 Found 16 hits with Last Name = 'abou-taleb' and Initial = 'n'
Found 16 hits with Last Name = 'abou-taleb' and Initial = 'n' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM7533
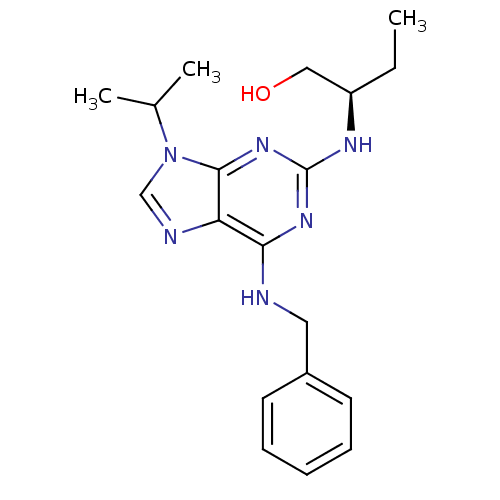
((2R)-2-[[6-(benzylamino)-9-isopropyl-purin-2-yl]am...)Show SMILES CC[C@H](CO)Nc1nc(NCc2ccccc2)c2ncn(C(C)C)c2n1 |r| Show InChI InChI=1S/C19H26N6O/c1-4-15(11-26)22-19-23-17(20-10-14-8-6-5-7-9-14)16-18(24-19)25(12-21-16)13(2)3/h5-9,12-13,15,26H,4,10-11H2,1-3H3,(H2,20,22,23,24)/t15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 82 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CDK1/cyclinB (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.bmc.2020.115633
BindingDB Entry DOI: 10.7270/Q2HQ43NS |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase/G2/mitotic-specific cyclin- 1
(Homo sapiens (Human)) | BDBM50560543

(CHEMBL4797771)Show SMILES COc1ccc(c(OC)c1)-n1c2nc(C)nc(Cl)c2sc1=S |(11.56,-15.68,;12.66,-14.6,;12.27,-13.11,;10.78,-12.7,;10.39,-11.21,;11.49,-10.14,;12.97,-10.54,;14.06,-9.45,;15.55,-9.85,;13.36,-12.02,;11.1,-8.65,;9.63,-8.17,;8.29,-8.94,;6.96,-8.17,;5.62,-8.94,;6.96,-6.63,;8.29,-5.85,;8.28,-4.31,;9.62,-6.62,;11.1,-6.14,;12.01,-7.39,;13.56,-7.39,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 97 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of CDK1/cyclinB (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.bmc.2020.115633
BindingDB Entry DOI: 10.7270/Q2HQ43NS |
More data for this
Ligand-Target Pair | |
DNA polymerase alpha catalytic subunit
(Homo sapiens (Human)) | BDBM50090910
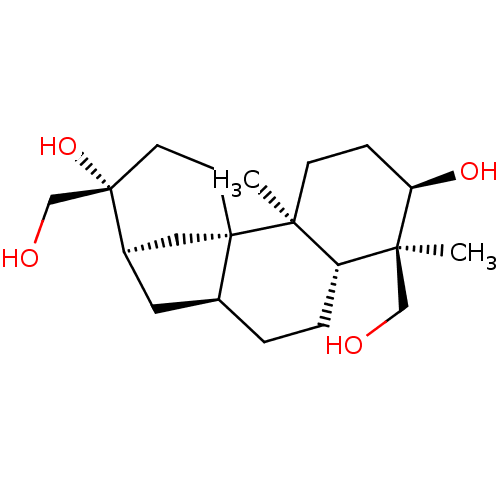
(3alpha,16,17,18-tetrahydroxyaphidicolone | 6,13-di...)Show SMILES C[C@@]1(CO)[C@H](O)CC[C@@]2(C)[C@H]1CC[C@H]1C[C@@H]3C[C@]21CC[C@]3(O)CO |r| Show InChI InChI=1S/C20H34O4/c1-17(11-21)15-4-3-13-9-14-10-19(13,7-8-20(14,24)12-22)18(15,2)6-5-16(17)23/h13-16,21-24H,3-12H2,1-2H3/t13-,14+,15-,16+,17-,18-,19-,20-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human DNA polymerase alpha using 5' end radiolabeled 24nt DNA/48nt DNA as primer/template after 5 mins by PAGE analysis |
J Med Chem 60: 5424-5437 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00067
BindingDB Entry DOI: 10.7270/Q2BZ6873 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM294045
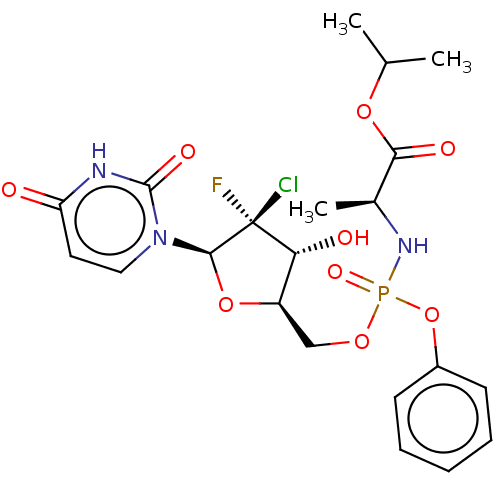
(US10106571, Example 2 | US10106571, Example 23)Show SMILES CC(C)OC(=O)[C@H](C)NP(=O)(OC[C@H]1O[C@@H](n2ccc(=O)[nH]c2=O)[C@@](F)(Cl)[C@@H]1O)Oc1ccccc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human CYP3A4 using ketoconazole as substrate |
J Med Chem 60: 5424-5437 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00067
BindingDB Entry DOI: 10.7270/Q2BZ6873 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50239940

