

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50370067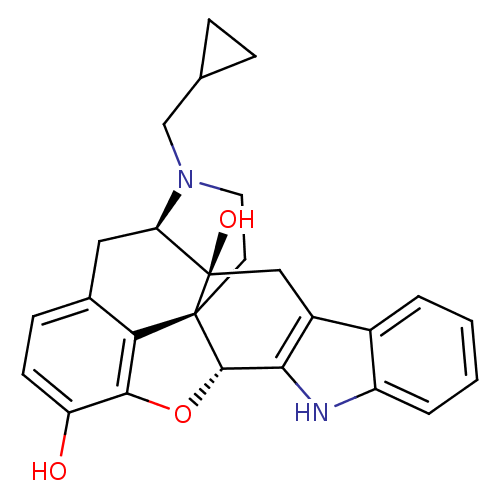 (CHEMBL1237164) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kitasato University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from human delta opioid receptor expressed in CHO cells | Bioorg Med Chem Lett 30: (2020) Article DOI: 10.1016/j.bmcl.2020.127176 BindingDB Entry DOI: 10.7270/Q2W380XV | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50545482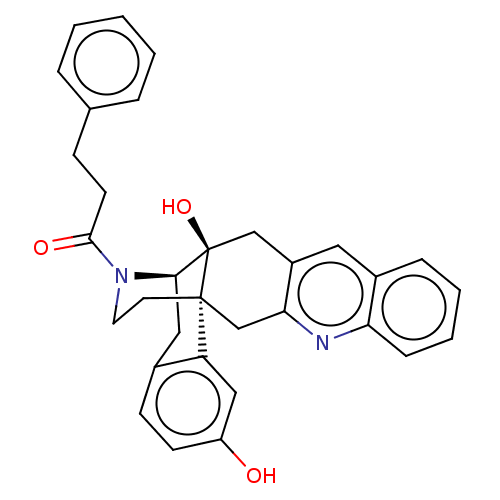 (CHEMBL4649733) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kitasato University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from human delta opioid receptor expressed in CHO cells | Bioorg Med Chem Lett 30: (2020) Article DOI: 10.1016/j.bmcl.2020.127176 BindingDB Entry DOI: 10.7270/Q2W380XV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP1 subtype (Homo sapiens (Human)) | BDBM50276262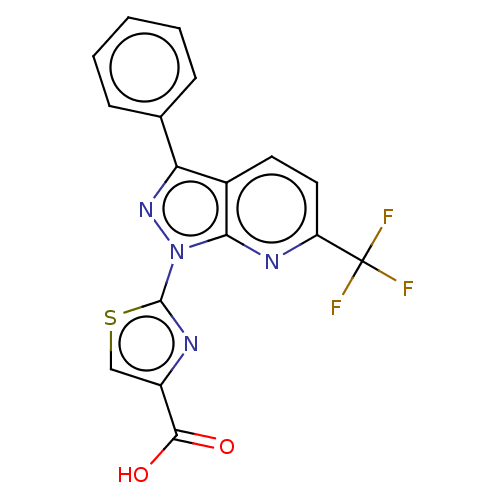 (CHEMBL4129609) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 8.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Asahi Kasei Pharma Corporation Curated by ChEMBL | Assay Description Antagonist activity at EP1 receptor (unknown origin) by reporter gene assay | Bioorg Med Chem Lett 28: 2408-2412 (2018) Article DOI: 10.1016/j.bmcl.2018.06.022 BindingDB Entry DOI: 10.7270/Q2T72KZT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50370067 (CHEMBL1237164) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kitasato University Curated by ChEMBL | Assay Description Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cells | Bioorg Med Chem Lett 30: (2020) Article DOI: 10.1016/j.bmcl.2020.127176 BindingDB Entry DOI: 10.7270/Q2W380XV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50545481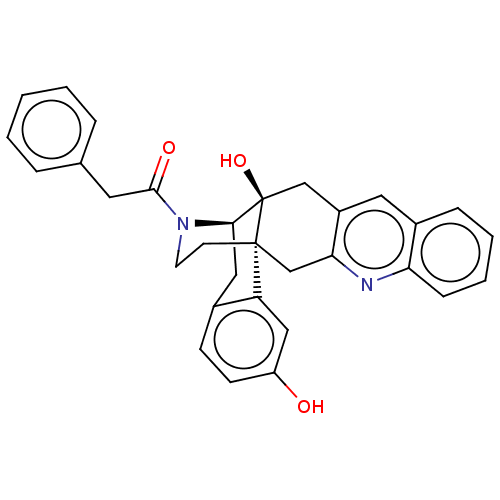 (CHEMBL4632584) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kitasato University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from human delta opioid receptor expressed in CHO cells | Bioorg Med Chem Lett 30: (2020) Article DOI: 10.1016/j.bmcl.2020.127176 BindingDB Entry DOI: 10.7270/Q2W380XV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50370067 (CHEMBL1237164) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | 31 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kitasato University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cells | Bioorg Med Chem Lett 30: (2020) Article DOI: 10.1016/j.bmcl.2020.127176 BindingDB Entry DOI: 10.7270/Q2W380XV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50545479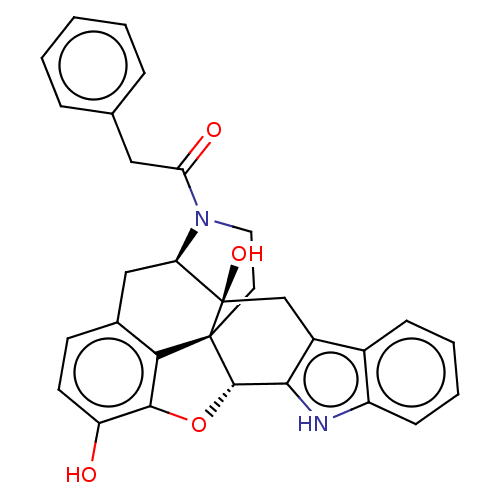 (CHEMBL4638693) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 42 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kitasato University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from human delta opioid receptor expressed in CHO cells | Bioorg Med Chem Lett 30: (2020) Article DOI: 10.1016/j.bmcl.2020.127176 BindingDB Entry DOI: 10.7270/Q2W380XV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tissue alpha-L-fucosidase (Homo sapiens (Human)) | BDBM50065257 ((2R,3R,4R,5R,6S)-2-Hydroxymethyl-6-methyl-piperidi...