 Found 30753 hits with Last Name = 'kim' and Initial = 'j'
Found 30753 hits with Last Name = 'kim' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Isoform PDE10A2 of cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A (PDE10A2)
(Homo sapiens (Human)) | BDBM194104
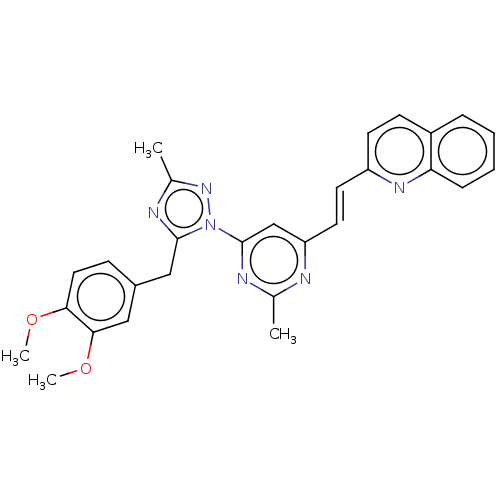
(US9200001, 18)Show SMILES COc1ccc(Cc2nc(C)nn2-c2cc(\C=C\c3ccc4ccccc4n3)nc(C)n2)cc1OC Show InChI InChI=1S/C28H26N6O2/c1-18-29-23(13-12-22-11-10-21-7-5-6-8-24(21)32-22)17-28(30-18)34-27(31-19(2)33-34)16-20-9-14-25(35-3)26(15-20)36-4/h5-15,17H,16H2,1-4H3/b13-12+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp.
US Patent
| Assay Description
In a typical experiment the PDE10 inhibitory activity of the compounds
of the present invention was determined in accordance with the following
exp... |
US Patent US9200001 (2015)
BindingDB Entry DOI: 10.7270/Q2BP01M3 |
More data for this
Ligand-Target Pair | |
Isoform PDE10A2 of cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A (PDE10A2)
(Homo sapiens (Human)) | BDBM194105
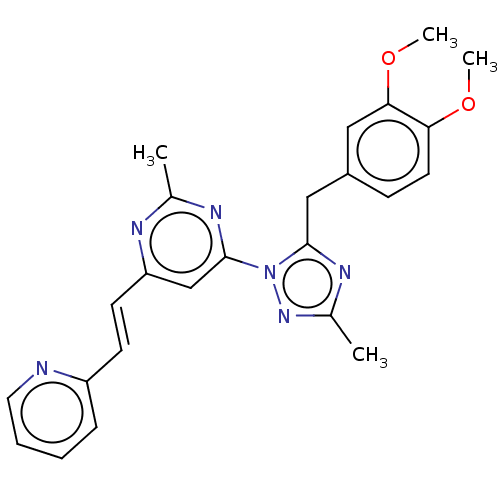
(US9200001, 19)Show SMILES COc1ccc(Cc2nc(C)nn2-c2cc(\C=C\c3ccccn3)nc(C)n2)cc1OC Show InChI InChI=1S/C24H24N6O2/c1-16-26-20(10-9-19-7-5-6-12-25-19)15-24(27-16)30-23(28-17(2)29-30)14-18-8-11-21(31-3)22(13-18)32-4/h5-13,15H,14H2,1-4H3/b10-9+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp.
US Patent
| Assay Description
In a typical experiment the PDE10 inhibitory activity of the compounds
of the present invention was determined in accordance with the following
exp... |
US Patent US9200001 (2015)
BindingDB Entry DOI: 10.7270/Q2BP01M3 |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM14361
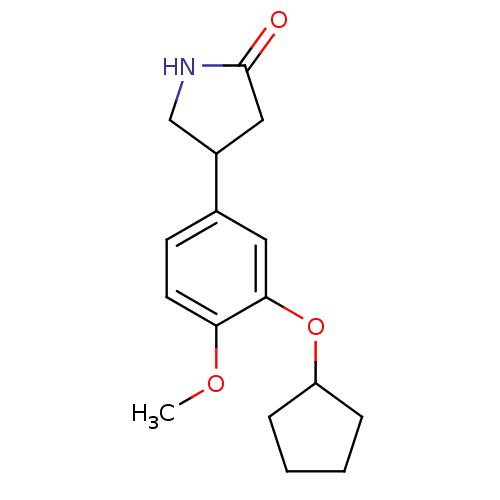
((R,S)-Rolipram | 4-(3-cyclopentyloxy-4-methoxy-phe...)Show InChI InChI=1S/C16H21NO3/c1-19-14-7-6-11(12-9-16(18)17-10-12)8-15(14)20-13-4-2-3-5-13/h6-8,12-13H,2-5,9-10H2,1H3,(H,17,18) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.00230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
Isoform PDE10A2 of cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A (PDE10A2)
(Homo sapiens (Human)) | BDBM194113
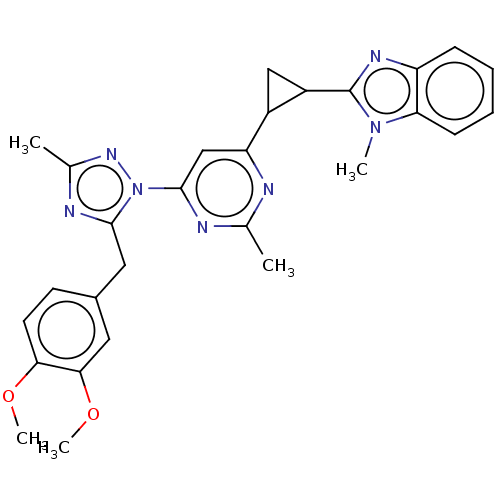
(US9200001, 28)Show SMILES COc1ccc(Cc2nc(C)nn2-c2cc(nc(C)n2)C2CC2c2nc3ccccc3n2C)cc1OC Show InChI InChI=1S/C28H29N7O2/c1-16-29-22(19-14-20(19)28-32-21-8-6-7-9-23(21)34(28)3)15-27(30-16)35-26(31-17(2)33-35)13-18-10-11-24(36-4)25(12-18)37-5/h6-12,15,19-20H,13-14H2,1-5H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp.
US Patent
| Assay Description
In a typical experiment the PDE10 inhibitory activity of the compounds
of the present invention was determined in accordance with the following
exp... |
US Patent US9200001 (2015)
BindingDB Entry DOI: 10.7270/Q2BP01M3 |
More data for this
Ligand-Target Pair | |
Isoform PDE10A2 of cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A (PDE10A2)
(Homo sapiens (Human)) | BDBM194090
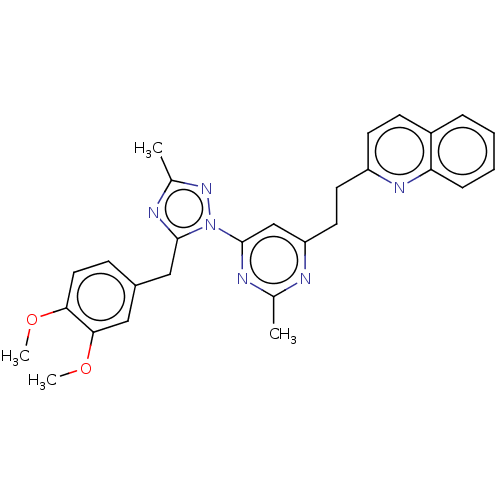
(US9200001, 3)Show SMILES COc1ccc(Cc2nc(C)nn2-c2cc(CCc3ccc4ccccc4n3)nc(C)n2)cc1OC Show InChI InChI=1S/C28H28N6O2/c1-18-29-23(13-12-22-11-10-21-7-5-6-8-24(21)32-22)17-28(30-18)34-27(31-19(2)33-34)16-20-9-14-25(35-3)26(15-20)36-4/h5-11,14-15,17H,12-13,16H2,1-4H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.00400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp.
US Patent
| Assay Description
In a typical experiment the PDE10 inhibitory activity of the compounds
of the present invention was determined in accordance with the following
exp... |
US Patent US9200001 (2015)
BindingDB Entry DOI: 10.7270/Q2BP01M3 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor delta
(Homo sapiens (Human)) | BDBM50214974
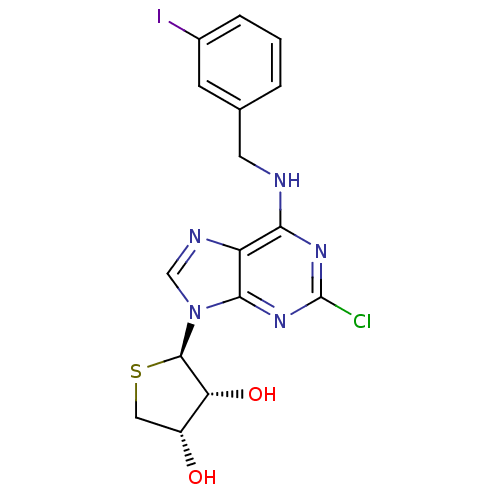
((2R,3R,4S)-2-(2-chloro-6-(3-iodobenzylamino)-9H-pu...)Show SMILES O[C@@H]1CS[C@H]([C@@H]1O)n1cnc2c(NCc3cccc(I)c3)nc(Cl)nc12 |r| Show InChI InChI=1S/C16H15ClIN5O2S/c17-16-21-13(19-5-8-2-1-3-9(18)4-8)11-14(22-16)23(7-20-11)15-12(25)10(24)6-26-15/h1-4,7,10,12,15,24-25H,5-6H2,(H,19,21,22)/t10-,12-,15-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00483 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University
Curated by ChEMBL
| Assay Description
Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARdelta LBD by TR-FRET assay |
J Med Chem 60: 7459-7475 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00805
BindingDB Entry DOI: 10.7270/Q2XK8HQH |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor delta
(Homo sapiens (Human)) | BDBM50214974