(CHEBI:85083 | PSI-7977, GS-7977 | Sofosbuvir | Sov...)Show SMILES CC(C)OC(=O)[C@H](C)N[P@](=O)(OC[C@H]1O[C@@H](n2ccc(=O)[nH]c2=O)[C@](C)(F)[C@@H]1O)Oc1ccccc1 |r| Show InChI InChI=1S/C22H29FN3O9P/c1-13(2)33-19(29)14(3)25-36(31,35-15-8-6-5-7-9-15)32-12-16-18(28)22(4,23)20(34-16)26-11-10-17(27)24-21(26)30/h5-11,13-14,16,18,20,28H,12H2,1-4H3,(H,25,31)(H,24,27,30)/t14-,16+,18+,20+,22+,36-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human CYP3A4 using ketoconazole as substrate |
J Med Chem 60: 5424-5437 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00067
BindingDB Entry DOI: 10.7270/Q2BZ6873 |
More data for this
Ligand-Target Pair | |
DNA polymerase beta
(Homo sapiens (Human)) | BDBM50020676

(CHEBI:41846 | CHEMBL566812)Show SMILES Cc1cn([C@H]2CC[C@@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)O2)c(=O)[nH]c1=O |r| Show InChI InChI=1S/C10H17N2O13P3/c1-6-4-12(10(14)11-9(6)13)8-3-2-7(23-8)5-22-27(18,19)25-28(20,21)24-26(15,16)17/h4,7-8H,2-3,5H2,1H3,(H,18,19)(H,20,21)(H,11,13,14)(H2,15,16,17)/t7-,8+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human DNA polymerase beta using 5' end radiolabeled 24nt DNA/48nt DNA as primer/template after 5 mins by PAGE analysis |
J Med Chem 60: 5424-5437 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00067
BindingDB Entry DOI: 10.7270/Q2BZ6873 |
More data for this
Ligand-Target Pair | |
DNA polymerase alpha catalytic subunit
(Homo sapiens (Human)) | BDBM50239939
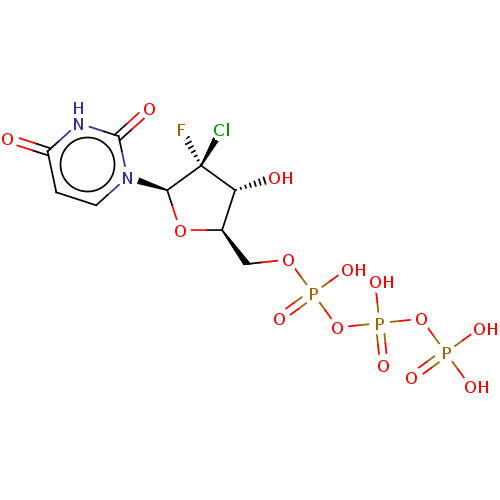
(CHEMBL4088430)Show SMILES O[C@@H]1[C@@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)O[C@@H](n2ccc(=O)[nH]c2=O)[C@@]1(F)Cl |r| Show InChI InChI=1S/C9H13ClFN2O14P3/c10-9(11)6(15)4(25-7(9)13-2-1-5(14)12-8(13)16)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h1-2,4,6-7,15H,3H2,(H,20,21)(H,22,23)(H,12,14,16)(H2,17,18,19)/t4-,6-,7-,9-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.02E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human DNA polymerase alpha using 5' end radiolabeled 24nt DNA/48nt DNA as primer/template after 5 mins by PAGE analysis |
J Med Chem 60: 5424-5437 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00067
BindingDB Entry DOI: 10.7270/Q2BZ6873 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM294045

