 Found 272 hits with Last Name = 'colletier' and Initial = 'jp'
Found 272 hits with Last Name = 'colletier' and Initial = 'jp' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cholinesterase
(Homo sapiens (Human)) | BDBM50247011

(CHEMBL4080419)Show SMILES Cl.CN(C)CCN(CC1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2ccccc2c1 Show InChI InChI=1S/C30H37N3O.ClH/c1-31(2)16-17-33(30(34)28-14-13-24-9-3-4-10-25(24)18-28)22-23-8-7-15-32(21-23)29-19-26-11-5-6-12-27(26)20-29;/h3-6,9-14,18,23,29H,7-8,15-17,19-22H2,1-2H3;1H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Competitive inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate at pH 8 by stopped flow assay |
J Med Chem 61: 119-139 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01086
BindingDB Entry DOI: 10.7270/Q2S75JRJ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50247011

(CHEMBL4080419)Show SMILES Cl.CN(C)CCN(CC1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2ccccc2c1 Show InChI InChI=1S/C30H37N3O.ClH/c1-31(2)16-17-33(30(34)28-14-13-24-9-3-4-10-25(24)18-28)22-23-8-7-15-32(21-23)29-19-26-11-5-6-12-27(26)20-29;/h3-6,9-14,18,23,29H,7-8,15-17,19-22H2,1-2H3;1H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Competitive inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate at pH 8 by stopped flow assay |
J Med Chem 61: 119-139 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01086
BindingDB Entry DOI: 10.7270/Q2S75JRJ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM8963
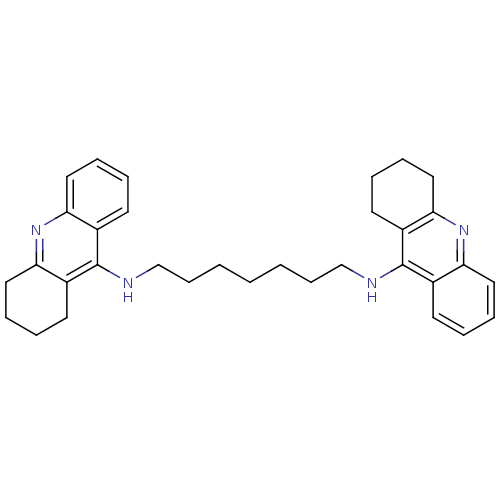
(CHEMBL32823 | Homodimeric Tacrine Analog 3b | N-[7...)Show SMILES C(CCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C33H40N4/c1(2-12-22-34-32-24-14-4-8-18-28(24)36-29-19-9-5-15-25(29)32)3-13-23-35-33-26-16-6-10-20-30(26)37-31-21-11-7-17-27(31)33/h4,6,8,10,14,16,18,20H,1-3,5,7,9,11-13,15,17,19,21-23H2,(H,34,36)(H,35,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.163 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Competitive inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate at pH 8 by stopped flow assay |
J Med Chem 61: 119-139 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01086
BindingDB Entry DOI: 10.7270/Q2S75JRJ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50556952
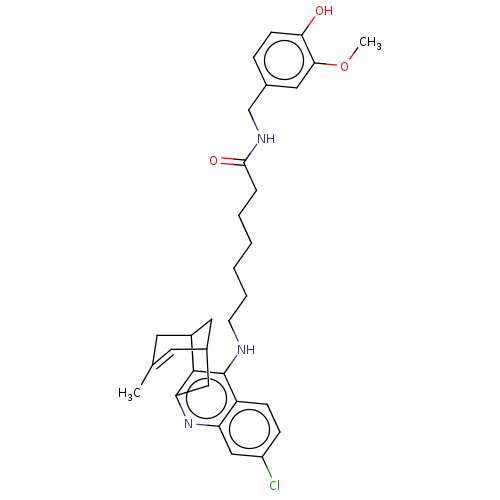
(CHEMBL4754487)Show SMILES COc1cc(CNC(=O)CCCCCCNc2c3C4CC(Cc3nc3cc(Cl)ccc23)C=C(C)C4)ccc1O |t:34,TLB:23:22:19:34.31.32,THB:16:17:19:34.31.32| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 0.680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Mixed-type inhibition of recombinant human AChE assessed as inhibition constant using acetylthiocholine iodide as substrate by Cornish-Bowden plot an... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01775
BindingDB Entry DOI: 10.7270/Q2K077XJ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50556952