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-L-fucosidase of bovine epididymis by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tissue alpha-L-fucosidase (Homo sapiens (Human)) | BDBM50065258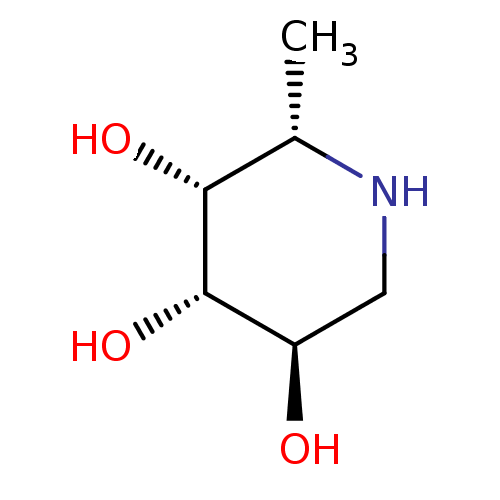 ((2S,3R,4S,5R)-2-Methyl-piperidine-3,4,5-triol | (2...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-L-fucosidase of bovine epididymis by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50545480 (CHEMBL4636799) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 89 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kitasato University Curated by ChEMBL | Assay Description Displacement of [3H]DPDPE from human delta opioid receptor expressed in CHO cells | Bioorg Med Chem Lett 30: (2020) Article DOI: 10.1016/j.bmcl.2020.127176 BindingDB Entry DOI: 10.7270/Q2W380XV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Maltase-glucoamylase (Homo sapiens (Human)) | BDBM50408432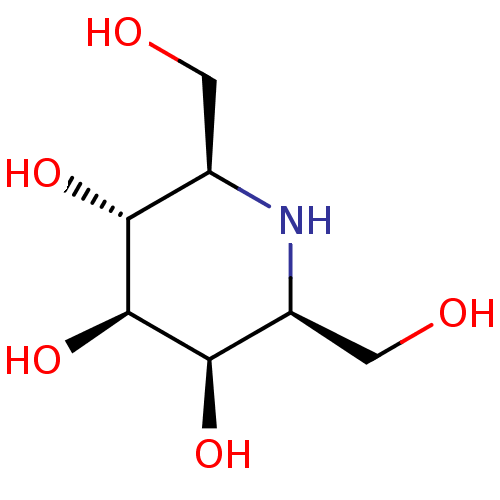 (CHEMBL2115215) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Compound was tested for binding affinity against alpha-glucosidase | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50545482 (CHEMBL4649733) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 1.79E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kitasato University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cells | Bioorg Med Chem Lett 30: (2020) Article DOI: 10.1016/j.bmcl.2020.127176 BindingDB Entry DOI: 10.7270/Q2W380XV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50545480 (CHEMBL4636799) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 2.15E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kitasato University Curated by ChEMBL | Assay Description Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cells | Bioorg Med Chem Lett 30: (2020) Article DOI: 10.1016/j.bmcl.2020.127176 BindingDB Entry DOI: 10.7270/Q2W380XV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP4 subtype (Homo sapiens (Human)) | BDBM50276262 (CHEMBL4129609) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | >2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Asahi Kasei Pharma Corporation Curated by ChEMBL | Assay Description Binding affinity to EP4 receptor (unknown origin) | Bioorg Med Chem Lett 28: 2408-2412 (2018) Article DOI: 10.1016/j.bmcl.2018.06.022 BindingDB Entry DOI: 10.7270/Q2T72KZT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50545480 (CHEMBL4636799) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 