 Found 72 hits with Last Name = 'bonacorsi' and Initial = 's'
Found 72 hits with Last Name = 'bonacorsi' and Initial = 's' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM50376416
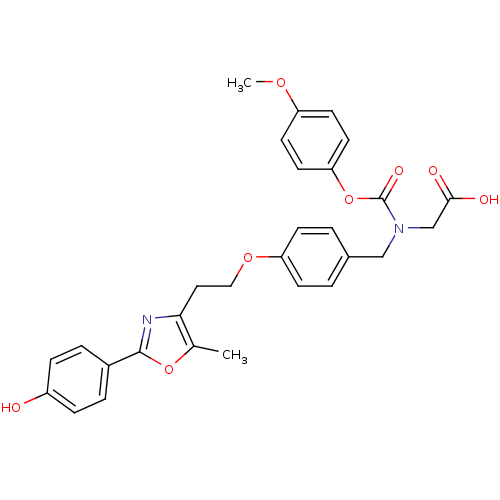
(CHEMBL253952)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCc3nc(oc3C)-c3ccc(O)cc3)cc2)cc1 Show InChI InChI=1S/C29H28N2O8/c1-19-26(30-28(38-19)21-5-7-22(32)8-6-21)15-16-37-24-9-3-20(4-10-24)17-31(18-27(33)34)29(35)39-25-13-11-23(36-2)12-14-25/h3-14,32H,15-18H2,1-2H3,(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 99 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR alpha receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM50150998
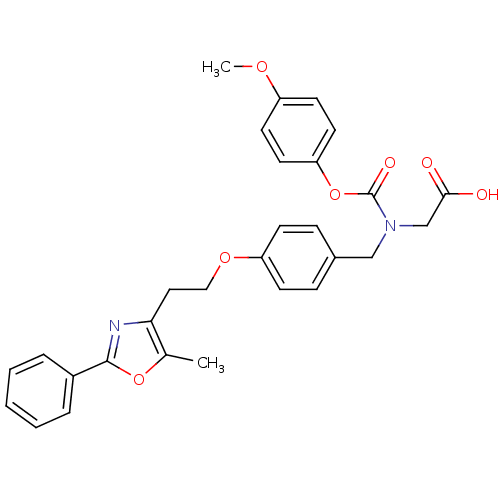
(((4-Methoxy-phenoxycarbonyl)-{4-[2-(5-methyl-2-phe...)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCc3nc(oc3C)-c3ccccc3)cc2)cc1 Show InChI InChI=1S/C29H28N2O7/c1-20-26(30-28(37-20)22-6-4-3-5-7-22)16-17-36-24-10-8-21(9-11-24)18-31(19-27(32)33)29(34)38-25-14-12-23(35-2)13-15-25/h3-15H,16-19H2,1-2H3,(H,32,33) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 188 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR alpha receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50150998

(((4-Methoxy-phenoxycarbonyl)-{4-[2-(5-methyl-2-phe...)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCc3nc(oc3C)-c3ccccc3)cc2)cc1 Show InChI InChI=1S/C29H28N2O7/c1-20-26(30-28(37-20)22-6-4-3-5-7-22)16-17-36-24-10-8-21(9-11-24)18-31(19-27(32)33)29(34)38-25-14-12-23(35-2)13-15-25/h3-15H,16-19H2,1-2H3,(H,32,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 247 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR gamma receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50150998

(((4-Methoxy-phenoxycarbonyl)-{4-[2-(5-methyl-2-phe...)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCc3nc(oc3C)-c3ccccc3)cc2)cc1 Show InChI InChI=1S/C29H28N2O7/c1-20-26(30-28(37-20)22-6-4-3-5-7-22)16-17-36-24-10-8-21(9-11-24)18-31(19-27(32)33)29(34)38-25-14-12-23(35-2)13-15-25/h3-15H,16-19H2,1-2H3,(H,32,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Agonist activity at PPAR gamma in human HEK cells by transactivation assay |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50376416

