44,45,46)(H,48,50,55) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of BCL-XL (unknown origin) incubated for 1 hr by TR-FRET assay |
ACS Med Chem Lett 5: 1088-93 (2014)
Article DOI: 10.1021/ml5001867
BindingDB Entry DOI: 10.7270/Q2VX0J43 |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50030758
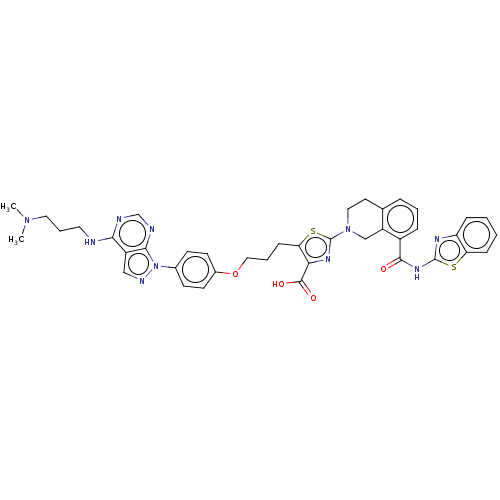
(CHEMBL3342195)Show SMILES CN(C)CCCNc1ncnc2n(ncc12)-c1ccc(OCCCc2sc(nc2C(O)=O)N2CCc3cccc(C(=O)Nc4nc5ccccc5s4)c3C2)cc1 Show InChI InChI=1S/C40H40N10O4S2/c1-48(2)19-7-18-41-35-29-22-44-50(36(29)43-24-42-35)26-13-15-27(16-14-26)54-21-6-12-33-34(38(52)53)46-40(56-33)49-20-17-25-8-5-9-28(30(25)23-49)37(51)47-39-45-31-10-3-4-11-32(31)55-39/h3-5,8-11,13-16,22,24H,6-7,12,17-21,23H2,1-2H3,(H,52,53)(H,41,42,43)(H,45,47,51) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of BCL-XL (unknown origin) incubated for 1 hr by TR-FRET assay |
ACS Med Chem Lett 5: 1088-93 (2014)
Article DOI: 10.1021/ml5001867
BindingDB Entry DOI: 10.7270/Q2VX0J43 |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50561528
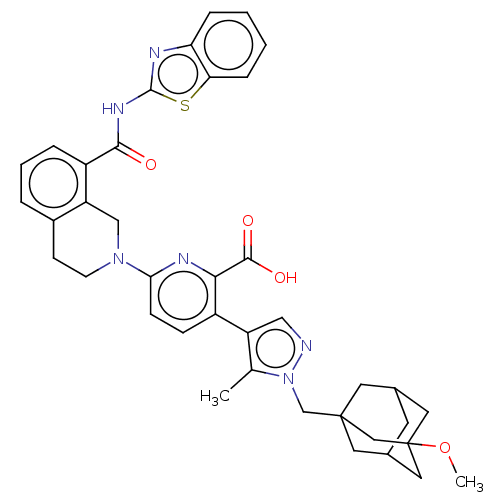
(CHEMBL4762875)Show SMILES COC12CC3CC(CC(Cn4ncc(c4C)-c4ccc(nc4C(O)=O)N4CCc5cccc(C(=O)Nc6nc7ccccc7s6)c5C4)(C3)C1)C2 |TLB:1:2:5.4.47:7,5:6:4.3.47:48,THB:5:4:6.49.7:48,3:4:7:49.2.48,3:2:5.4.47:7| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of F-Bak (GQVGRQLAIIGDK(6-FAM)INR-amide probe binding to BCL-xl (unknown origin) incubated for 1 hr by TR-FRET assay |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.9b00568
BindingDB Entry DOI: 10.7270/Q2542S8D |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50030759
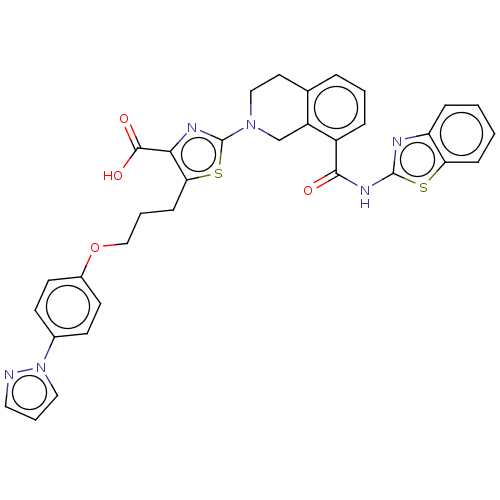