(CHEMBL4754487)Show SMILES COc1cc(CNC(=O)CCCCCCNc2c3C4CC(Cc3nc3cc(Cl)ccc23)C=C(C)C4)ccc1O |t:34,TLB:23:22:19:34.31.32,THB:16:17:19:34.31.32| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 0.940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Mixed-type inhibition of recombinant human AChE assessed as dissociation constant for protein-substrate-compound complex using acetylthiocholine iodi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01775
BindingDB Entry DOI: 10.7270/Q2K077XJ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50468733
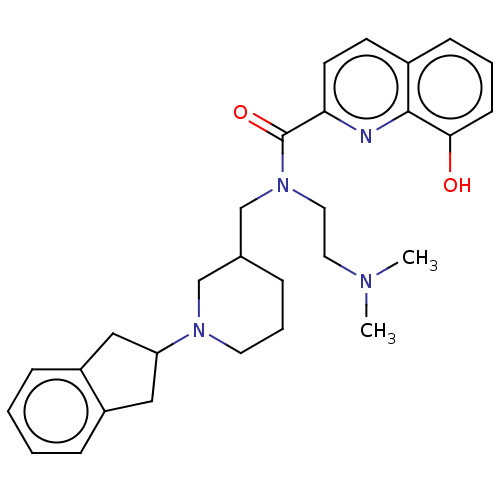
(CHEMBL4294570)Show SMILES CN(C)CCN(CC1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2cccc(O)c2n1 Show InChI InChI=1S/C29H36N4O2/c1-31(2)15-16-33(29(35)26-13-12-22-10-5-11-27(34)28(22)30-26)20-21-7-6-14-32(19-21)25-17-23-8-3-4-9-24(23)18-25/h3-5,8-13,21,25,34H,6-7,14-20H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Competitive inhibition of human BChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition and measured... |
Eur J Med Chem 156: 598-617 (2018)
Article DOI: 10.1016/j.ejmech.2018.07.033
BindingDB Entry DOI: 10.7270/Q27W6FW2 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50468736
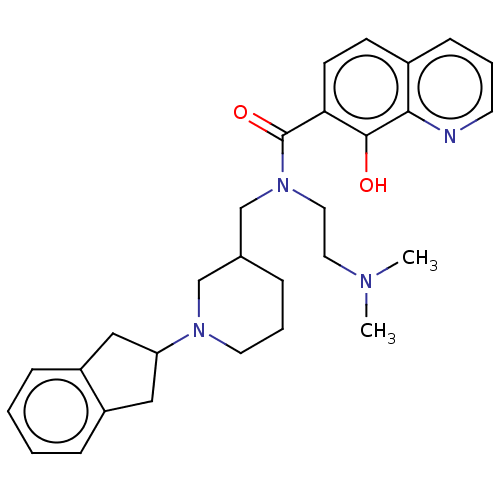
(CHEMBL4283585)Show SMILES CN(C)CCN(CC1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2cccnc2c1O Show InChI InChI=1S/C29H36N4O2/c1-31(2)15-16-33(29(35)26-12-11-22-10-5-13-30-27(22)28(26)34)20-21-7-6-14-32(19-21)25-17-23-8-3-4-9-24(23)18-25/h3-5,8-13,21,25,34H,6-7,14-20H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Competitive inhibition of human BChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition and measured... |
Eur J Med Chem 156: 598-617 (2018)
Article DOI: 10.1016/j.ejmech.2018.07.033
BindingDB Entry DOI: 10.7270/Q27W6FW2 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50027375

(CHEMBL3338394)Show SMILES COCCN(C[C@@H]1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2ccccc2c1 |r| Show InChI InChI=1S/C29H34N2O2/c1-33-16-15-31(29(32)27-13-12-23-8-2-3-9-24(23)17-27)21-22-7-6-14-30(20-22)28-18-25-10-4-5-11-26(25)19-28/h2-5,8-13,17,22,28H,6-7,14-16,18-21H2,1H3/t22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human BChE at 50 nM by stopped flow apparatus method |
J Med Chem 57: 8167-79 (2014)
Article DOI: 10.1021/jm501195e
BindingDB Entry DOI: 10.7270/Q22V2HQ7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8960