((2R,3R,4S)-2-(2-chloro-6-(3-iodobenzylamino)-9H-pu...)Show SMILES O[C@@H]1CS[C@H]([C@@H]1O)n1cnc2c(NCc3cccc(I)c3)nc(Cl)nc12 |r| Show InChI InChI=1S/C16H15ClIN5O2S/c17-16-21-13(19-5-8-2-1-3-9(18)4-8)11-14(22-16)23(7-20-11)15-12(25)10(24)6-26-15/h1-4,7,10,12,15,24-25H,5-6H2,(H,19,21,22)/t10-,12-,15-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00483 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University
Curated by ChEMBL
| Assay Description
Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARdelta LBD by TR-FRET assay |
J Med Chem 60: 7459-7475 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00805
BindingDB Entry DOI: 10.7270/Q2XK8HQH |
More data for this
Ligand-Target Pair | |
Isoform PDE10A2 of cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A (PDE10A2)
(Homo sapiens (Human)) | BDBM194223
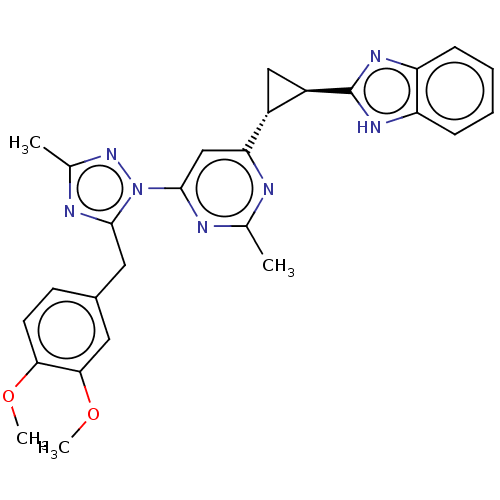
(US9200001, 26)Show SMILES COc1ccc(Cc2nc(C)nn2-c2cc(nc(C)n2)[C@@H]2C[C@H]2c2nc3ccccc3[nH]2)cc1OC |r| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp.
US Patent
| Assay Description
In a typical experiment the PDE10 inhibitory activity of the compounds
of the present invention was determined in accordance with the following
exp... |
US Patent US9200001 (2015)
BindingDB Entry DOI: 10.7270/Q2BP01M3 |
More data for this
Ligand-Target Pair | |
Isoform PDE10A2 of cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A (PDE10A2)
(Homo sapiens (Human)) | BDBM194122

(US9200001, 38)Show SMILES COc1ccc(Cc2nc(C)nn2-c2cc(nc(C)n2)C2CC2c2nc3ccccc3[nH]2)cc1OC | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp.
US Patent
| Assay Description
In a typical experiment the PDE10 inhibitory activity of the compounds
of the present invention was determined in accordance with the following
exp... |
US Patent US9200001 (2015)
BindingDB Entry DOI: 10.7270/Q2BP01M3 |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50156455
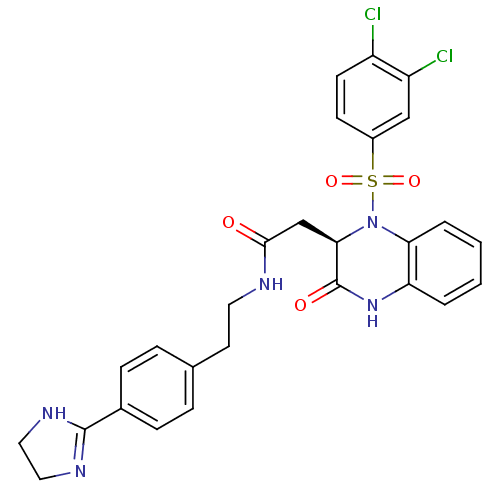
((R)-N-(4-(4,5-dihydro-1H-imidazol-2-yl)phenethyl)-...)Show SMILES Clc1ccc(cc1Cl)S(=O)(=O)N1[C@H](CC(=O)NCCc2ccc(cc2)C2=NCCN2)C(=O)Nc2ccccc12 |r,t:27| Show InChI InChI=1S/C27H25Cl2N5O4S/c28-20-10-9-19(15-21(20)29)39(37,38)34-23-4-2-1-3-22(23)33-27(36)24(34)16-25(35)30-12-11-17-5-7-18(8-6-17)26-31-13-14-32-26/h1-10,15,24H,11-14,16H2,(H,30,35)(H,31,32)(H,33,36)/t24-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.00780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human BK1 receptor E273 mutant |
Bioorg Med Chem Lett 16: 2791-5 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.112
BindingDB Entry DOI: 10.7270/Q28915GX |
More data for this
Ligand-Target Pair | |
Isoform PDE10A2 of cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A (PDE10A2)
(Homo sapiens (Human)) | BDBM194099
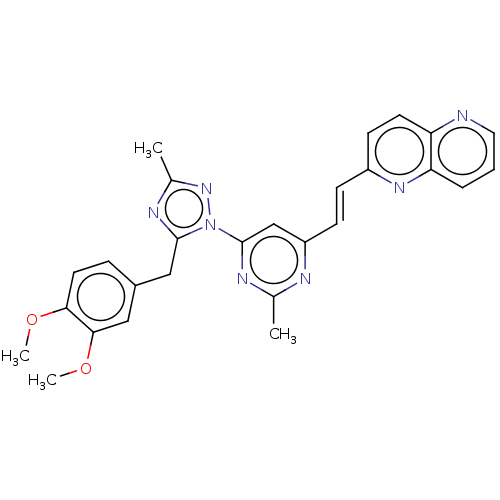
(US9200001, 13)Show SMILES COc1ccc(Cc2nc(C)nn2-c2cc(\C=C\c3ccc4ncccc4n3)nc(C)n2)cc1OC Show InChI InChI=1S/C27H25N7O2/c1-17-29-21(9-8-20-10-11-22-23(32-20)6-5-13-28-22)16-27(30-17)34-26(31-18(2)33-34)15-19-7-12-24(35-3)25(14-19)36-4/h5-14,16H,15H2,1-4H3/b9-8+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp.
US Patent
| Assay Description
In a typical experiment the PDE10 inhibitory activity of the compounds
of the present invention was determined in accordance with the following
exp... |
US Patent US9200001 (2015)
BindingDB Entry DOI: 10.7270/Q2BP01M3 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor delta
(Homo sapiens (Human)) | BDBM50214981
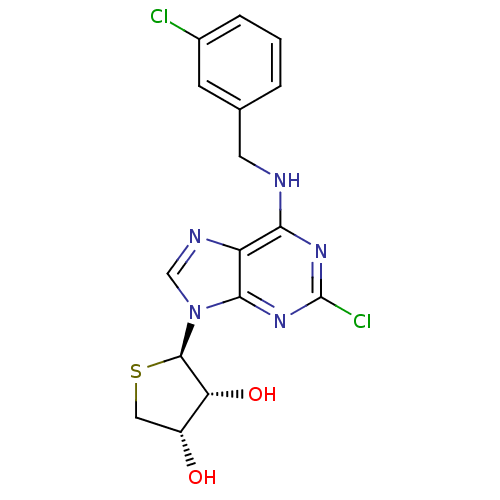
((2R,3R,4S)-2-(2-chloro-6-(3-chlorobenzylamino)-9H-...)Show SMILES O[C@@H]1CS[C@H]([C@@H]1O)n1cnc2c(NCc3cccc(Cl)c3)nc(Cl)nc12 |r| Show InChI InChI=1S/C16H15Cl2N5O2S/c17-9-3-1-2-8(4-9)5-19-13-11-14(22-16(18)21-13)23(7-20-11)15-12(25)10(24)6-26-15/h1-4,7,10,12,15,24-25H,5-6H2,(H,19,21,22)/t10-,12-,15-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0102 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University
Curated by ChEMBL
| Assay Description
Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARdelta LBD by TR-FRET assay |
J Med Chem 60: 7459-7475 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00805
BindingDB Entry DOI: 10.7270/Q2XK8HQH |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor delta
(Homo sapiens (Human)) | BDBM50214981