(CHEMBL3342194)Show SMILES OC(=O)c1nc(sc1CCCOc1ccc(cc1)-n1cccn1)N1CCc2cccc(C(=O)Nc3nc4ccccc4s3)c2C1 Show InChI InChI=1S/C33H28N6O4S2/c40-30(37-32-35-26-8-1-2-9-27(26)44-32)24-7-3-6-21-15-18-38(20-25(21)24)33-36-29(31(41)42)28(45-33)10-4-19-43-23-13-11-22(12-14-23)39-17-5-16-34-39/h1-3,5-9,11-14,16-17H,4,10,15,18-20H2,(H,41,42)(H,35,37,40) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of BCL-XL (unknown origin) incubated for 1 hr by TR-FRET assay |
ACS Med Chem Lett 5: 1088-93 (2014)
Article DOI: 10.1021/ml5001867
BindingDB Entry DOI: 10.7270/Q2VX0J43 |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50030752
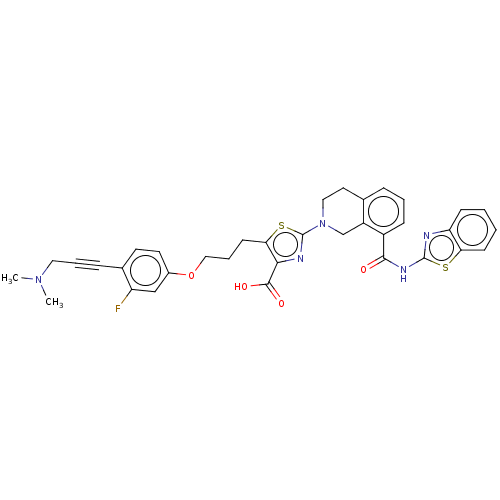
(CHEMBL3342333)Show SMILES CN(C)CC#Cc1ccc(OCCCc2sc(nc2C(O)=O)N2CCc3cccc(C(=O)Nc4nc5ccccc5s4)c3C2)cc1F Show InChI InChI=1S/C35H32FN5O4S2/c1-40(2)17-6-9-23-14-15-24(20-27(23)36)45-19-7-13-30-31(33(43)44)38-35(47-30)41-18-16-22-8-5-10-25(26(22)21-41)32(42)39-34-37-28-11-3-4-12-29(28)46-34/h3-5,8,10-12,14-15,20H,7,13,16-19,21H2,1-2H3,(H,43,44)(H,37,39,42) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of BCL-XL (unknown origin) incubated for 1 hr by TR-FRET assay |
ACS Med Chem Lett 5: 1088-93 (2014)
Article DOI: 10.1021/ml5001867
BindingDB Entry DOI: 10.7270/Q2VX0J43 |
More data for this
Ligand-Target Pair | |
Purine nucleoside phosphorylase
(Homo sapiens (Human)) | BDBM22109
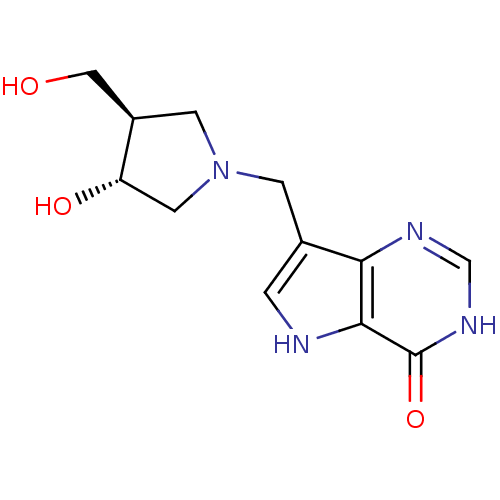
(7-{[(3R,4R)-3-hydroxy-4-(hydroxymethyl)pyrrolidin-...)Show SMILES OC[C@H]1CN(Cc2c[nH]c3c2nc[nH]c3=O)C[C@@H]1O |r| Show InChI InChI=1S/C12H16N4O3/c17-5-8-3-16(4-9(8)18)2-7-1-13-11-10(7)14-6-15-12(11)19/h1,6,8-9,13,17-18H,2-5H2,(H,14,15,19)/t8-,9+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| 0.0110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Industrial Research Limited