((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...)Show SMILES COc1cc2CC(CC3CCN(Cc4ccccc4)CC3)C(=O)c2cc1OC Show InChI InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bari Aldo Moro
Curated by ChEMBL
| Assay Description
Mixed type inhibition of human AChE using acetylthiocholineiodide as substrate measured for every 30 sec for 5 mins by Ellman's method |
ACS Med Chem Lett 11: 869-876 (2020)
Article DOI: 10.1021/acsmedchemlett.9b00656
BindingDB Entry DOI: 10.7270/Q2WS8XS4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50540764

(CHEMBL4648322)Show SMILES COc1cc2cc(NC(=O)[C@@H]3CCC[C@@H](C3)NCc3ccccc3)c(=O)oc2cc1OC |r| Show InChI InChI=1S/C25H28N2O5/c1-30-22-13-18-12-20(25(29)32-21(18)14-23(22)31-2)27-24(28)17-9-6-10-19(11-17)26-15-16-7-4-3-5-8-16/h3-5,7-8,12-14,17,19,26H,6,9-11,15H2,1-2H3,(H,27,28)/t17-,19+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bari Aldo Moro
Curated by ChEMBL
| Assay Description
Mixed type inhibition of human AChE using acetylthiocholineiodide as substrate measured for every 30 sec for 5 mins by Ellman's method |
ACS Med Chem Lett 11: 869-876 (2020)
Article DOI: 10.1021/acsmedchemlett.9b00656
BindingDB Entry DOI: 10.7270/Q2WS8XS4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50027377
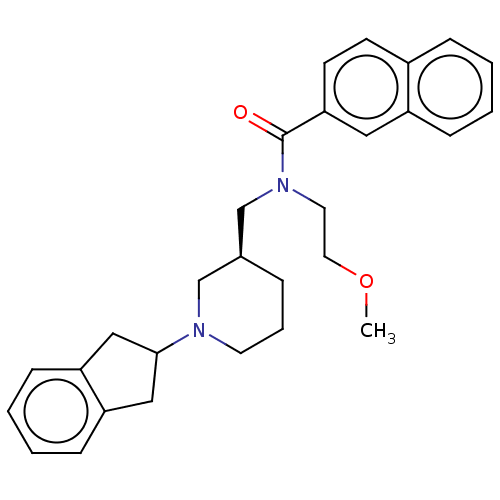
(CHEMBL3338395)Show SMILES COCCN(C[C@H]1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2ccccc2c1 |r| Show InChI InChI=1S/C29H34N2O2/c1-33-16-15-31(29(32)27-13-12-23-8-2-3-9-24(23)17-27)21-22-7-6-14-30(20-22)28-18-25-10-4-5-11-26(25)19-28/h2-5,8-13,17,22,28H,6-7,14-16,18-21H2,1H3/t22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human BChE at 50 nM by stopped flow apparatus method |
J Med Chem 57: 8167-79 (2014)
Article DOI: 10.1021/jm501195e
BindingDB Entry DOI: 10.7270/Q22V2HQ7 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50424045
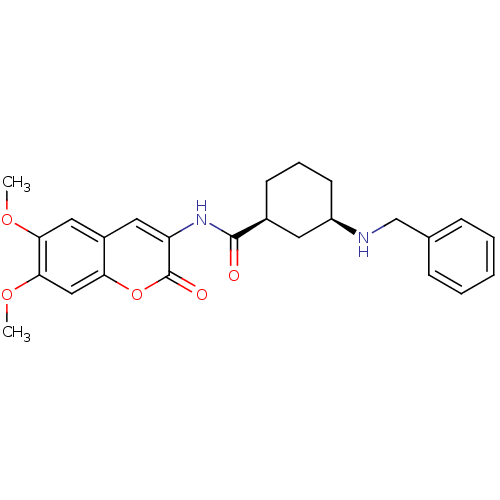
(CHEMBL2314726)Show SMILES COc1cc2cc(NC(=O)[C@H]3CCC[C@H](C3)NCc3ccccc3)c(=O)oc2cc1OC |r| Show InChI InChI=1S/C25H28N2O5/c1-30-22-13-18-12-20(25(29)32-21(18)14-23(22)31-2)27-24(28)17-9-6-10-19(11-17)26-15-16-7-4-3-5-8-16/h3-5,7-8,12-14,17,19,26H,6,9-11,15H2,1-2H3,(H,27,28)/t17-,19+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 93 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bari Aldo Moro
Curated by ChEMBL
| Assay Description
Mixed type inhibition of human AChE using acetylthiocholineiodide as substrate measured for every 30 sec for 5 mins by Ellman's method |
ACS Med Chem Lett 11: 869-876 (2020)
Article DOI: 10.1021/acsmedchemlett.9b00656
BindingDB Entry DOI: 10.7270/Q2WS8XS4 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50424045