((2R,3R,4S)-2-(2-chloro-6-(3-chlorobenzylamino)-9H-...)Show SMILES O[C@@H]1CS[C@H]([C@@H]1O)n1cnc2c(NCc3cccc(Cl)c3)nc(Cl)nc12 |r| Show InChI InChI=1S/C16H15Cl2N5O2S/c17-9-3-1-2-8(4-9)5-19-13-11-14(22-16(18)21-13)23(7-20-11)15-12(25)10(24)6-26-15/h1-4,7,10,12,15,24-25H,5-6H2,(H,19,21,22)/t10-,12-,15-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0102 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University
Curated by ChEMBL
| Assay Description
Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARdelta LBD by TR-FRET assay |
J Med Chem 60: 7459-7475 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00805
BindingDB Entry DOI: 10.7270/Q2XK8HQH |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450717

(CHEMBL317087)Show InChI InChI=1S/C9H10BrClN2O/c10-8-3-7(4-13-9(8)11)14-5-6-1-2-12-6/h3-4,6,12H,1-2,5H2/t6-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220995

(CHEMBL77788)Show InChI InChI=1S/C15H20N2O3/c1-11(18)17-16-10-12-7-8-14(19-2)15(9-12)20-13-5-3-4-6-13/h7-10,13H,3-6H2,1-2H3,(H,17,18)/b16-10+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450726
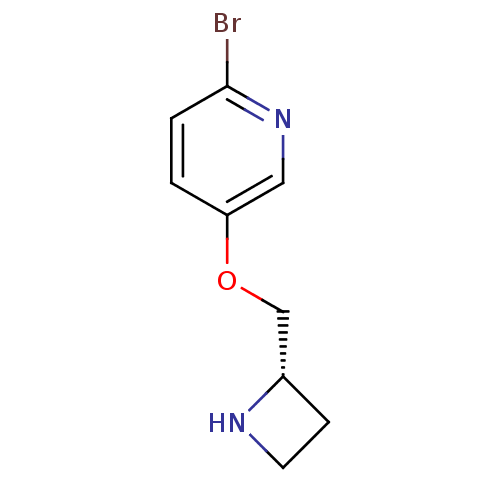
(CHEMBL407258)Show InChI InChI=1S/C9H11BrN2O/c10-9-2-1-8(5-12-9)13-6-7-3-4-11-7/h1-2,5,7,11H,3-4,6H2/t7-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450734
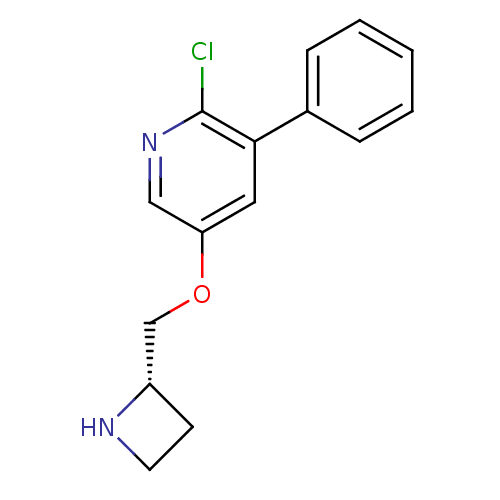
(CHEMBL318869)Show InChI InChI=1S/C15H15ClN2O/c16-15-14(11-4-2-1-3-5-11)8-13(9-18-15)19-10-12-6-7-17-12/h1-5,8-9,12,17H,6-7,10H2/t12-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Rattus norvegicus (rat)) | BDBM50366620
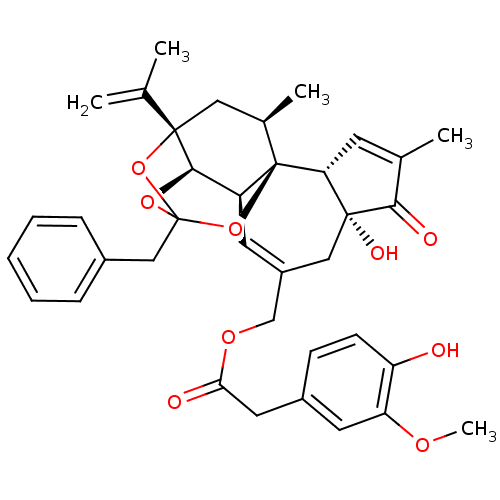
(RESINIFERATOXIN)Show SMILES COc1cc(CC(=O)OCC2=C[C@H]3[C@H]4OC5(Cc6ccccc6)O[C@@]4(C[C@@H](C)[C@]3(O5)[C@@H]3C=C(C)C(=O)[C@@]3(O)C2)C(C)=C)ccc1O |r,t:10,35,TLB:23:15:12:24.25.26,THB:16:15:12:24.25.26| Show InChI InChI=1S/C37H40O9/c1-21(2)35-17-23(4)37-27(33(35)44-36(45-35,46-37)19-24-9-7-6-8-10-24)14-26(18-34(41)30(37)13-22(3)32(34)40)20-43-31(39)16-25-11-12-28(38)29(15-25)42-5/h6-15,23,27,30,33,38,41H,1,16-20H2,2-5H3/t23-,27+,30-,33-,34-,35+,36?,37-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards vanilloid receptor by [3H]RTX displacement. |
Bioorg Med Chem Lett 9: 2909-14 (1999)
BindingDB Entry DOI: 10.7270/Q2HT2PTB |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450710
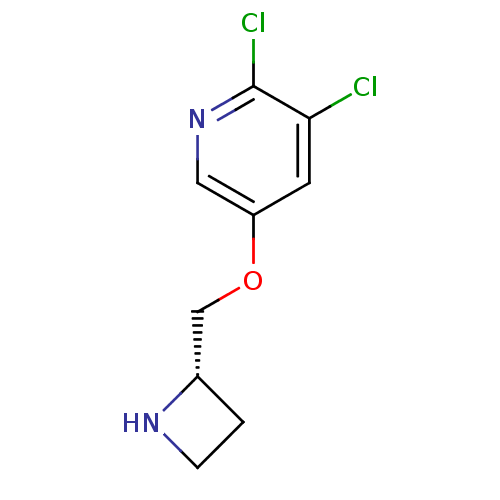
(CHEMBL97555)Show InChI InChI=1S/C9H10Cl2N2O/c10-8-3-7(4-13-9(8)11)14-5-6-1-2-12-6/h3-4,6,12H,1-2,5H2/t6-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 1
(Rattus norvegicus (rat)) | BDBM50366620