Curated by ChEMBL
| Assay Description
Inhibition of human purine nucleoside phosphorylase by xanthine-oxidase coupled assay |
J Med Chem 51: 5880-4 (2008)
Article DOI: 10.1021/jm800792b
BindingDB Entry DOI: 10.7270/Q2VQ32H9 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Estrogen receptor beta
(Homo sapiens (Human)) | BDBM50471255
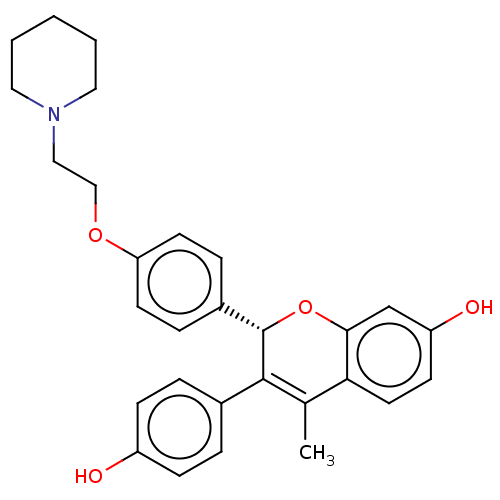
(Acolbifene | EM-652 | SCH-57068)Show SMILES CC1=C([C@@H](Oc2cc(O)ccc12)c1ccc(OCCN2CCCCC2)cc1)c1ccc(O)cc1 |t:1| Show InChI InChI=1S/C29H31NO4/c1-20-26-14-11-24(32)19-27(26)34-29(28(20)21-5-9-23(31)10-6-21)22-7-12-25(13-8-22)33-18-17-30-15-3-2-4-16-30/h5-14,19,29,31-32H,2-4,15-18H2,1H3/t29-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Le Centre Hospitalier Universitaire de Qu£bec
Curated by ChEMBL
| Assay Description
Apparent binding affinity against estradiol-stimulated T-47D cell proliferation |
J Med Chem 40: 2117-22 (1997)
Article DOI: 10.1021/jm970095o
BindingDB Entry DOI: 10.7270/Q2474DKC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450717

(CHEMBL317087)Show InChI InChI=1S/C9H10BrClN2O/c10-8-3-7(4-13-9(8)11)14-5-6-1-2-12-6/h3-4,6,12H,1-2,5H2/t6-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50577958
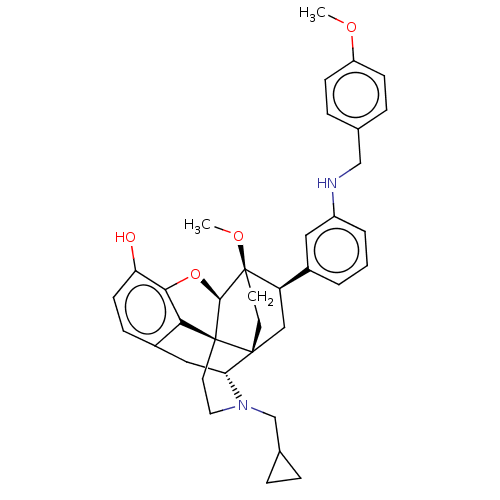
(CHEMBL4859512)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC6CC6)CC[C@@]14[C@@]51CC[C@@]2(OC)[C@H](C1)c1cccc(NCc2ccc(OC)cc2)c1)ccc3O |r,THB:10:9:5.4.6:17| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]U69,593 from human kappa opioid receptor expressed in CHO cell membrane measured after 30 mins by liquid scintillation counting m... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01082