(CHEMBL253952)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCc3nc(oc3C)-c3ccc(O)cc3)cc2)cc1 Show InChI InChI=1S/C29H28N2O8/c1-19-26(30-28(38-19)21-5-7-22(32)8-6-21)15-16-37-24-9-3-20(4-10-24)17-31(18-27(33)34)29(35)39-25-13-11-23(36-2)12-14-25/h3-14,32H,15-18H2,1-2H3,(H,33,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.05E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR gamma receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM50376415
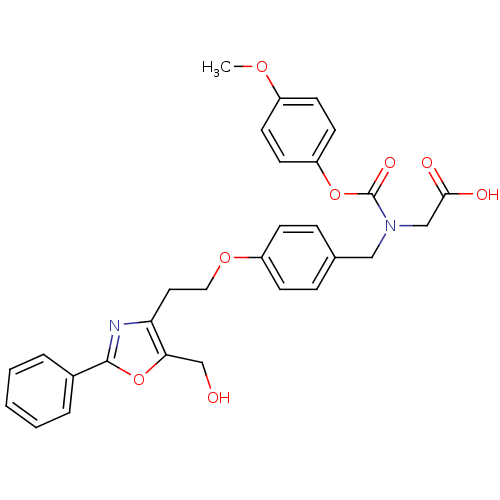
(CHEMBL429671)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCc3nc(oc3CO)-c3ccccc3)cc2)cc1 Show InChI InChI=1S/C29H28N2O8/c1-36-22-11-13-24(14-12-22)38-29(35)31(18-27(33)34)17-20-7-9-23(10-8-20)37-16-15-25-26(19-32)39-28(30-25)21-5-3-2-4-6-21/h2-14,32H,15-19H2,1H3,(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.09E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR alpha receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM50376414
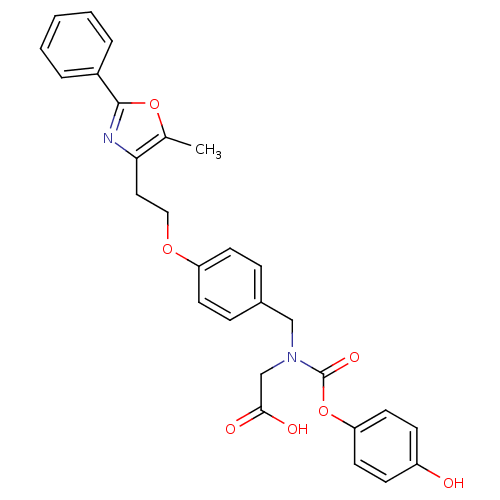
(CHEMBL254989)Show SMILES Cc1oc(nc1CCOc1ccc(CN(CC(O)=O)C(=O)Oc2ccc(O)cc2)cc1)-c1ccccc1 Show InChI InChI=1S/C28H26N2O7/c1-19-25(29-27(36-19)21-5-3-2-4-6-21)15-16-35-23-11-7-20(8-12-23)17-30(18-26(32)33)28(34)37-24-13-9-22(31)10-14-24/h2-14,31H,15-18H2,1H3,(H,32,33) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.74E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR alpha receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50376412
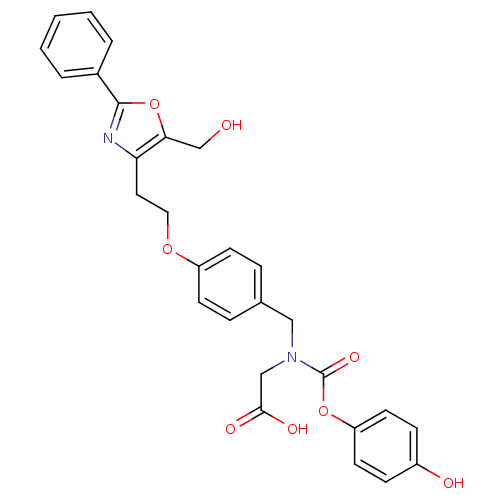
(CHEMBL254347)Show SMILES OCc1oc(nc1CCOc1ccc(CN(CC(O)=O)C(=O)Oc2ccc(O)cc2)cc1)-c1ccccc1 Show InChI InChI=1S/C28H26N2O8/c31-18-25-24(29-27(38-25)20-4-2-1-3-5-20)14-15-36-22-10-6-19(7-11-22)16-30(17-26(33)34)28(35)37-23-12-8-21(32)9-13-23/h1-13,31-32H,14-18H2,(H,33,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR gamma receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM50376410