(CHEMBL2314726)Show SMILES COc1cc2cc(NC(=O)[C@H]3CCC[C@H](C3)NCc3ccccc3)c(=O)oc2cc1OC |r| Show InChI InChI=1S/C25H28N2O5/c1-30-22-13-18-12-20(25(29)32-21(18)14-23(22)31-2)27-24(28)17-9-6-10-19(11-17)26-15-16-7-4-3-5-8-16/h3-5,7-8,12-14,17,19,26H,6,9-11,15H2,1-2H3,(H,27,28)/t17-,19+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 93.4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bari Aldo Moro
Curated by ChEMBL
| Assay Description
Mixed type inhibition of human AChE using acetylthiocholineiodide as substrate measured for every 30 sec for 5 mins by Ellman's method |
ACS Med Chem Lett 11: 869-876 (2020)
Article DOI: 10.1021/acsmedchemlett.9b00656
BindingDB Entry DOI: 10.7270/Q2WS8XS4 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50556952

(CHEMBL4754487)Show SMILES COc1cc(CNC(=O)CCCCCCNc2c3C4CC(Cc3nc3cc(Cl)ccc23)C=C(C)C4)ccc1O |t:34,TLB:23:22:19:34.31.32,THB:16:17:19:34.31.32| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 0.770 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01775
BindingDB Entry DOI: 10.7270/Q2K077XJ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50523281

(CHEMBL4583650)Show SMILES O=C(Nc1ccc(cn1)-c1cn(CCNc2c3CCCCc3nc3ccccc23)nn1)Nc1cccc2C(=O)N3CCCCC3c12 Show InChI InChI=1S/C35H35N9O2/c45-34-25-10-7-13-28(32(25)30-14-5-6-18-44(30)34)39-35(46)40-31-16-15-22(20-37-31)29-21-43(42-41-29)19-17-36-33-23-8-1-3-11-26(23)38-27-12-4-2-9-24(27)33/h1,3,7-8,10-11,13,15-16,20-21,30H,2,4-6,9,12,14,17-19H2,(H,36,38)(H2,37,39,40,46) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant AChE using ATCh as substrate preincubated for 20 mins followed by substrate addition and measured for 3 mins by Ellma... |
Eur J Med Chem 168: 58-77 (2019)
Article DOI: 10.1016/j.ejmech.2018.12.063
BindingDB Entry DOI: 10.7270/Q2J969S0 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50247022

(CHEMBL4089230)Show SMILES CN(C)CCN(CC1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2ccccc2c1 Show InChI InChI=1S/C30H37N3O/c1-31(2)16-17-33(30(34)28-14-13-24-9-3-4-10-25(24)18-28)22-23-8-7-15-32(21-23)29-19-26-11-5-6-12-27(26)20-29/h3-6,9-14,18,23,29H,7-8,15-17,19-22H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 300 secs followed by substrate addition measured f... |
J Med Chem 61: 119-139 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01086
BindingDB Entry DOI: 10.7270/Q2S75JRJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50556958
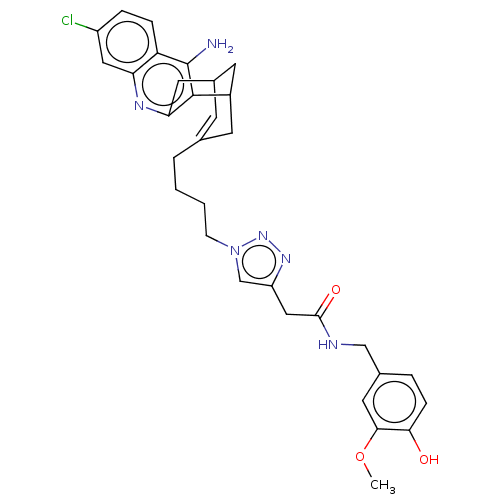
(CHEMBL4744528)Show SMILES COc1cc(CNC(=O)Cc2cn(CCCCC3=CC4CC(C3)c3c(N)c5ccc(Cl)cc5nc3C4)nn2)ccc1O |t:17,TLB:33:34:20:22.18.17,THB:24:23:20:22.18.17| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01775
BindingDB Entry DOI: 10.7270/Q2K077XJ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM10592