BindingDB Entry DOI: 10.7270/Q2NZ8CG1 |
More data for this
Ligand-Target Pair | |
Estrogen receptor beta
(Homo sapiens (Human)) | BDBM50471254
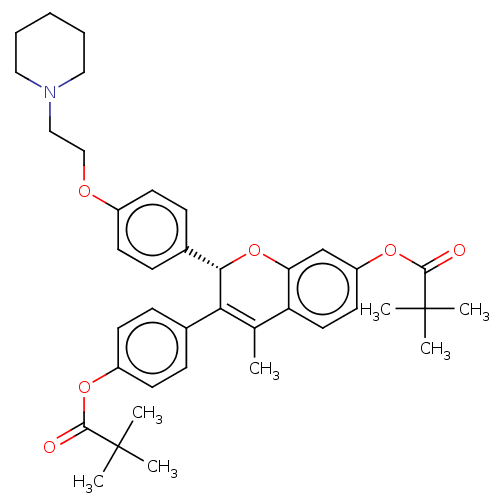
(CHEMBL308234)Show SMILES CC1=C([C@@H](Oc2cc(OC(=O)C(C)(C)C)ccc12)c1ccc(OCCN2CCCCC2)cc1)c1ccc(OC(=O)C(C)(C)C)cc1 |r,t:1| Show InChI InChI=1S/C39H47NO6/c1-26-32-20-19-31(45-37(42)39(5,6)7)25-33(32)46-35(34(26)27-11-17-30(18-12-27)44-36(41)38(2,3)4)28-13-15-29(16-14-28)43-24-23-40-21-9-8-10-22-40/h11-20,25,35H,8-10,21-24H2,1-7H3/t35-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Le Centre Hospitalier Universitaire de Qu£bec
Curated by ChEMBL
| Assay Description
Apparent binding affinity against estradiol-stimulated T-47D cell proliferation |
J Med Chem 40: 2117-22 (1997)
Article DOI: 10.1021/jm970095o
BindingDB Entry DOI: 10.7270/Q2474DKC |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50498573
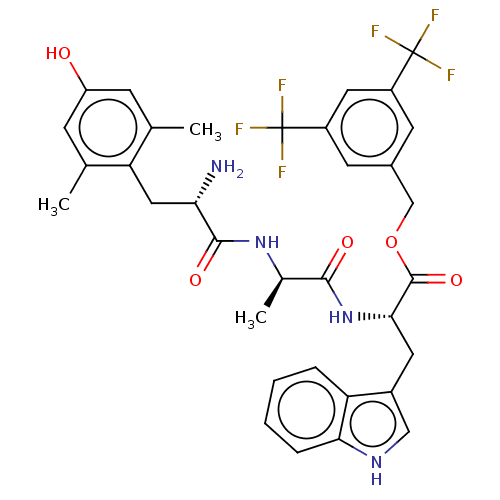
(CHEMBL3609620)Show SMILES C[C@@H](NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)OCc1cc(cc(c1)C(F)(F)F)C(F)(F)F |r| Show InChI InChI=1S/C34H34F6N4O5/c1-17-8-24(45)9-18(2)26(17)14-27(41)31(47)43-19(3)30(46)44-29(12-21-15-42-28-7-5-4-6-25(21)28)32(48)49-16-20-10-22(33(35,36)37)13-23(11-20)34(38,39)40/h4-11,13,15,19,27,29,42,45H,12,14,16,41H2,1-3H3,(H,43,47)(H,44,46)/t19-,27+,29+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]-substance P from human NK1 receptor transfected in CHO cells |
Bioorg Med Chem Lett 25: 3716-20 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.030
BindingDB Entry DOI: 10.7270/Q2R78J7N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM207217