(CHEMBL257761)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCC(=O)N(C(C)=O)C(=O)c3ccccc3)cc2)cc1 Show InChI InChI=1S/C29H28N2O9/c1-20(32)31(28(36)22-6-4-3-5-7-22)26(33)16-17-39-24-10-8-21(9-11-24)18-30(19-27(34)35)29(37)40-25-14-12-23(38-2)13-15-25/h3-15H,16-19H2,1-2H3,(H,34,35) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR alpha receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50376415

(CHEMBL429671)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCc3nc(oc3CO)-c3ccccc3)cc2)cc1 Show InChI InChI=1S/C29H28N2O8/c1-36-22-11-13-24(14-12-22)38-29(35)31(18-27(33)34)17-20-7-9-23(10-8-20)37-16-15-25-26(19-32)39-28(30-25)21-5-3-2-4-6-21/h2-14,32H,15-19H2,1H3,(H,33,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR gamma receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50376414

(CHEMBL254989)Show SMILES Cc1oc(nc1CCOc1ccc(CN(CC(O)=O)C(=O)Oc2ccc(O)cc2)cc1)-c1ccccc1 Show InChI InChI=1S/C28H26N2O7/c1-19-25(29-27(36-19)21-5-3-2-4-6-21)15-16-35-23-11-7-20(8-12-23)17-30(18-26(32)33)28(34)37-24-13-9-22(31)10-14-24/h2-14,31H,15-18H2,1H3,(H,32,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR gamma receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50376410

(CHEMBL257761)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCC(=O)N(C(C)=O)C(=O)c3ccccc3)cc2)cc1 Show InChI InChI=1S/C29H28N2O9/c1-20(32)31(28(36)22-6-4-3-5-7-22)26(33)16-17-39-24-10-8-21(9-11-24)18-30(19-27(34)35)29(37)40-25-14-12-23(38-2)13-15-25/h3-15H,16-19H2,1-2H3,(H,34,35) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR gamma receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM50376413
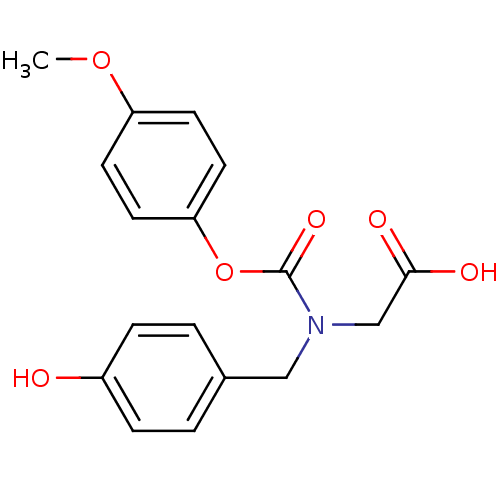
(CHEMBL444373)Show InChI InChI=1S/C17H17NO6/c1-23-14-6-8-15(9-7-14)24-17(22)18(11-16(20)21)10-12-2-4-13(19)5-3-12/h2-9,19H,10-11H2,1H3,(H,20,21) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR alpha receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM50376412