(7-chloro-15-methyl-10-azatetracyclo[11.3.1.0^{2,11...)Show InChI InChI=1S/C17H17ClN2/c1-9-4-10-6-11(5-9)16-15(7-10)20-14-8-12(18)2-3-13(14)17(16)19/h2-4,8,10-11H,5-7H2,1H3,(H2,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01775
BindingDB Entry DOI: 10.7270/Q2K077XJ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50556953

(CHEMBL4748114)Show SMILES COc1cc(CNC(=O)CCCCCCCNc2c3C4CC(Cc3nc3cc(Cl)ccc23)C=C(C)C4)ccc1O |t:35,TLB:24:23:20:35.32.33,THB:17:18:20:35.32.33| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01775
BindingDB Entry DOI: 10.7270/Q2K077XJ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50556954

(CHEMBL4782921)Show SMILES COc1cc(CNC(=O)CCCCCCCCNc2c3C4CC(Cc3nc3cc(Cl)ccc23)C=C(C)C4)ccc1O |t:36,TLB:25:24:21:36.33.34,THB:18:19:21:36.33.34| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01775
BindingDB Entry DOI: 10.7270/Q2K077XJ |
More data for this
Ligand-Target Pair | |
Amine oxidase [flavin-containing] A
(Homo sapiens (Human)) | BDBM15581
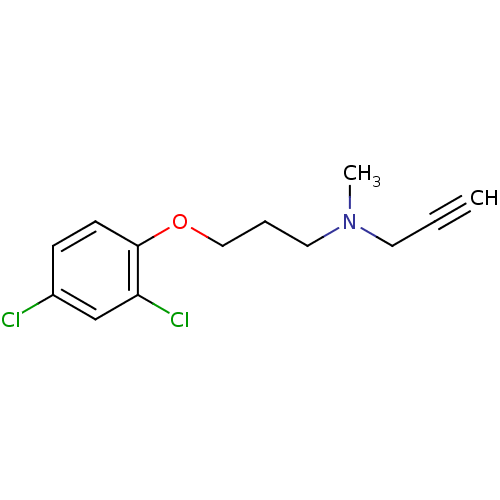
(CHEMBL8706 | CLG | CLORGILINE | Clorgyline | N-[3-...)Show InChI InChI=1S/C13H15Cl2NO/c1-3-7-16(2)8-4-9-17-13-6-5-11(14)10-12(13)15/h1,5-6,10H,4,7-9H2,2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 3.40 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant microsomal MAO-A expressed in baculovirus-infected insect cells using p-tyramine as substrate preincubated for 15 min... |
Bioorg Med Chem 25: 633-645 (2017)
Article DOI: 10.1016/j.bmc.2016.11.032
BindingDB Entry DOI: 10.7270/Q2HH6N2G |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50556951
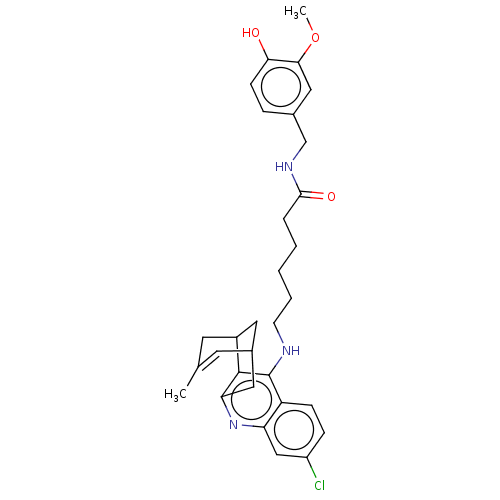
(CHEMBL4788434)Show SMILES COc1cc(CNC(=O)CCCCCNc2c3C4CC(Cc3nc3cc(Cl)ccc23)C=C(C)C4)ccc1O |t:33,TLB:22:21:18:33.30.31,THB:15:16:18:33.30.31| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 4.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01775
BindingDB Entry DOI: 10.7270/Q2K077XJ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50556956
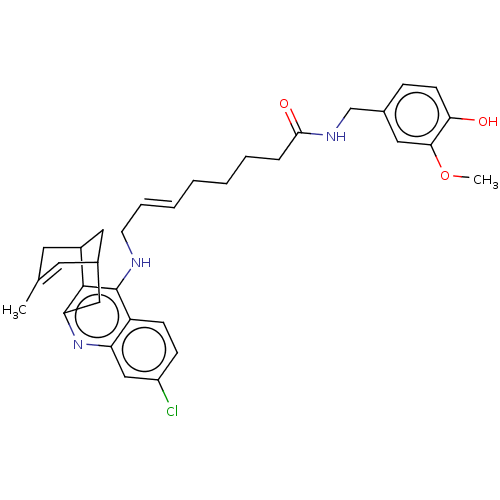
(CHEMBL4741162)Show SMILES COc1cc(CNC(=O)CCCC\C=C\CNc2c3C4CC(Cc3nc3cc(Cl)ccc23)C=C(C)C4)ccc1O |t:35,TLB:24:23:20:35.32.33,THB:17:18:20:35.32.33| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01775
BindingDB Entry DOI: 10.7270/Q2K077XJ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50431886