(US10106571, Example 2 | US10106571, Example 23)Show SMILES CC(C)OC(=O)[C@H](C)NP(=O)(OC[C@H]1O[C@@H](n2ccc(=O)[nH]c2=O)[C@@](F)(Cl)[C@@H]1O)Oc1ccccc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2C9 using sulfaphenazole as substrate |
J Med Chem 60: 5424-5437 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00067
BindingDB Entry DOI: 10.7270/Q2BZ6873 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50239940

(CHEBI:85083 | PSI-7977, GS-7977 | Sofosbuvir | Sov...)Show SMILES CC(C)OC(=O)[C@H](C)N[P@](=O)(OC[C@H]1O[C@@H](n2ccc(=O)[nH]c2=O)[C@](C)(F)[C@@H]1O)Oc1ccccc1 |r| Show InChI InChI=1S/C22H29FN3O9P/c1-13(2)33-19(29)14(3)25-36(31,35-15-8-6-5-7-9-15)32-12-16-18(28)22(4,23)20(34-16)26-11-10-17(27)24-21(26)30/h5-11,13-14,16,18,20,28H,12H2,1-4H3,(H,25,31)(H,24,27,30)/t14-,16+,18+,20+,22+,36-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2C9 using sulfaphenazole as substrate |
J Med Chem 60: 5424-5437 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00067
BindingDB Entry DOI: 10.7270/Q2BZ6873 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50239940

(CHEBI:85083 | PSI-7977, GS-7977 | Sofosbuvir | Sov...)Show SMILES CC(C)OC(=O)[C@H](C)N[P@](=O)(OC[C@H]1O[C@@H](n2ccc(=O)[nH]c2=O)[C@](C)(F)[C@@H]1O)Oc1ccccc1 |r| Show InChI InChI=1S/C22H29FN3O9P/c1-13(2)33-19(29)14(3)25-36(31,35-15-8-6-5-7-9-15)32-12-16-18(28)22(4,23)20(34-16)26-11-10-17(27)24-21(26)30/h5-11,13-14,16,18,20,28H,12H2,1-4H3,(H,25,31)(H,24,27,30)/t14-,16+,18+,20+,22+,36-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2D6 using quinidine as substrate |
J Med Chem 60: 5424-5437 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00067
BindingDB Entry DOI: 10.7270/Q2BZ6873 |
More data for this
Ligand-Target Pair | |
DNA polymerase beta
(Homo sapiens (Human)) | BDBM50333129
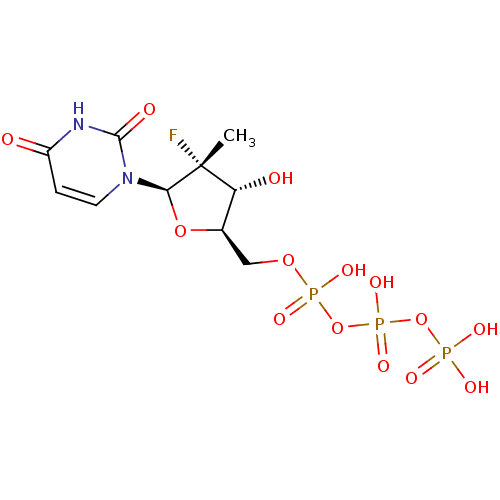
(((2R,3R,4R,5R)-5-(2,4-dioxo-3,4-dihydropyrimidin-1...)Show SMILES C[C@@]1(F)[C@H](O)[C@@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)O[C@H]1n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C10H16FN2O14P3/c1-10(11)7(15)5(25-8(10)13-3-2-6(14)12-9(13)16)4-24-29(20,21)27-30(22,23)26-28(17,18)19/h2-3,5,7-8,15H,4H2,1H3,(H,20,21)(H,22,23)(H,12,14,16)(H2,17,18,19)/t5-,7-,8-,10-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human DNA polymerase beta using 5' end radiolabeled 24nt DNA/48nt DNA as primer/template after 5 mins by PAGE analysis |
J Med Chem 60: 5424-5437 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00067
BindingDB Entry DOI: 10.7270/Q2BZ6873 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50239940