(CHEMBL254347)Show SMILES OCc1oc(nc1CCOc1ccc(CN(CC(O)=O)C(=O)Oc2ccc(O)cc2)cc1)-c1ccccc1 Show InChI InChI=1S/C28H26N2O8/c31-18-25-24(29-27(38-25)20-4-2-1-3-5-20)14-15-36-22-10-6-19(7-11-22)16-30(17-26(33)34)28(35)37-23-12-8-21(32)9-13-23/h1-13,31-32H,14-18H2,(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR alpha receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50376411
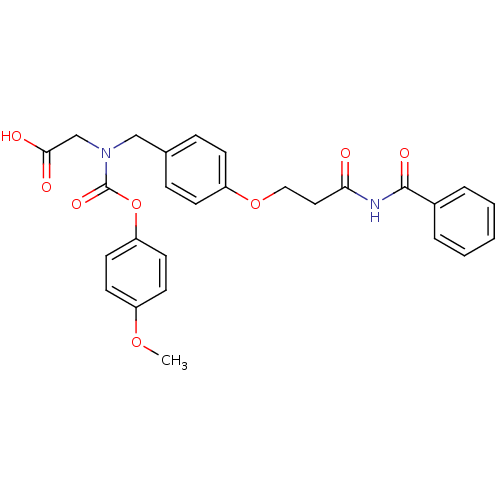
(CHEMBL401829)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCC(=O)NC(=O)c3ccccc3)cc2)cc1 Show InChI InChI=1S/C27H26N2O8/c1-35-21-11-13-23(14-12-21)37-27(34)29(18-25(31)32)17-19-7-9-22(10-8-19)36-16-15-24(30)28-26(33)20-5-3-2-4-6-20/h2-14H,15-18H2,1H3,(H,31,32)(H,28,30,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR gamma receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor alpha
(Homo sapiens (Human)) | BDBM50376411

(CHEMBL401829)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCC(=O)NC(=O)c3ccccc3)cc2)cc1 Show InChI InChI=1S/C27H26N2O8/c1-35-21-11-13-23(14-12-21)37-27(34)29(18-25(31)32)17-19-7-9-22(10-8-19)36-16-15-24(30)28-26(33)20-5-3-2-4-6-20/h2-14H,15-18H2,1H3,(H,31,32)(H,28,30,33) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR alpha receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50376413

(CHEMBL444373)Show InChI InChI=1S/C17H17NO6/c1-23-14-6-8-15(9-7-14)24-17(22)18(11-16(20)21)10-12-2-4-13(19)5-3-12/h2-9,19H,10-11H2,1H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Binding affinity at PPAR gamma receptor |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50376416

(CHEMBL253952)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCc3nc(oc3C)-c3ccc(O)cc3)cc2)cc1 Show InChI InChI=1S/C29H28N2O8/c1-19-26(30-28(38-19)21-5-7-22(32)8-6-21)15-16-37-24-9-3-20(4-10-24)17-31(18-27(33)34)29(35)39-25-13-11-23(36-2)12-14-25/h3-14,32H,15-18H2,1-2H3,(H,33,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.98E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Agonist activity at PPAR gamma in human HEK cells by transactivation assay |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50376413

(CHEMBL444373)Show InChI InChI=1S/C17H17NO6/c1-23-14-6-8-15(9-7-14)24-17(22)18(11-16(20)21)10-12-2-4-13(19)5-3-12/h2-9,19H,10-11H2,1H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Agonist activity at PPAR gamma in human HEK cells by transactivation assay |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50376415

(CHEMBL429671)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCc3nc(oc3CO)-c3ccccc3)cc2)cc1 Show InChI InChI=1S/C29H28N2O8/c1-36-22-11-13-24(14-12-22)38-29(35)31(18-27(33)34)17-20-7-9-23(10-8-20)37-16-15-25-26(19-32)39-28(30-25)21-5-3-2-4-6-21/h2-14,32H,15-19H2,1H3,(H,33,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Agonist activity at PPAR gamma in human HEK cells by transactivation assay |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50376412