3.48E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kitasato University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cells | Bioorg Med Chem Lett 30: (2020) Article DOI: 10.1016/j.bmcl.2020.127176 BindingDB Entry DOI: 10.7270/Q2W380XV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tissue alpha-L-fucosidase (Homo sapiens (Human)) | BDBM50065259 ((2R,3R,4R,5R)-2-(Hydroxymethyl)piperidine-3,4,5-tr...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid UniChem Patents Similars | Article PubMed | 4.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-L-fucosidase of bovine epididymis by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50276262 (CHEMBL4129609) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | >5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Asahi Kasei Pharma Corporation Curated by ChEMBL | Assay Description Binding affinity to EP3 receptor (unknown origin) | Bioorg Med Chem Lett 28: 2408-2412 (2018) Article DOI: 10.1016/j.bmcl.2018.06.022 BindingDB Entry DOI: 10.7270/Q2T72KZT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Homo sapiens (Human)) | BDBM50545479 (CHEMBL4638693) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 5.99E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kitasato University Curated by ChEMBL | Assay Description Displacement of [3H]U69593 from human kappa opioid receptor expressed in CHO cells | Bioorg Med Chem Lett 30: (2020) Article DOI: 10.1016/j.bmcl.2020.127176 BindingDB Entry DOI: 10.7270/Q2W380XV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Maltase-glucoamylase (Homo sapiens (Human)) | BDBM50408431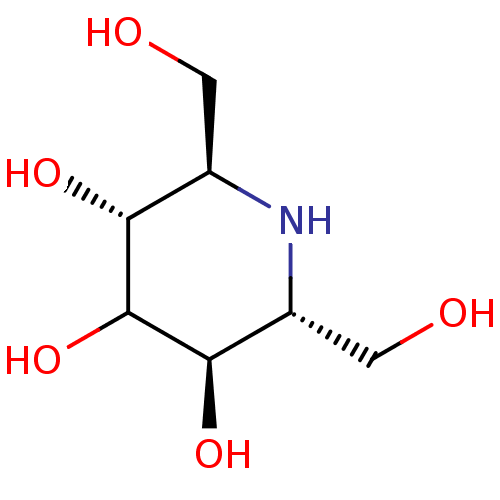 (CHEMBL2114210) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Compound was tested for binding affinity against alpha-glucosidase | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Maltase-glucoamylase (Homo sapiens (Human)) | BDBM50065255 ((R)-2,6-Bis-hydroxymethyl-piperidine-3,4,5-triol |...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Compound was tested for binding affinity against alpha-glucosidase | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (Homo sapiens (Human)) | BDBM50545479 (CHEMBL4638693) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 8.67E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kitasato University Curated by ChEMBL | Assay Description Displacement of [3H]DAMGO from human mu opioid receptor expressed in CHO cells | Bioorg Med Chem Lett 30: (2020) Article DOI: 10.1016/j.bmcl.2020.127176 BindingDB Entry DOI: 10.7270/Q2W380XV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP2 subtype (Homo sapiens (Human)) | BDBM50276262 (CHEMBL4129609) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | >8.