(CHEMBL1413473)Show InChI InChI=1S/C13H15N3O2S/c1-9(17)14-12-15-16(10(2)18)13(3,19-12)11-7-5-4-6-8-11/h4-8H,1-3H3,(H,14,15,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.90 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BChE using butyrylthiocholine as substrate preincubated for 300 secs followed by substrate addition and measured for ... |
Eur J Med Chem 156: 598-617 (2018)
Article DOI: 10.1016/j.ejmech.2018.07.033
BindingDB Entry DOI: 10.7270/Q27W6FW2 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50246979
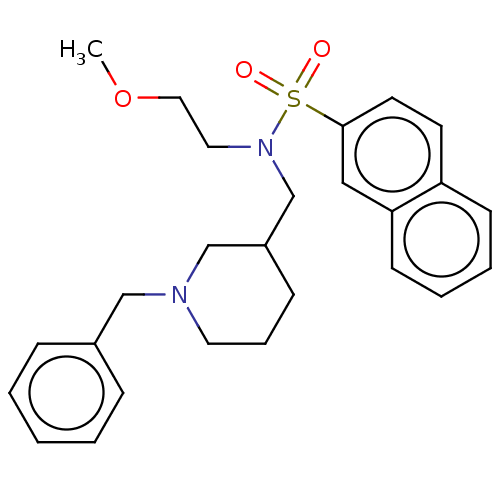
(CHEMBL4105611 | US20230331674, Table 1.1)Show SMILES COCCN(CC1CCCN(Cc2ccccc2)C1)S(=O)(=O)c1ccc2ccccc2c1 Show InChI InChI=1S/C26H32N2O3S/c1-31-17-16-28(32(29,30)26-14-13-24-11-5-6-12-25(24)18-26)21-23-10-7-15-27(20-23)19-22-8-3-2-4-9-22/h2-6,8-9,11-14,18,23H,7,10,15-17,19-21H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 4.90 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 300 secs followed by substrate addition measured f... |
J Med Chem 61: 119-139 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01086
BindingDB Entry DOI: 10.7270/Q2S75JRJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50556950
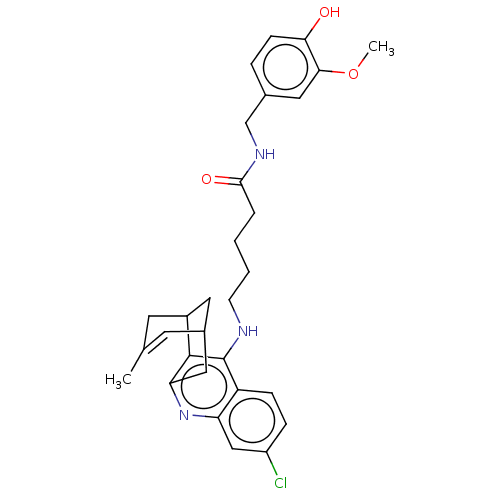
(CHEMBL4743210)Show SMILES COc1cc(CNC(=O)CCCCNc2c3C4CC(Cc3nc3cc(Cl)ccc23)C=C(C)C4)ccc1O |t:32,TLB:21:20:17:32.29.30,THB:14:15:17:32.29.30| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01775
BindingDB Entry DOI: 10.7270/Q2K077XJ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50556957