(RESINIFERATOXIN)Show SMILES COc1cc(CC(=O)OCC2=C[C@H]3[C@H]4OC5(Cc6ccccc6)O[C@@]4(C[C@@H](C)[C@]3(O5)[C@@H]3C=C(C)C(=O)[C@@]3(O)C2)C(C)=C)ccc1O |r,t:10,35,TLB:23:15:12:24.25.26,THB:16:15:12:24.25.26| Show InChI InChI=1S/C37H40O9/c1-21(2)35-17-23(4)37-27(33(35)44-36(45-35,46-37)19-24-9-7-6-8-10-24)14-26(18-34(41)30(37)13-22(3)32(34)40)20-43-31(39)16-25-11-12-28(38)29(15-25)42-5/h6-15,23,27,30,33,38,41H,1,16-20H2,2-5H3/t23-,27+,30-,33-,34-,35+,36?,37-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University
Curated by ChEMBL
| Assay Description
In vitro binding affinity towards vanilloid receptor by [3H]RTX displacement. |
Bioorg Med Chem Lett 9: 2909-14 (1999)
BindingDB Entry DOI: 10.7270/Q2HT2PTB |
More data for this
Ligand-Target Pair | |
Isoform PDE10A2 of cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A (PDE10A2)
(Homo sapiens (Human)) | BDBM194089

(US9200001, 2)Show SMILES COc1ccc(Cc2nc(C)nn2-c2cc(nc(C)n2)C#Cc2ccc3ccccc3n2)cc1OC Show InChI InChI=1S/C28H24N6O2/c1-18-29-23(13-12-22-11-10-21-7-5-6-8-24(21)32-22)17-28(30-18)34-27(31-19(2)33-34)16-20-9-14-25(35-3)26(15-20)36-4/h5-11,14-15,17H,16H2,1-4H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp.
US Patent
| Assay Description
In a typical experiment the PDE10 inhibitory activity of the compounds
of the present invention was determined in accordance with the following
exp... |
US Patent US9200001 (2015)
BindingDB Entry DOI: 10.7270/Q2BP01M3 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450720

(CHEMBL94683)Show InChI InChI=1S/C10H11N3O/c11-4-8-3-10(6-12-5-8)14-7-9-1-2-13-9/h3,5-6,9,13H,1-2,7H2/t9-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Isoform PDE10A2 of cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A (PDE10A2)
(Homo sapiens (Human)) | BDBM194102
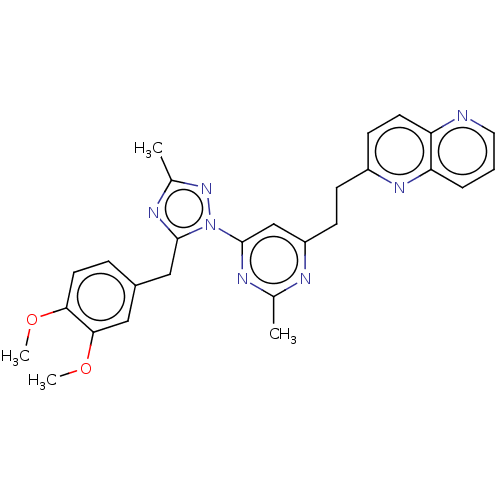
(US9200001, 16)Show SMILES COc1ccc(Cc2nc(C)nn2-c2cc(CCc3ccc4ncccc4n3)nc(C)n2)cc1OC Show InChI InChI=1S/C27H27N7O2/c1-17-29-21(9-8-20-10-11-22-23(32-20)6-5-13-28-22)16-27(30-17)34-26(31-18(2)33-34)15-19-7-12-24(35-3)25(14-19)36-4/h5-7,10-14,16H,8-9,15H2,1-4H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp.
US Patent
| Assay Description
In a typical experiment the PDE10 inhibitory activity of the compounds
of the present invention was determined in accordance with the following
exp... |
US Patent US9200001 (2015)
BindingDB Entry DOI: 10.7270/Q2BP01M3 |
More data for this
Ligand-Target Pair | |
Isoform PDE10A2 of cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A (PDE10A2)
(Homo sapiens (Human)) | BDBM194111
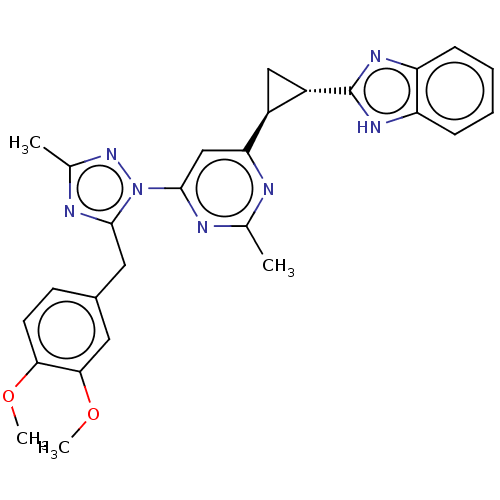
(US9200001, 25)Show SMILES COc1ccc(Cc2nc(C)nn2-c2cc(nc(C)n2)[C@H]2C[C@@H]2c2nc3ccccc3[nH]2)cc1OC |r| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp.
US Patent
| Assay Description
In a typical experiment the PDE10 inhibitory activity of the compounds
of the present invention was determined in accordance with the following
exp... |
US Patent US9200001 (2015)
BindingDB Entry DOI: 10.7270/Q2BP01M3 |
More data for this
Ligand-Target Pair | |
Isoform PDE10A2 of cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A (PDE10A2)
(Homo sapiens (Human)) | BDBM194095

(US9200001, 8)Show SMILES COc1ccc(Cc2nc(C)nn2-c2cc(CCc3ccc4ccccc4n3)nc(C)n2)nc1OC Show InChI InChI=1S/C27H27N7O2/c1-17-28-21(12-11-20-10-9-19-7-5-6-8-23(19)31-20)15-25(29-17)34-26(30-18(2)33-34)16-22-13-14-24(35-3)27(32-22)36-4/h5-10,13-15H,11-12,16H2,1-4H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp.
US Patent
| Assay Description
In a typical experiment the PDE10 inhibitory activity of the compounds
of the present invention was determined in accordance with the following
exp... |
US Patent US9200001 (2015)
BindingDB Entry DOI: 10.7270/Q2BP01M3 |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50156455