(CHEBI:85083 | PSI-7977, GS-7977 | Sofosbuvir | Sov...)Show SMILES CC(C)OC(=O)[C@H](C)N[P@](=O)(OC[C@H]1O[C@@H](n2ccc(=O)[nH]c2=O)[C@](C)(F)[C@@H]1O)Oc1ccccc1 |r| Show InChI InChI=1S/C22H29FN3O9P/c1-13(2)33-19(29)14(3)25-36(31,35-15-8-6-5-7-9-15)32-12-16-18(28)22(4,23)20(34-16)26-11-10-17(27)24-21(26)30/h5-11,13-14,16,18,20,28H,12H2,1-4H3,(H,25,31)(H,24,27,30)/t14-,16+,18+,20+,22+,36-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human CYP1A2 using alpha-naphthoflavone as substrate |
J Med Chem 60: 5424-5437 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00067
BindingDB Entry DOI: 10.7270/Q2BZ6873 |
More data for this
Ligand-Target Pair | |
DNA polymerase beta
(Homo sapiens (Human)) | BDBM50239939

(CHEMBL4088430)Show SMILES O[C@@H]1[C@@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)O[C@@H](n2ccc(=O)[nH]c2=O)[C@@]1(F)Cl |r| Show InChI InChI=1S/C9H13ClFN2O14P3/c10-9(11)6(15)4(25-7(9)13-2-1-5(14)12-8(13)16)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h1-2,4,6-7,15H,3H2,(H,20,21)(H,22,23)(H,12,14,16)(H2,17,18,19)/t4-,6-,7-,9-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human DNA polymerase beta using 5' end radiolabeled 24nt DNA/48nt DNA as primer/template after 5 mins by PAGE analysis |
J Med Chem 60: 5424-5437 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00067
BindingDB Entry DOI: 10.7270/Q2BZ6873 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM294045

(US10106571, Example 2 | US10106571, Example 23)Show SMILES CC(C)OC(=O)[C@H](C)NP(=O)(OC[C@H]1O[C@@H](n2ccc(=O)[nH]c2=O)[C@@](F)(Cl)[C@@H]1O)Oc1ccccc1 |r| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human CYP2D6 using quinidine as substrate |
J Med Chem 60: 5424-5437 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00067
BindingDB Entry DOI: 10.7270/Q2BZ6873 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM294045

(US10106571, Example 2 | US10106571, Example 23)Show SMILES CC(C)OC(=O)[C@H](C)NP(=O)(OC[C@H]1O[C@@H](n2ccc(=O)[nH]c2=O)[C@@](F)(Cl)[C@@H]1O)Oc1ccccc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of human CYP1A2 using alpha-naphthoflavone as substrate |
J Med Chem 60: 5424-5437 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00067
BindingDB Entry DOI: 10.7270/Q2BZ6873 |
More data for this
Ligand-Target Pair | |
DNA polymerase alpha catalytic subunit
(Homo sapiens (Human)) | BDBM50333129

(((2R,3R,4R,5R)-5-(2,4-dioxo-3,4-dihydropyrimidin-1...)Show SMILES C[C@@]1(F)[C@H](O)[C@@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)O[C@H]1n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C10H16FN2O14P3/c1-10(11)7(15)5(25-8(10)13-3-2-6(14)12-9(13)16)4-24-29(20,21)27-30(22,23)26-28(17,18)19/h2-3,5,7-8,15H,4H2,1H3,(H,20,21)(H,22,23)(H,12,14,16)(H2,17,18,19)/t5-,7-,8-,10-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Competitive inhibition of leucine aminopeptidase; Ki value reporting the slope effect(Kis) |
J Med Chem 60: 5424-5437 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00067
BindingDB Entry DOI: 10.7270/Q2BZ6873 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data