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Asahi Kasei Pharma Corporation Curated by ChEMBL | Assay Description Binding affinity to EP2 receptor (unknown origin) | Bioorg Med Chem Lett 28: 2408-2412 (2018) Article DOI: 10.1016/j.bmcl.2018.06.022 BindingDB Entry DOI: 10.7270/Q2T72KZT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP1 subtype (Homo sapiens (Human)) | BDBM50276262 (CHEMBL4129609) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Asahi Kasei Pharma Corporation Curated by ChEMBL | Assay Description Antagonist activity at EP1 receptor (unknown origin) by reporter gene assay | Bioorg Med Chem Lett 28: 2408-2412 (2018) Article DOI: 10.1016/j.bmcl.2018.06.022 BindingDB Entry DOI: 10.7270/Q2T72KZT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP1 subtype (Homo sapiens (Human)) | BDBM50449137 (CHEMBL3127163) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Asahi Kasei Pharma Corporation Curated by ChEMBL | Assay Description Antagonist activity at EP1 receptor (unknown origin) by reporter gene assay | Bioorg Med Chem Lett 28: 2408-2412 (2018) Article DOI: 10.1016/j.bmcl.2018.06.022 BindingDB Entry DOI: 10.7270/Q2T72KZT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP1 subtype (Homo sapiens (Human)) | BDBM50276250 (CHEMBL4126319) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Asahi Kasei Pharma Corporation Curated by ChEMBL | Assay Description Antagonist activity at EP1 receptor (unknown origin) by reporter gene assay | Bioorg Med Chem Lett 28: 2408-2412 (2018) Article DOI: 10.1016/j.bmcl.2018.06.022 BindingDB Entry DOI: 10.7270/Q2T72KZT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP1 subtype (Homo sapiens (Human)) | BDBM50276264 (CHEMBL4128163) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Asahi Kasei Pharma Corporation Curated by ChEMBL | Assay Description Antagonist activity at EP1 receptor (unknown origin) by reporter gene assay | Bioorg Med Chem Lett 28: 2408-2412 (2018) Article DOI: 10.1016/j.bmcl.2018.06.022 BindingDB Entry DOI: 10.7270/Q2T72KZT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP1 subtype (Homo sapiens (Human)) | BDBM50276263 (CHEMBL4129401) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Asahi Kasei Pharma Corporation Curated by ChEMBL | Assay Description Antagonist activity at EP1 receptor (unknown origin) by reporter gene assay | Bioorg Med Chem Lett 28: 2408-2412 (2018) Article DOI: 10.1016/j.bmcl.2018.06.022 BindingDB Entry DOI: 10.7270/Q2T72KZT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP1 subtype (Homo sapiens (Human)) | BDBM50276265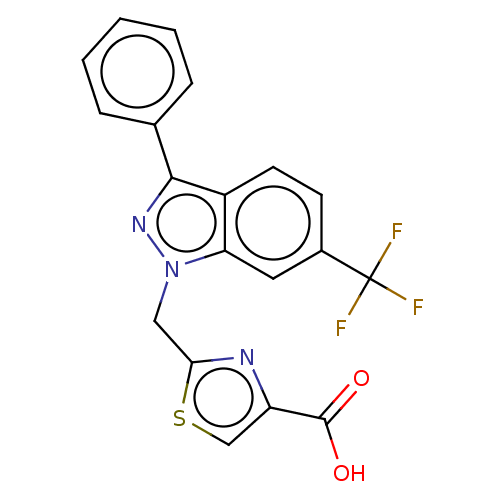 (CHEMBL4129545) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Asahi Kasei Pharma Corporation Curated by ChEMBL | Assay Description Antagonist activity at EP1 receptor (unknown origin) by reporter gene assay | Bioorg Med Chem Lett 28: 2408-2412 (2018) Article DOI: 