((R)-N-(4-(4,5-dihydro-1H-imidazol-2-yl)phenethyl)-...)Show SMILES Clc1ccc(cc1Cl)S(=O)(=O)N1[C@H](CC(=O)NCCc2ccc(cc2)C2=NCCN2)C(=O)Nc2ccccc12 |r,t:27| Show InChI InChI=1S/C27H25Cl2N5O4S/c28-20-10-9-19(15-21(20)29)39(37,38)34-23-4-2-1-3-22(23)33-27(36)24(34)16-25(35)30-12-11-17-5-7-18(8-6-17)26-31-13-14-32-26/h1-10,15,24H,11-14,16H2,(H,30,35)(H,31,32)(H,33,36)/t24-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human BK1 receptor |
Bioorg Med Chem Lett 16: 2791-5 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.112
BindingDB Entry DOI: 10.7270/Q28915GX |
More data for this
Ligand-Target Pair | |
Calcitonin gene-related peptide type 1 receptor
(Homo sapiens (Human)) | BDBM50301971
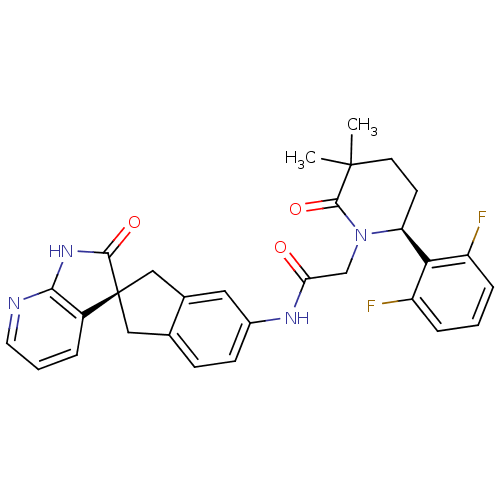
(2-((S)-6-(2,6-difluorophenyl)-3,3-dimethyl-2-oxopi...)Show SMILES CC1(C)CC[C@H](N(CC(=O)Nc2ccc3C[C@@]4(Cc3c2)C(=O)Nc2ncccc42)C1=O)c1c(F)cccc1F |r| Show InChI InChI=1S/C30H28F2N4O3/c1-29(2)11-10-23(25-21(31)6-3-7-22(25)32)36(28(29)39)16-24(37)34-19-9-8-17-14-30(15-18(17)13-19)20-5-4-12-33-26(20)35-27(30)38/h3-9,12-13,23H,10-11,14-16H2,1-2H3,(H,34,37)(H,33,35,38)/t23-,30-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck& Co.
Curated by ChEMBL
| Assay Description
Displacement of [125I]hCGRP from human cloned CGRP receptor expressed in HEK293 cells |
Bioorg Med Chem Lett 19: 5787-90 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.134
BindingDB Entry DOI: 10.7270/Q2PG1RTW |
More data for this
Ligand-Target Pair | |
Calcitonin gene-related peptide type 1 receptor
(Homo sapiens (Human)) | BDBM50329174

(CHEMBL1269644 | N-(((R)-2,3-dihydro-1H-inden-1-yl)...)Show SMILES CC(C)(C)C(=O)N(C[C@@H]1CCc2ccccc12)Cc1cnc2cc3C[C@]4(Cc3cc2c1)C(=O)Nc1ncccc41 |r| Show InChI InChI=1S/C34H34N4O2/c1-33(2,3)32(40)38(20-23-11-10-22-7-4-5-8-27(22)23)19-21-13-24-14-25-16-34(17-26(25)15-29(24)36-18-21)28-9-6-12-35-30(28)37-31(34)39/h4-9,12-15,18,23H,10-11,16-17,19-20H2,1-3H3,(H,35,37,39)/t23-,34-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck& Co.
Curated by ChEMBL
| Assay Description
Displacement of [125I]hCGRP from human cloned CGRP receptor expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6827-30 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.105
BindingDB Entry DOI: 10.7270/Q2Z89CNJ |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50156455

((R)-N-(4-(4,5-dihydro-1H-imidazol-2-yl)phenethyl)-...)Show SMILES Clc1ccc(cc1Cl)S(=O)(=O)N1[C@H](CC(=O)NCCc2ccc(cc2)C2=NCCN2)C(=O)Nc2ccccc12 |r,t:27| Show InChI InChI=1S/C27H25Cl2N5O4S/c28-20-10-9-19(15-21(20)29)39(37,38)34-23-4-2-1-3-22(23)33-27(36)24(34)16-25(35)30-12-11-17-5-7-18(8-6-17)26-31-13-14-32-26/h1-10,15,24H,11-14,16H2,(H,30,35)(H,31,32)(H,33,36)/t24-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human BK1 receptor N298 mutant |
Bioorg Med Chem Lett 16: 2791-5 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.112
BindingDB Entry DOI: 10.7270/Q28915GX |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450712

(CHEMBL96604)Show InChI InChI=1S/C11H16N2O2/c1-2-14-10-5-11(7-12-6-10)15-8-9-3-4-13-9/h5-7,9,13H,2-4,8H2,1H3/t9-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450713