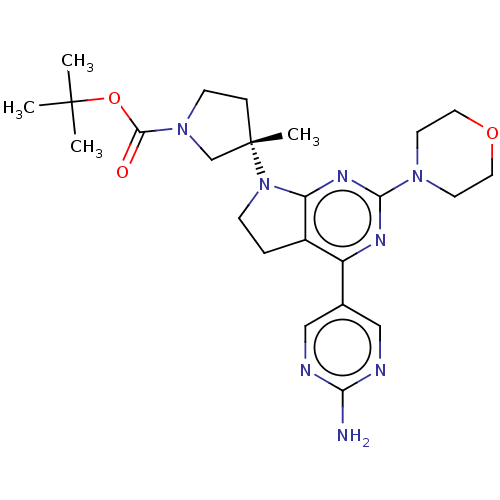
(US9260439, 194 | US9260439, 238 | US9260439, 239)Show SMILES CC(C)(C)OC(=O)N1CC[C@@](C)(C1)N1CCc2c1nc(nc2-c1cnc(N)nc1)N1CCOCC1 |r| Show InChI InChI=1S/C24H34N8O3/c1-23(2,3)35-22(33)31-8-6-24(4,15-31)32-7-5-17-18(16-13-26-20(25)27-14-16)28-21(29-19(17)32)30-9-11-34-12-10-30/h13-14H,5-12,15H2,1-4H3,(H2,25,26,27)/t24-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| <0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length PI3K p110alpha/p85alpha (322 to 600) expressed in baculovirus infected Sf21 cells using phosphatidylinosi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01652
BindingDB Entry DOI: 10.7270/Q2TH8RC9 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM340384
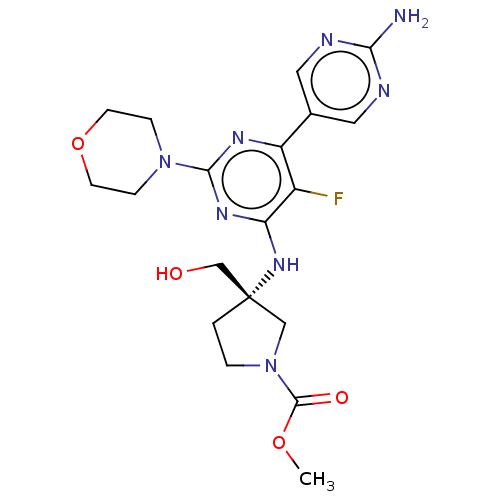
(US9758538, Example 72)Show SMILES COC(=O)N1CC[C@@](CO)(C1)Nc1nc(nc(c1F)-c1cnc(N)nc1)N1CCOCC1 |r| Show InChI InChI=1S/C19H25FN8O4/c1-31-18(30)28-3-2-19(10-28,11-29)26-15-13(20)14(12-8-22-16(21)23-9-12)24-17(25-15)27-4-6-32-7-5-27/h8-9,29H,2-7,10-11H2,1H3,(H2,21,22,23)(H,24,25,26)/t19-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length PI3K p110alpha/p85alpha (322 to 600) expressed in baculovirus infected Sf21 cells using phosphatidylinosi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01652
BindingDB Entry DOI: 10.7270/Q2TH8RC9 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM207196
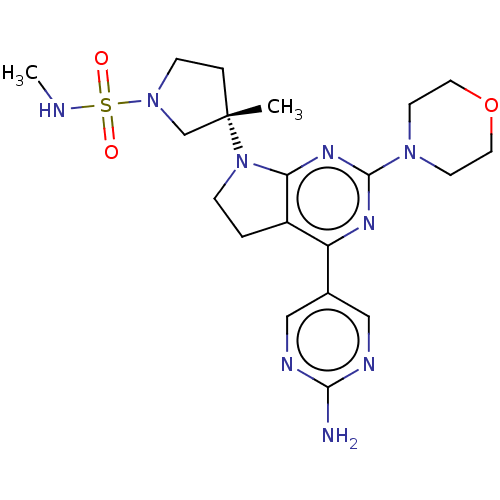
(US9260439, 173)Show SMILES CNS(=O)(=O)N1CC[C@@](C)(C1)N1CCc2c1nc(nc2-c1cnc(N)nc1)N1CCOCC1 |r| Show InChI InChI=1S/C20H29N9O3S/c1-20(4-6-28(13-20)33(30,31)22-2)29-5-3-15-16(14-11-23-18(21)24-12-14)25-19(26-17(15)29)27-7-9-32-10-8-27/h11-12,22H,3-10,13H2,1-2H3,(H2,21,23,24)/t20-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length PI3K p110alpha/p85alpha (322 to 600) expressed in baculovirus infected Sf21 cells using phosphatidylinosi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01652