(CHEMBL254347)Show SMILES OCc1oc(nc1CCOc1ccc(CN(CC(O)=O)C(=O)Oc2ccc(O)cc2)cc1)-c1ccccc1 Show InChI InChI=1S/C28H26N2O8/c31-18-25-24(29-27(38-25)20-4-2-1-3-5-20)14-15-36-22-10-6-19(7-11-22)16-30(17-26(33)34)28(35)37-23-12-8-21(32)9-13-23/h1-13,31-32H,14-18H2,(H,33,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Agonist activity at PPAR gamma in human HEK cells by transactivation assay |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50376414

(CHEMBL254989)Show SMILES Cc1oc(nc1CCOc1ccc(CN(CC(O)=O)C(=O)Oc2ccc(O)cc2)cc1)-c1ccccc1 Show InChI InChI=1S/C28H26N2O7/c1-19-25(29-27(36-19)21-5-3-2-4-6-21)15-16-35-23-11-7-20(8-12-23)17-30(18-26(32)33)28(34)37-24-13-9-22(31)10-14-24/h2-14,31H,15-18H2,1H3,(H,32,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Agonist activity at PPAR gamma in human HEK cells by transactivation assay |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50376410

(CHEMBL257761)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCC(=O)N(C(C)=O)C(=O)c3ccccc3)cc2)cc1 Show InChI InChI=1S/C29H28N2O9/c1-20(32)31(28(36)22-6-4-3-5-7-22)26(33)16-17-39-24-10-8-21(9-11-24)18-30(19-27(34)35)29(37)40-25-14-12-23(38-2)13-15-25/h3-15H,16-19H2,1-2H3,(H,34,35) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Agonist activity at PPAR gamma in human HEK cells by transactivation assay |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Peroxisome proliferator-activated receptor gamma
(Homo sapiens (Human)) | BDBM50376411

(CHEMBL401829)Show SMILES COc1ccc(OC(=O)N(CC(O)=O)Cc2ccc(OCCC(=O)NC(=O)c3ccccc3)cc2)cc1 Show InChI InChI=1S/C27H26N2O8/c1-35-21-11-13-23(14-12-21)37-27(34)29(18-25(31)32)17-19-7-9-22(10-8-19)36-16-15-24(30)28-26(33)20-5-3-2-4-6-20/h2-14H,15-18H2,1H3,(H,31,32)(H,28,30,33) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb
Curated by ChEMBL
| Assay Description
Agonist activity at PPAR gamma in human HEK cells by transactivation assay |
Drug Metab Dispos 35: 150-67 (2006)
Article DOI: 10.1124/dmd.106.012450
BindingDB Entry DOI: 10.7270/Q2JS9R92 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 in human liver microsomes assessed as phenacetin O-deethylation after 3 mins by LC-MS/MS analysis in presence of NADPH |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2A6 in human liver microsomes assessed as coumarin 7-hydroxylation after 3 mins by LC-MS/MS analysis in presence of NADPH |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2B6
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2B6 in human liver microsomes assessed as bupropion hydroxylation after 3 mins by LC-MS/MS analysis in presence of NADPH |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C8 in human liver microsomes assessed as paclitaxel 6alpha-hydroxylation after 3 mins by LC-MS/MS analysis in presence of NADPH |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes assessed as paclitaxel 6alpha-hydroxylation after 3 mins by LC-MS/MS analysis in presence of NADPH |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 in human liver microsomes assessed as (S)-mephenytoin 4'-hydroxylation after 3 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes assessed as bufuralol 1'-hydroxylation after 3 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2E1
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2E1 in human liver microsomes assessed as chlorzoxazone 6-hydroxylation after 3 mins by LC-MS/MS analysis in presence of NADPH |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as midazolam 1'-hydroxylation after 3 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as testosterone 6beta-hydroxylation after 3 mins by LC-MS/MS analysis in presence of NADPH |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 in human liver microsomes assessed as phenacetin O-deethylation preincubated for 15 mins by LC-MS/MS analysis in presence of NAD... |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2A6 in human liver microsomes assessed as coumarin 7-hydroxylation preincubated for 15 mins by LC-MS/MS analysis in presence of NADP... |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2B6
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2B6 in human liver microsomes assessed as bupropion hydroxylation preincubated for 15 mins by LC-MS/MS analysis in presence of NADPH |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C8 in human liver microsomes assessed as paclitaxel 6alpha-hydroxylation preincubated for 15 mins by LC-MS/MS analysis in presence ... |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes assessed as paclitaxel 6alpha-hydroxylation preincubated for 15 mins by LC-MS/MS analysis in presence ... |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 in human liver microsomes assessed as (S)-mephenytoin 4'-hydroxylation preincubated for 15 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes assessed as bufuralol 1'-hydroxylation preincubated for 15 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2E1
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2E1 in human liver microsomes assessed as chlorzoxazone 6-hydroxylation preincubated for 15 mins by LC-MS/MS analysis in presence of... |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as midazolam 1'-hydroxylation preincubated for 15 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50225074