(CHEMBL4794769)Show SMILES COc1cc(CNC(=O)CCCC\C=C\CCCNc2c3C4CC(Cc3nc3cc(Cl)ccc23)C=C(C)C4)ccc1O |t:37,TLB:26:25:22:37.34.35,THB:19:20:22:37.34.35| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.60 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human AChE using acetylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's m... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01775
BindingDB Entry DOI: 10.7270/Q2K077XJ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Mus musculus (mouse)) | BDBM8960

((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...)Show SMILES COc1cc2CC(CC3CCN(Cc4ccccc4)CC3)C(=O)c2cc1OC Show InChI InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 5.70 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of AChE in mouse brain homogenate using acetylthiocholine as substrate preincubated for 300 secs followed by substrate addition by Ellman'... |
J Med Chem 61: 119-139 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01086
BindingDB Entry DOI: 10.7270/Q2S75JRJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glycogen synthase kinase-3 beta/[Tau protein] kinase
(Sus scrofa) | BDBM50523276

(CHEMBL4443989)Show SMILES [H][C@]12CCCCN1C(=O)c1cccc(NC(=O)Nc3cc(ccn3)-c3cn(CCNc4c5CCCCc5nc5ccccc45)nn3)c21 |r| Show InChI InChI=1S/C35H35N9O2/c45-34-25-10-7-13-28(32(25)30-14-5-6-18-44(30)34)39-35(46)40-31-20-22(15-16-36-31)29-21-43(42-41-29)19-17-37-33-23-8-1-3-11-26(23)38-27-12-4-2-9-24(27)33/h1,3,7-8,10-11,13,15-16,20-21,30H,2,4-6,9,12,14,17-19H2,(H,37,38)(H2,36,39,40,46)/t30-/m1/s1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS
Curated by ChEMBL
| Assay Description
Inhibition of native porcine brain GSK-3alpha/beta using GS-1 as substrate incubated for 30 mins in presence of [gamma-33P]ATP by liquid scintillatio... |
Eur J Med Chem 168: 58-77 (2019)
Article DOI: 10.1016/j.ejmech.2018.12.063
BindingDB Entry DOI: 10.7270/Q2J969S0 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50556958

(CHEMBL4744528)Show SMILES COc1cc(CNC(=O)Cc2cn(CCCCC3=CC4CC(C3)c3c(N)c5ccc(Cl)cc5nc3C4)nn2)ccc1O |t:17,TLB:33:34:20:22.18.17,THB:24:23:20:22.18.17| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human serum BChE using butyrylthiocholineiodide as substrate preincubated for 20 mins followed by substrate addition by Ellman's method |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01775
BindingDB Entry DOI: 10.7270/Q2K077XJ |
More data for this
Ligand-Target Pair | |
Glycogen synthase kinase-3 beta/[Tau protein] kinase
(Sus scrofa) | BDBM50523265

(CHEMBL4531222)Show SMILES O=C(Nc1cc(COCc2cn(CCCc3ccccc3)nn2)ccn1)Nc1cccc2C(=O)N3CCCCC3c12 Show InChI InChI=1S/C31H33N7O3/c39-30-25-11-6-12-26(29(25)27-13-4-5-17-38(27)30)33-31(40)34-28-18-23(14-15-32-28)20-41-21-24-19-37(36-35-24)16-7-10-22-8-2-1-3-9-22/h1-3,6,8-9,11-12,14-15,18-19,27H,4-5,7,10,13,16-17,20-21H2,(H2,32,33,34,40) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS
Curated by ChEMBL
| Assay Description
Inhibition of native porcine brain GSK-3alpha/beta using GS-1 as substrate incubated for 30 mins in presence of [gamma-33P]ATP by liquid scintillatio... |
Eur J Med Chem 168: 58-77 (2019)
Article DOI: 10.1016/j.ejmech.2018.12.063
BindingDB Entry DOI: 10.7270/Q2J969S0 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50523276