(CHEMBL96837)Show InChI InChI=1S/C12H18N2O/c1-2-3-10-6-12(8-13-7-10)15-9-11-4-5-14-11/h6-8,11,14H,2-5,9H2,1H3/t11-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50062641
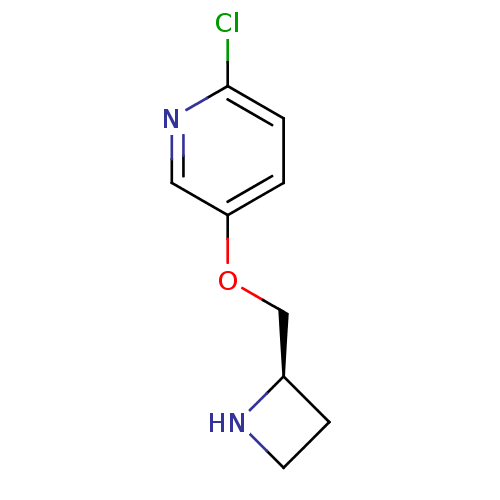
(5-((R)-1-Azetidin-2-ylmethoxy)-2-chloro-pyridine |...)Show InChI InChI=1S/C9H11ClN2O/c10-9-2-1-8(5-12-9)13-6-7-3-4-11-7/h1-2,5,7,11H,3-4,6H2/t7-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM50049750
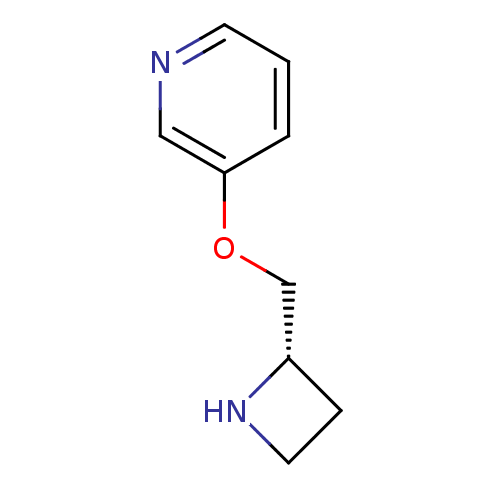
((S)-3-(azetidin-2-ylmethoxy)pyridine | 3-((S)-1-Az...)Show InChI InChI=1S/C9H12N2O/c1-2-9(6-10-4-1)12-7-8-3-5-11-8/h1-2,4,6,8,11H,3,5,7H2/t8-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratory
Curated by ChEMBL
| Assay Description
Compound was evaluated for its ability to displace [3H]-cytisine binding from high-affinity Nicotinic acetylcholine receptor in rat brain (principall... |
J Med Chem 41: 407-12 (1998)
Article DOI: 10.1021/jm9706224
BindingDB Entry DOI: 10.7270/Q2CJ8F56 |
More data for this
Ligand-Target Pair | |
Calcitonin gene-related peptide type 1 receptor/Receptor activity-modifying protein 1
(Homo sapiens (Human)) | BDBM50444982
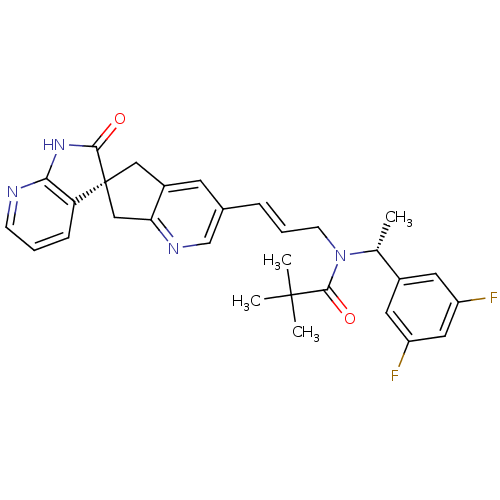
(CHEMBL3099918)Show SMILES C[C@@H](N(C\C=C\c1cnc2C[C@]3(Cc2c1)C(=O)Nc1ncccc31)C(=O)C(C)(C)C)c1cc(F)cc(F)c1 |r| Show InChI InChI=1S/C30H30F2N4O2/c1-18(20-12-22(31)14-23(32)13-20)36(28(38)29(2,3)4)10-6-7-19-11-21-15-30(16-25(21)34-17-19)24-8-5-9-33-26(24)35-27(30)37/h5-9,11-14,17-18H,10,15-16H2,1-4H3,(H,33,35,37)/b7-6+/t18-,30+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck& Co.
Curated by ChEMBL
| Assay Description
Displacement of [125I]-hCGRP from human CALCRL/RAMP1 expressed in HEK293 cell membranes |
Bioorg Med Chem Lett 24: 258-61 (2013)
Article DOI: 10.1016/j.bmcl.2013.11.027
BindingDB Entry DOI: 10.7270/Q2DB8393 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50062639
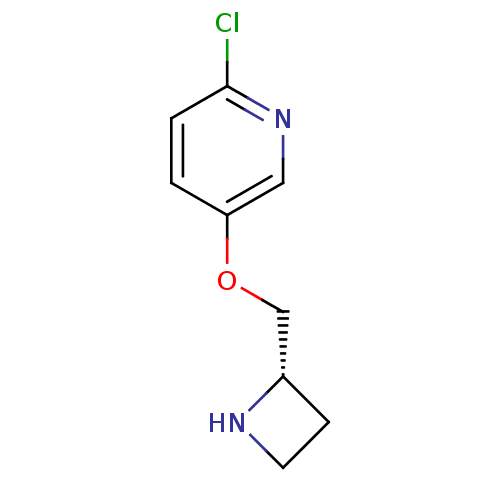
(5-((S)-1-Azetidin-2-ylmethoxy)-2-chloro-pyridine |...)Show InChI InChI=1S/C9H11ClN2O/c10-9-2-1-8(5-12-9)13-6-7-3-4-11-7/h1-2,5,7,11H,3-4,6H2/t7-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Calcitonin gene-related peptide type 1 receptor
(Homo sapiens (Human)) | BDBM103495
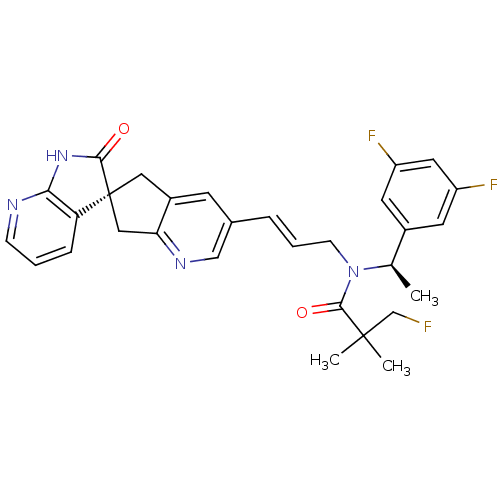
(US8552023, 7)Show SMILES C[C@@H](N(C\C=C\c1cnc2C[C@]3(Cc2c1)C(=O)Nc1ncccc31)C(=O)C(C)(C)CF)c1cc(F)cc(F)c1 |r| Show InChI InChI=1S/C30H29F3N4O2/c1-18(20-11-22(32)13-23(33)12-20)37(28(39)29(2,3)17-31)9-5-6-19-10-21-14-30(15-25(21)35-16-19)24-7-4-8-34-26(24)36-27(30)38/h4-8,10-13,16,18H,9,14-15,17H2,1-3H3,(H,34,36,38)/b6-5+/t18-,30+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0410 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Merck Sharp & Dohme Corp.
US Patent
| Assay Description
Inhibition of the binding of 125I-CGRP to receptors and functional antagonism of CGRP receptors were determined using native receptor binding assay. |
US Patent US8552023 (2013)
BindingDB Entry DOI: 10.7270/Q2VX0F4V |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50221005
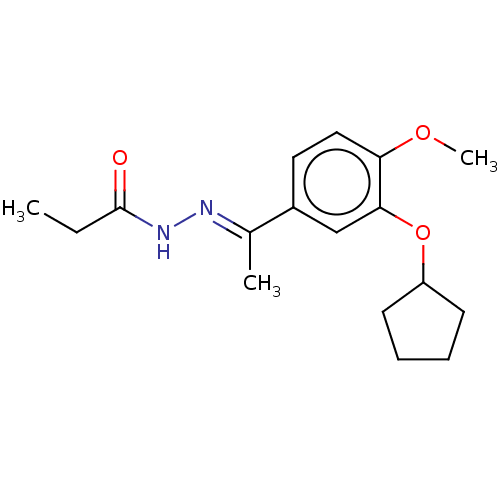
(CHEMBL75684)Show InChI InChI=1S/C17H24N2O3/c1-4-17(20)19-18-12(2)13-9-10-15(21-3)16(11-13)22-14-7-5-6-8-14/h9-11,14H,4-8H2,1-3H3,(H,19,20)/b18-12+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450723