10.1016/j.bmcl.2018.06.022 BindingDB Entry DOI: 10.7270/Q2T72KZT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP1 subtype (Homo sapiens (Human)) | BDBM50276249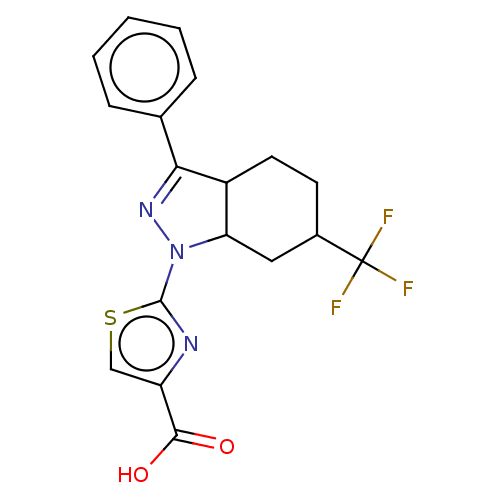 (CHEMBL4126167) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Asahi Kasei Pharma Corporation Curated by ChEMBL | Assay Description Antagonist activity at EP1 receptor (unknown origin) by reporter gene assay | Bioorg Med Chem Lett 28: 2408-2412 (2018) Article DOI: 10.1016/j.bmcl.2018.06.022 BindingDB Entry DOI: 10.7270/Q2T72KZT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50065252 (2,6-Bis-hydroxymethyl-1-methyl-piperidine-3,4,5-tr...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rat intestinal sucrase by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Maltase-glucoamylase (Homo sapiens (Human)) | BDBM50259956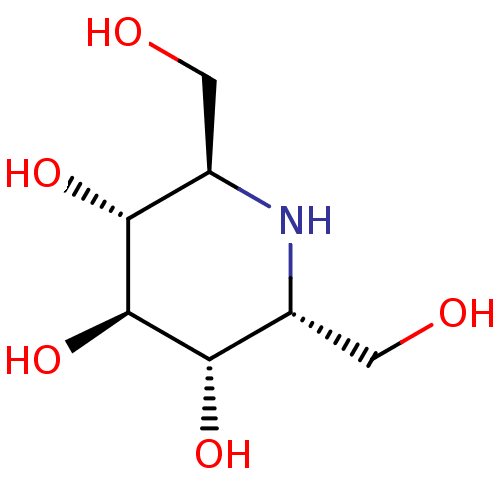 (2,6-Bis-hydroxymethyl-piperidine-3,4,5-triol | CHE...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rice by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP1 subtype (Homo sapiens (Human)) | BDBM50276266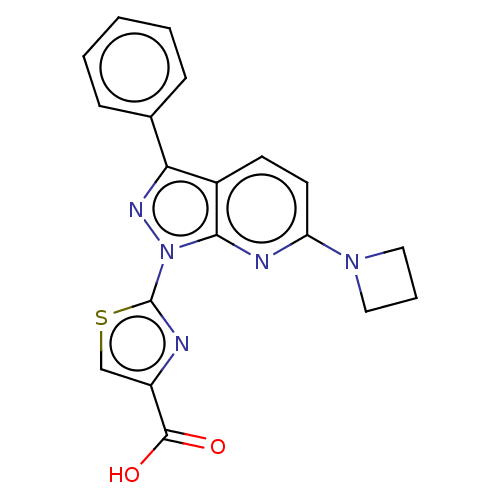 (CHEMBL4126096) | KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Asahi Kasei Pharma Corporation Curated by ChEMBL | Assay Description Antagonist activity at EP1 receptor (unknown origin) by reporter gene assay | Bioorg Med Chem Lett 28: 2408-2412 (2018) Article DOI: 10.1016/j.bmcl.2018.06.022 BindingDB Entry DOI: 10.7270/Q2T72KZT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50259956 (2,6-Bis-hydroxymethyl-piperidine-3,4,5-triol | CHE...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rat intestinal sucrase by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50259956 (2,6-Bis-hydroxymethyl-piperidine-3,4,5-triol | CHE...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rat liver lysosomal by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50259956 (2,6-Bis-hydroxymethyl-piperidine-3,4,5-triol | CHE...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rat intestinal maltase by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Maltase-glucoamylase (Homo sapiens (Human)) | BDBM50065255 ((R)-2,6-Bis-hydroxymethyl-piperidine-3,4,5-triol |...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rice by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50259956 (2,6-Bis-hydroxymethyl-piperidine-3,4,5-triol | CHE...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rat intestinal isomaltase by colorimetric assay using the D-glucose oxidase-peroxidase meth... | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50065255 ((R)-2,6-Bis-hydroxymethyl-piperidine-3,4,5-triol |...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rat intestinal sucrase by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50065252 (2,6-Bis-hydroxymethyl-1-methyl-piperidine-3,4,5-tr...