BindingDB Entry DOI: 10.7270/Q2TH8RC9 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM207378
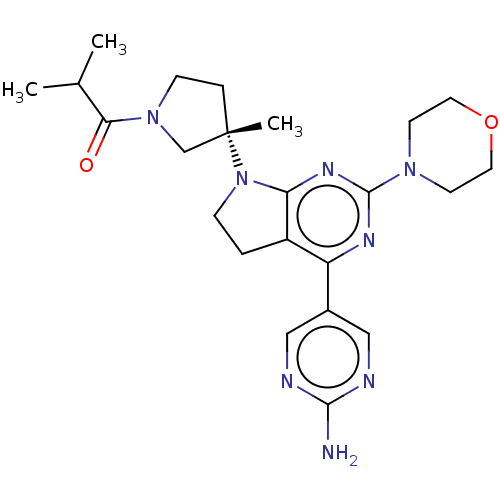
(US9260439, 262)Show SMILES CC(C)C(=O)N1CC[C@@](C)(C1)N1CCc2c1nc(nc2-c1cnc(N)nc1)N1CCOCC1 Show InChI InChI=1S/C23H32N8O2/c1-15(2)20(32)30-7-5-23(3,14-30)31-6-4-17-18(16-12-25-21(24)26-13-16)27-22(28-19(17)31)29-8-10-33-11-9-29/h12-13,15H,4-11,14H2,1-3H3,(H2,24,25,26)/t23-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length PI3K p110alpha/p85alpha (322 to 600) expressed in baculovirus infected Sf21 cells using phosphatidylinosi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01652
BindingDB Entry DOI: 10.7270/Q2TH8RC9 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM340336
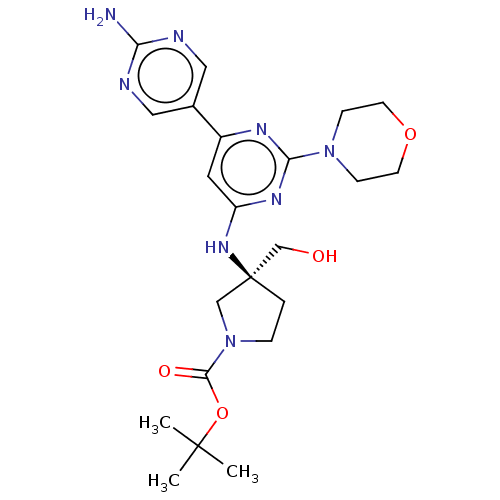
(US9758538, Example 24)Show SMILES CC(C)(C)OC(=O)N1CC[C@@](CO)(C1)Nc1cc(nc(n1)N1CCOCC1)-c1cnc(N)nc1 |r| Show InChI InChI=1S/C22H32N8O4/c1-21(2,3)34-20(32)30-5-4-22(13-30,14-31)28-17-10-16(15-11-24-18(23)25-12-15)26-19(27-17)29-6-8-33-9-7-29/h10-12,31H,4-9,13-14H2,1-3H3,(H2,23,24,25)(H,26,27,28)/t22-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length PI3K p110alpha/p85alpha (322 to 600) expressed in baculovirus infected Sf21 cells using phosphatidylinosi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01652
BindingDB Entry DOI: 10.7270/Q2TH8RC9 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM340314
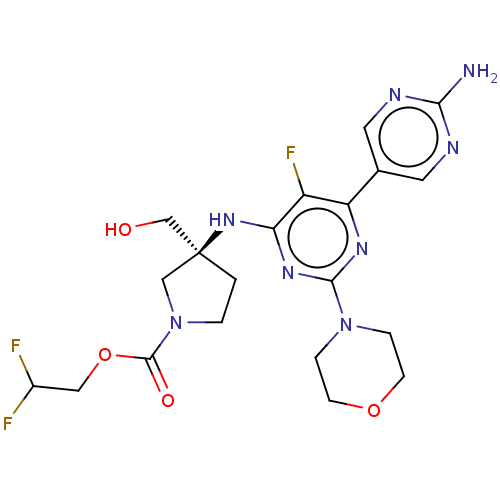