((1S,3S,5S)-2-[(S)-2-amino-2-(3-hydroxy-adamantan-1...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(CC(O)(C3)C1)C2 |TLB:1:12:15:20.18.17,THB:13:14:17:22.12.21,13:12:15.14.20:17,21:12:15:20.18.17,21:18:15:22.13.12,19:18:15:22.13.12| Show InChI InChI=1S/C18H25N3O2/c19-8-13-2-12-3-14(12)21(13)16(22)15(20)17-4-10-1-11(5-17)7-18(23,6-10)9-17/h10-15,23H,1-7,9,20H2/t10?,11?,12-,13+,14+,15-,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as testosterone 6beta-hydroxylation preincubated for 15 mins by LC-MS/MS analysis in presence... |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM11543
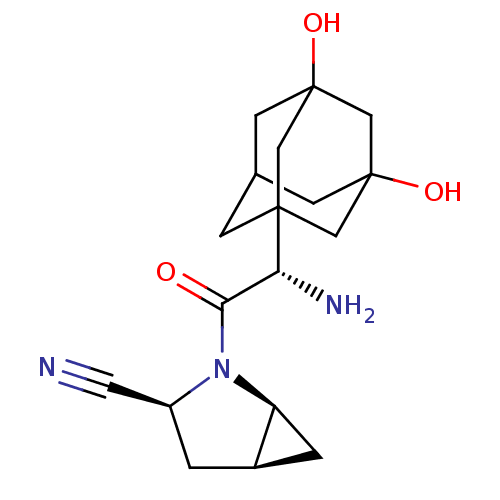
((1S,3S,5S)-2-[(2S)-2-amino-2-(3,5-dihydroxyadamant...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(O)(CC(O)(C3)C1)C2 |r,TLB:21:14:23:18.22.19,21:19:14.15.13:23,20:19:14.15.13:23,17:16:14.13.21:22,15:16:14.13.21:22,THB:15:14:22:18.23.16| Show InChI InChI=1S/C18H25N3O3/c19-6-12-1-11-2-13(11)21(12)15(22)14(20)16-3-10-4-17(23,7-16)9-18(24,5-10)8-16/h10-14,23-24H,1-5,7-9,20H2/t10?,11-,12+,13+,14-,16?,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 in human liver microsomes assessed as phenacetin O-deethylation after 3 mins by LC-MS/MS analysis in presence of NADPH |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM11543

((1S,3S,5S)-2-[(2S)-2-amino-2-(3,5-dihydroxyadamant...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(O)(CC(O)(C3)C1)C2 |r,TLB:21:14:23:18.22.19,21:19:14.15.13:23,20:19:14.15.13:23,17:16:14.13.21:22,15:16:14.13.21:22,THB:15:14:22:18.23.16| Show InChI InChI=1S/C18H25N3O3/c19-6-12-1-11-2-13(11)21(12)15(22)14(20)16-3-10-4-17(23,7-16)9-18(24,5-10)8-16/h10-14,23-24H,1-5,7-9,20H2/t10?,11-,12+,13+,14-,16?,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2A6 in human liver microsomes assessed as coumarin 7-hydroxylation after 3 mins by LC-MS/MS analysis in presence of NADPH |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM11543