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rat liver lysosomal by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50065252 (2,6-Bis-hydroxymethyl-1-methyl-piperidine-3,4,5-tr...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rat intestinal maltase by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Maltase-glucoamylase (Homo sapiens (Human)) | BDBM50065252 (2,6-Bis-hydroxymethyl-1-methyl-piperidine-3,4,5-tr...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rice by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50065255 ((R)-2,6-Bis-hydroxymethyl-piperidine-3,4,5-triol |...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rat intestinal maltase by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50065253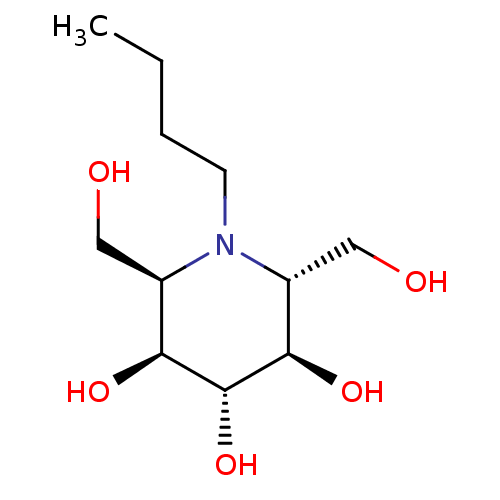 (1-Butyl-2,6-bis-hydroxymethyl-piperidine-3,4,5-tri...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rat liver lysosomal by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tissue alpha-L-fucosidase (Homo sapiens (Human)) | BDBM50408432 (CHEMBL2115215) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-L-fucosidase of bovine epididymis by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50065255 ((R)-2,6-Bis-hydroxymethyl-piperidine-3,4,5-triol |...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rat liver lysosomal by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50065252 (2,6-Bis-hydroxymethyl-1-methyl-piperidine-3,4,5-tr...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rat intestinal isomaltase by colorimetric assay using the D-glucose oxidase-peroxidase meth... | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50408432 (CHEMBL2115215) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rat intestinal sucrase by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysosomal alpha-glucosidase (Rattus norvegicus) | BDBM50065253 (1-Butyl-2,6-bis-hydroxymethyl-piperidine-3,4,5-tri...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rat intestinal sucrase by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Maltase-glucoamylase (Homo sapiens (Human)) | BDBM50408432 (CHEMBL2115215) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-glucosidase of rice by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tissue alpha-L-fucosidase (Rattus norvegicus) | BDBM50408432 (CHEMBL2115215) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokuriku University Curated by ChEMBL | Assay Description Inhibitory activity measured against alpha-L-fucosidase of rat epididymis by colorimetric assay using the D-glucose oxidase-peroxidase method | J Med Chem 41: 2565-71 (1998) Article DOI: 10.1021/jm970836l BindingDB Entry DOI: 10.7270/Q25D8SHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 90 total ) | Next | Last >> |