((Scheme A): Preparation of 2,2-difluoroethyl (3S)-...)Show SMILES Nc1ncc(cn1)-c1nc(nc(N[C@@]2(CO)CCN(C2)C(=O)OCC(F)F)c1F)N1CCOCC1 |r| Show InChI InChI=1S/C20H25F3N8O4/c21-13(22)9-35-19(33)31-2-1-20(10-31,11-32)29-16-14(23)15(12-7-25-17(24)26-8-12)27-18(28-16)30-3-5-34-6-4-30/h7-8,13,32H,1-6,9-11H2,(H2,24,25,26)(H,27,28,29)/t20-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| <0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length PI3K p110alpha/p85alpha (322 to 600) expressed in baculovirus infected Sf21 cells using phosphatidylinosi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01652
BindingDB Entry DOI: 10.7270/Q2TH8RC9 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM340346

(US9758538, Example 34)Show SMILES COC(=O)N1CC[C@@](CO)(C1)Nc1cc(nc(n1)N1CCOCC1)-c1cnc(N)nc1 |r| Show InChI InChI=1S/C19H26N8O4/c1-30-18(29)27-3-2-19(11-27,12-28)25-15-8-14(13-9-21-16(20)22-10-13)23-17(24-15)26-4-6-31-7-5-26/h8-10,28H,2-7,11-12H2,1H3,(H2,20,21,22)(H,23,24,25)/t19-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length PI3K p110alpha/p85alpha (322 to 600) expressed in baculovirus infected Sf21 cells using phosphatidylinosi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01652
BindingDB Entry DOI: 10.7270/Q2TH8RC9 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(BOVINE) | BDBM50060602

(2-[3-((2R,8aS)-1,4-Dioxo-hexahydro-pyrrolo[1,2-a]p...)Show InChI InChI=1S/C13H18N4O4/c14-10(18)6-15-5-3-9(12(15)20)17-7-11(19)16-4-1-2-8(16)13(17)21/h8-9H,1-7H2,(H2,14,18)/t8-,9?/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota
Curated by ChEMBL
| Assay Description
Inhibitory constant of compound in presence of Gpp(NH)p (Pre treated with 1 nM) calculated for the high affinity components of the [3H]spiroperidol b... |