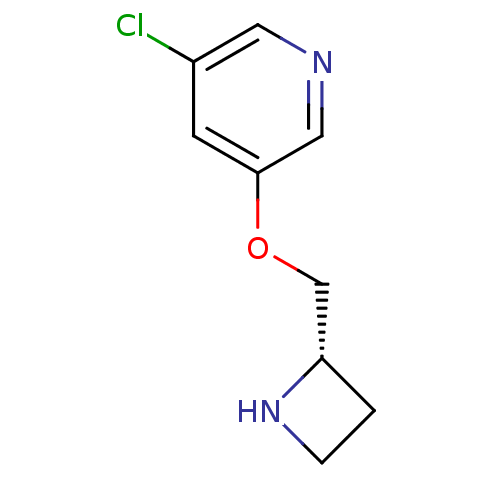
(CHEMBL95113)Show InChI InChI=1S/C9H11ClN2O/c10-7-3-9(5-11-4-7)13-6-8-1-2-12-8/h3-5,8,12H,1-2,6H2/t8-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50184183
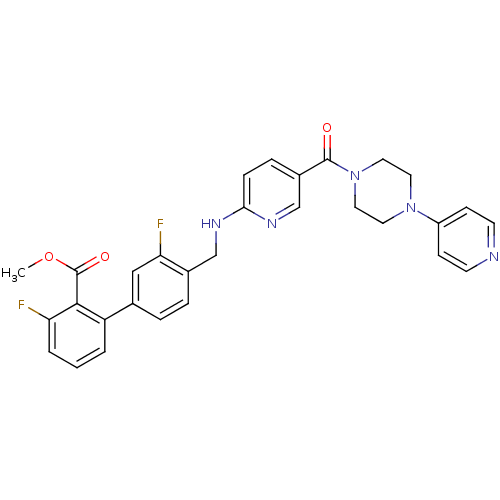
(3,3'-difluoro-4'-{[5-(4-pyridin-4-yl-piperazine-1-...)Show SMILES COC(=O)c1c(F)cccc1-c1ccc(CNc2ccc(cn2)C(=O)N2CCN(CC2)c2ccncc2)c(F)c1 Show InChI InChI=1S/C30H27F2N5O3/c1-40-30(39)28-24(3-2-4-25(28)31)20-5-6-21(26(32)17-20)18-34-27-8-7-22(19-35-27)29(38)37-15-13-36(14-16-37)23-9-11-33-12-10-23/h2-12,17,19H,13-16,18H2,1H3,(H,34,35) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human BK1 receptor |
Bioorg Med Chem Lett 16: 2791-5 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.112
BindingDB Entry DOI: 10.7270/Q28915GX |
More data for this
Ligand-Target Pair | |
cAMP-specific 3',5'-cyclic phosphodiesterase 4C
(Rattus norvegicus) | BDBM50220998
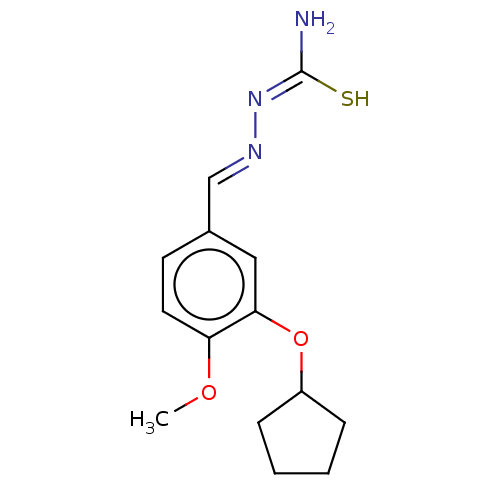
(CHEMBL76382)Show InChI InChI=1S/C14H19N3O2S/c1-18-12-7-6-10(9-16-17-14(15)20)8-13(12)19-11-4-2-3-5-11/h6-9,11H,2-5H2,1H3,(H3,15,17,20)/b16-9+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
LEAD GENE CO., LTD
Curated by ChEMBL
| Assay Description
Binding affinity towards rolipram binding site on phosphodiesterase 4 using [3H]rolipram as radioligand in crude rat brain homogenate |
Bioorg Med Chem Lett 13: 2355-8 (2003)
BindingDB Entry DOI: 10.7270/Q2CZ39CG |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50072110
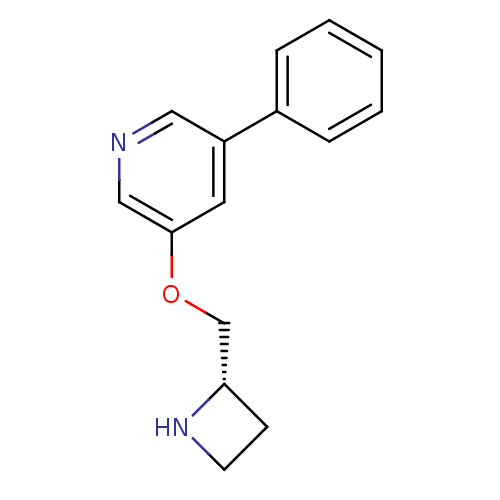
(3-((S)-1-Azetidin-2-ylmethoxy)-5-phenyl-pyridine |...)Show InChI InChI=1S/C15H16N2O/c1-2-4-12(5-3-1)13-8-15(10-16-9-13)18-11-14-6-7-17-14/h1-5,8-10,14,17H,6-7,11H2/t14-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50450728
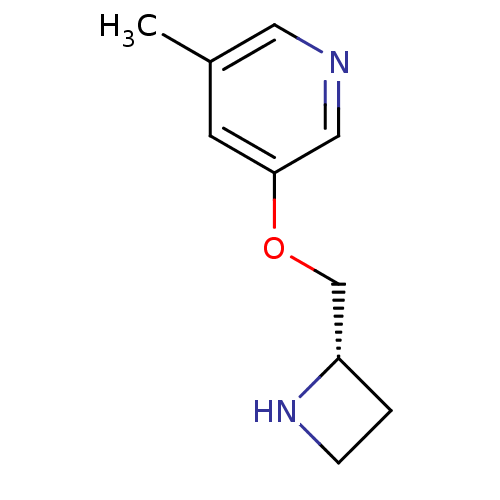
(CHEMBL96431)Show InChI InChI=1S/C10H14N2O/c1-8-4-10(6-11-5-8)13-7-9-2-3-12-9/h4-6,9,12H,2-3,7H2,1H3/t9-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor delta
(Homo sapiens (Human)) | BDBM28661
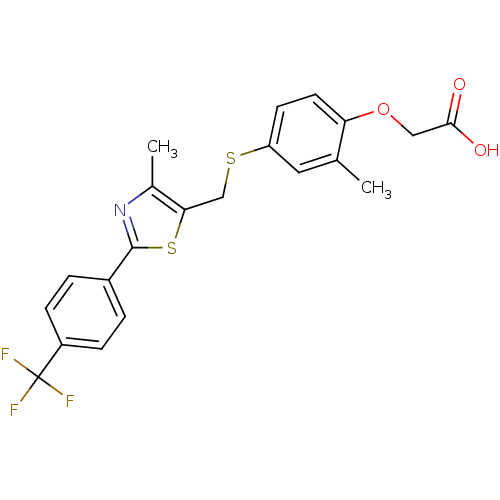
(2-{2-methyl-4-[({4-methyl-2-[4-(trifluoromethyl)ph...)Show SMILES Cc1nc(sc1CSc1ccc(OCC(O)=O)c(C)c1)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C21H18F3NO3S2/c1-12-9-16(7-8-17(12)28-10-19(26)27)29-11-18-13(2)25-20(30-18)14-3-5-15(6-4-14)21(22,23)24/h3-9H,10-11H2,1-2H3,(H,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University
Curated by ChEMBL
| Assay Description
Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARdelta LBD by TR-FRET assay |
J Med Chem 60: 7459-7475 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00805
BindingDB Entry DOI: 10.7270/Q2XK8HQH |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50066788
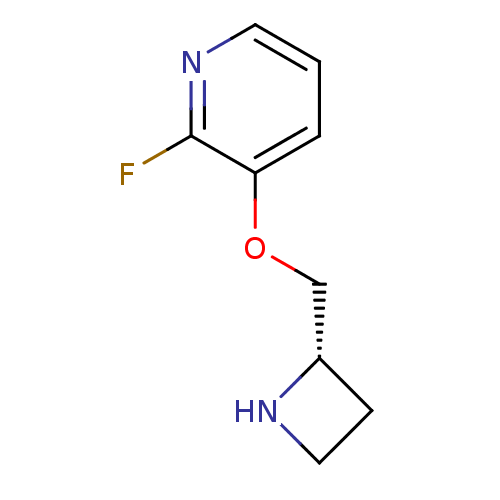
((S)-3-(azetidin-2-ylmethoxy)-2-fluoropyridine | 3-...)Show InChI InChI=1S/C9H11FN2O/c10-9-8(2-1-4-12-9)13-6-7-3-5-11-7/h1-2,4,7,11H,3,5-6H2/t7-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards Nicotinic acetylcholine receptor alpha4-beta2 in rat brain using [3H]-cytisine as radioligand |
Bioorg Med Chem Lett 8: 2797-802 (1999)
BindingDB Entry DOI: 10.7270/Q23J3FGV |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor delta
(Homo sapiens (Human)) | BDBM28661