((1S,3S,5S)-2-[(2S)-2-amino-2-(3,5-dihydroxyadamant...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(O)(CC(O)(C3)C1)C2 |r,TLB:21:14:23:18.22.19,21:19:14.15.13:23,20:19:14.15.13:23,17:16:14.13.21:22,15:16:14.13.21:22,THB:15:14:22:18.23.16| Show InChI InChI=1S/C18H25N3O3/c19-6-12-1-11-2-13(11)21(12)15(22)14(20)16-3-10-4-17(23,7-16)9-18(24,5-10)8-16/h10-14,23-24H,1-5,7-9,20H2/t10?,11-,12+,13+,14-,16?,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as testosterone 6beta-hydroxylation preincubated for 15 mins by LC-MS/MS analysis in presence... |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM11543

((1S,3S,5S)-2-[(2S)-2-amino-2-(3,5-dihydroxyadamant...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(O)(CC(O)(C3)C1)C2 |r,TLB:21:14:23:18.22.19,21:19:14.15.13:23,20:19:14.15.13:23,17:16:14.13.21:22,15:16:14.13.21:22,THB:15:14:22:18.23.16| Show InChI InChI=1S/C18H25N3O3/c19-6-12-1-11-2-13(11)21(12)15(22)14(20)16-3-10-4-17(23,7-16)9-18(24,5-10)8-16/h10-14,23-24H,1-5,7-9,20H2/t10?,11-,12+,13+,14-,16?,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as midazolam 1'-hydroxylation preincubated for 15 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2E1
(Homo sapiens (Human)) | BDBM11543

((1S,3S,5S)-2-[(2S)-2-amino-2-(3,5-dihydroxyadamant...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(O)(CC(O)(C3)C1)C2 |r,TLB:21:14:23:18.22.19,21:19:14.15.13:23,20:19:14.15.13:23,17:16:14.13.21:22,15:16:14.13.21:22,THB:15:14:22:18.23.16| Show InChI InChI=1S/C18H25N3O3/c19-6-12-1-11-2-13(11)21(12)15(22)14(20)16-3-10-4-17(23,7-16)9-18(24,5-10)8-16/h10-14,23-24H,1-5,7-9,20H2/t10?,11-,12+,13+,14-,16?,17?,18?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2E1 in human liver microsomes assessed as chlorzoxazone 6-hydroxylation preincubated for 15 mins by LC-MS/MS analysis in presence of... |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM11543

((1S,3S,5S)-2-[(2S)-2-amino-2-(3,5-dihydroxyadamant...)Show SMILES N[C@H](C(=O)N1[C@H]2C[C@H]2C[C@H]1C#N)C12CC3CC(O)(CC(O)(C3)C1)C2 |r,TLB:21:14:23:18.22.19,21:19:14.15.13:23,20:19:14.15.13:23,17:16:14.13.21:22,15:16:14.13.21:22,THB:15:14:22:18.23.16| Show InChI InChI=1S/C18H25N3O3/c19-6-12-1-11-2-13(11)21(12)15(22)14(20)16-3-10-4-17(23,7-16)9-18(24,5-10)8-16/h10-14,23-24H,1-5,7-9,20H2/t10?,11-,12+,13+,14-,16?,17?,18?/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 in human liver microsomes assessed as bufuralol 1'-hydroxylation preincubated for 15 mins by LC-MS/MS analysis |
Drug Metab Dispos 40: 1345-56 (2012)
Article DOI: 10.1124/dmd.112.045450
BindingDB Entry DOI: 10.7270/Q27D2WVN |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data