J Med Chem 40: 3594-600 (1997)
Article DOI: 10.1021/jm970328b
BindingDB Entry DOI: 10.7270/Q2HX1DBD |
More data for this
Ligand-Target Pair | |
WD repeat-containing protein 5
(Homo sapiens (Human)) | BDBM50605356
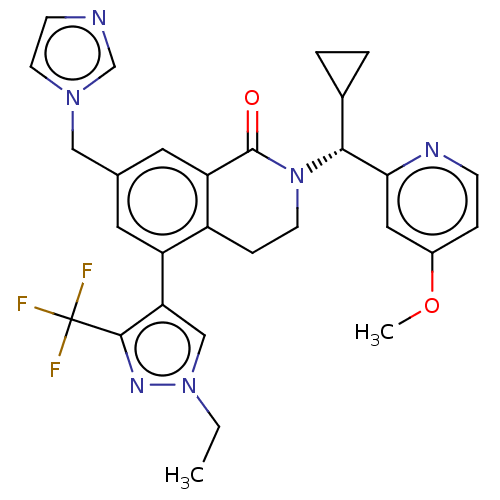
(CHEMBL5170071)Show SMILES CCn1cc(c(n1)C(F)(F)F)-c1cc(Cn2ccnc2)cc2C(=O)N(CCc12)[C@H](C1CC1)c1cc(OC)ccn1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00195
BindingDB Entry DOI: 10.7270/Q2JQ153M |
More data for this
Ligand-Target Pair | |
WD repeat-containing protein 5
(Homo sapiens (Human)) | BDBM50605355
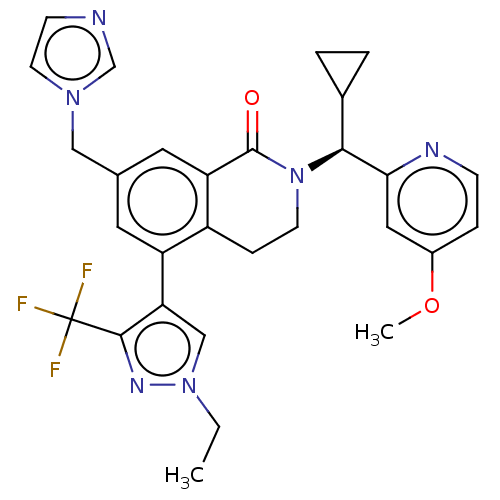
(CHEMBL5170717)Show SMILES CCn1cc(c(n1)C(F)(F)F)-c1cc(Cn2ccnc2)cc2C(=O)N(CCc12)[C@@H](C1CC1)c1cc(OC)ccn1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00195
BindingDB Entry DOI: 10.7270/Q2JQ153M |
More data for this
Ligand-Target Pair | |
WD repeat-containing protein 5
(Homo sapiens (Human)) | BDBM50605350
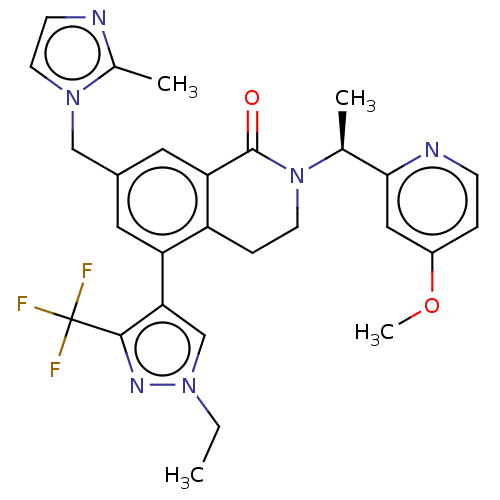
(CHEMBL5177656)Show SMILES CCn1cc(c(n1)C(F)(F)F)-c1cc(Cn2ccnc2C)cc2C(=O)N(CCc12)[C@@H](C)c1cc(OC)ccn1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00195
BindingDB Entry DOI: 10.7270/Q2JQ153M |
More data for this
Ligand-Target Pair | |
WD repeat-containing protein 5
(Homo sapiens (Human)) | BDBM50605353
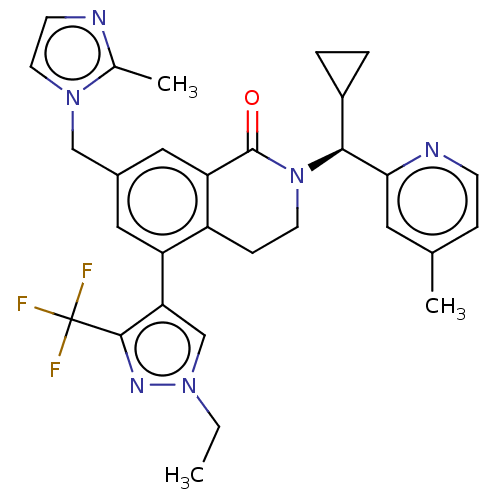
(CHEMBL5190717)Show SMILES CCn1cc(c(n1)C(F)(F)F)-c1cc(Cn2ccnc2C)cc2C(=O)N(CCc12)[C@@H](C1CC1)c1cc(C)ccn1 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| <0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00195
BindingDB Entry DOI: 10.7270/Q2JQ153M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data