(2-{2-methyl-4-[({4-methyl-2-[4-(trifluoromethyl)ph...)Show SMILES Cc1nc(sc1CSc1ccc(OCC(O)=O)c(C)c1)-c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C21H18F3NO3S2/c1-12-9-16(7-8-17(12)28-10-19(26)27)29-11-18-13(2)25-20(30-18)14-3-5-15(6-4-14)21(22,23)24/h3-9H,10-11H2,1-2H3,(H,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University
Curated by ChEMBL
| Assay Description
Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARdelta LBD by TR-FRET assay |
J Med Chem 60: 7459-7475 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00805
BindingDB Entry DOI: 10.7270/Q2XK8HQH |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50156455

((R)-N-(4-(4,5-dihydro-1H-imidazol-2-yl)phenethyl)-...)Show SMILES Clc1ccc(cc1Cl)S(=O)(=O)N1[C@H](CC(=O)NCCc2ccc(cc2)C2=NCCN2)C(=O)Nc2ccccc12 |r,t:27| Show InChI InChI=1S/C27H25Cl2N5O4S/c28-20-10-9-19(15-21(20)29)39(37,38)34-23-4-2-1-3-22(23)33-27(36)24(34)16-25(35)30-12-11-17-5-7-18(8-6-17)26-31-13-14-32-26/h1-10,15,24H,11-14,16H2,(H,30,35)(H,31,32)(H,33,36)/t24-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity to human BK1 receptor D291 mutant |
Bioorg Med Chem Lett 16: 2791-5 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.112
BindingDB Entry DOI: 10.7270/Q28915GX |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50085048
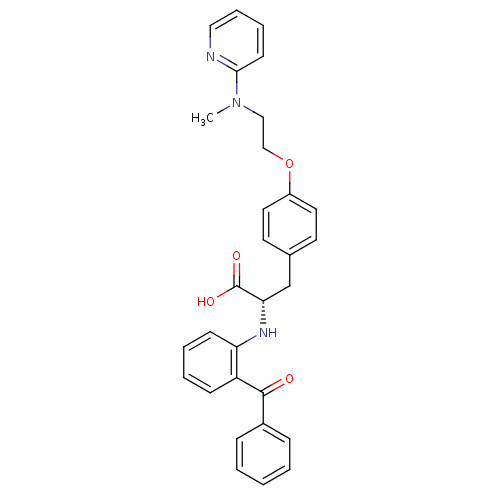
((S)-2-(2-Benzoyl-phenylamino)-3-{4-[2-(methyl-pyri...)Show SMILES CN(CCOc1ccc(C[C@H](Nc2ccccc2C(=O)c2ccccc2)C(O)=O)cc1)c1ccccn1 Show InChI InChI=1S/C30H29N3O4/c1-33(28-13-7-8-18-31-28)19-20-37-24-16-14-22(15-17-24)21-27(30(35)36)32-26-12-6-5-11-25(26)29(34)23-9-3-2-4-10-23/h2-18,27,32H,19-21H2,1H3,(H,35,36)/t27-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0497 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University
Curated by ChEMBL
| Assay Description
Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARgamma LBD by TR-FRET assay |
J Med Chem 60: 7459-7475 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00805
BindingDB Entry DOI: 10.7270/Q2XK8HQH |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50085048

((S)-2-(2-Benzoyl-phenylamino)-3-{4-[2-(methyl-pyri...)Show SMILES CN(CCOc1ccc(C[C@H](Nc2ccccc2C(=O)c2ccccc2)C(O)=O)cc1)c1ccccn1 Show InChI InChI=1S/C30H29N3O4/c1-33(28-13-7-8-18-31-28)19-20-37-24-16-14-22(15-17-24)21-27(30(35)36)32-26-12-6-5-11-25(26)29(34)23-9-3-2-4-10-23/h2-18,27,32H,19-21H2,1H3,(H,35,36)/t27-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0497 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sahmyook University
Curated by ChEMBL
| Assay Description
Displacement of Fluormone-Pan-PPAR Green from human GST-tagged PPARgamma LBD by TR-FRET assay |
J Med Chem 60: 7459-7475 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00805
BindingDB Entry DOI: 10.7270/Q2XK8HQH |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Calcitonin gene-related peptide type 1 receptor/Receptor activity-modifying protein 1
(Homo sapiens (Human)) | BDBM103499

(US8552023, 11)Show SMILES C[C@@H](N(C\C=C\c1cnc2C[C@]3(Cc2c1)C(=O)Nc1ncccc31)C(=O)C1(CC1)C(F)(F)F)c1cc(F)cc(F)c1 |r| Show InChI InChI=1S/C30H25F5N4O2/c1-17(19-11-21(31)13-22(32)12-19)39(27(41)29(6-7-29)30(33,34)35)9-3-4-18-10-20-14-28(15-24(20)37-16-18)23-5-2-8-36-25(23)38-26(28)40/h2-5,8,10-13,16-17H,6-7,9,14-15H2,1H3,(H,36,38,40)/b4-3+/t17-,28+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck& Co.
Curated by ChEMBL
| Assay Description
Displacement of [125I]-hCGRP from human CALCRL/RAMP1 expressed in HEK293 cell membranes |
Bioorg Med Chem Lett 24: 258-61 (2013)
Article DOI: 10.1016/j.bmcl.2013.11.027
BindingDB Entry DOI: 10.7270/Q2DB8393 |
More data for this
Ligand-Target Pair | |
Calcitonin gene-related peptide type 1 receptor/Receptor activity-modifying protein 1
(Homo sapiens (Human)) | BDBM50444972
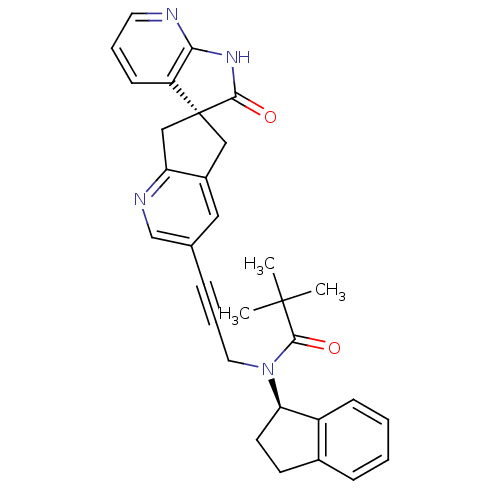
(CHEMBL3099931)Show SMILES CC(C)(C)C(=O)N(CC#Cc1cnc2C[C@]3(Cc2c1)C(=O)Nc1ncccc31)[C@@H]1CCc2ccccc12 |r| Show InChI InChI=1S/C31H30N4O2/c1-30(2,3)29(37)35(26-13-12-21-9-4-5-10-23(21)26)15-7-8-20-16-22-17-31(18-25(22)33-19-20)24-11-6-14-32-27(24)34-28(31)36/h4-6,9-11,14,16,19,26H,12-13,15,17-18H2,1-3H3,(H,32,34,36)/t26-,31+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck& Co.
Curated by ChEMBL
| Assay Description
Displacement of [125I]-hCGRP from human CALCRL/RAMP1 expressed in HEK293 cell membranes |
Bioorg Med Chem Lett 24: 258-61 (2013)
Article DOI: 10.1016/j.bmcl.2013.11.027
BindingDB Entry DOI: 10.7270/Q2DB8393 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data