(CHEMBL4443989)Show SMILES [H][C@]12CCCCN1C(=O)c1cccc(NC(=O)Nc3cc(ccn3)-c3cn(CCNc4c5CCCCc5nc5ccccc45)nn3)c21 |r| Show InChI InChI=1S/C35H35N9O2/c45-34-25-10-7-13-28(32(25)30-14-5-6-18-44(30)34)39-35(46)40-31-20-22(15-16-36-31)29-21-43(42-41-29)19-17-37-33-23-8-1-3-11-26(23)38-27-12-4-2-9-24(27)33/h1,3,7-8,10-11,13,15-16,20-21,30H,2,4-6,9,12,14,17-19H2,(H,37,38)(H2,36,39,40,46)/t30-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9.5 | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant AChE using ATCh as substrate preincubated for 20 mins followed by substrate addition and measured for 3 mins by Ellma... |
Eur J Med Chem 168: 58-77 (2019)
Article DOI: 10.1016/j.ejmech.2018.12.063
BindingDB Entry DOI: 10.7270/Q2J969S0 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50468744
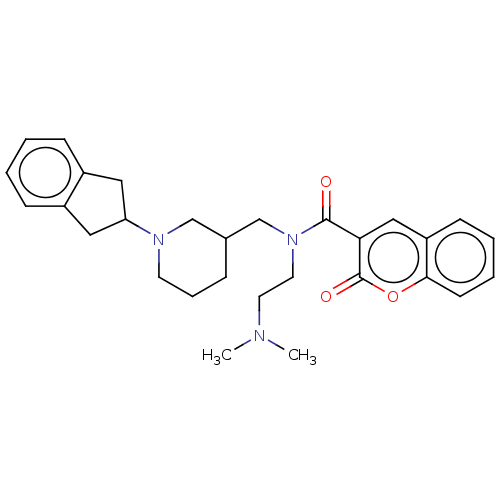
(CHEMBL4280947)Show SMILES CN(C)CCN(CC1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1cc2ccccc2oc1=O Show InChI InChI=1S/C29H35N3O3/c1-30(2)14-15-32(28(33)26-18-24-11-5-6-12-27(24)35-29(26)34)20-21-8-7-13-31(19-21)25-16-22-9-3-4-10-23(22)17-25/h3-6,9-12,18,21,25H,7-8,13-17,19-20H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9.5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of human BChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition and measured for 1 min b... |
Eur J Med Chem 156: 598-617 (2018)
Article DOI: 10.1016/j.ejmech.2018.07.033
BindingDB Entry DOI: 10.7270/Q27W6FW2 |
More data for this
Ligand-Target Pair | |
Glycogen synthase kinase-3 beta/[Tau protein] kinase
(Sus scrofa) | BDBM50523269

(CHEMBL4446287)Show SMILES CN(Cc1cn(CCNc2c3CCCCc3nc3ccccc23)nn1)Cc1ccnc(NC(=O)Nc2cccc3C(=O)N4CCCCC4c23)c1 Show InChI InChI=1S/C38H42N10O2/c1-46(23-26-24-47(45-44-26)20-18-40-36-27-9-2-4-12-30(27)41-31-13-5-3-10-28(31)36)22-25-16-17-39-34(21-25)43-38(50)42-32-14-8-11-29-35(32)33-15-6-7-19-48(33)37(29)49/h2,4,8-9,11-12,14,16-17,21,24,33H,3,5-7,10,13,15,18-20,22-23H2,1H3,(H,40,41)(H2,39,42,43,50) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS
Curated by ChEMBL
| Assay Description
Inhibition of native porcine brain GSK-3alpha/beta using GS-1 as substrate incubated for 30 mins in presence of [gamma-33P]ATP by liquid scintillatio... |
Eur J Med Chem 168: 58-77 (2019)
Article DOI: 10.1016/j.ejmech.2018.12.063
BindingDB Entry DOI: 10.7270/Q2J969S0 |
More data for this
Ligand-Target Pair | |
Glycogen synthase kinase-3 beta/[Tau protein] kinase
(Sus scrofa) | BDBM50523277
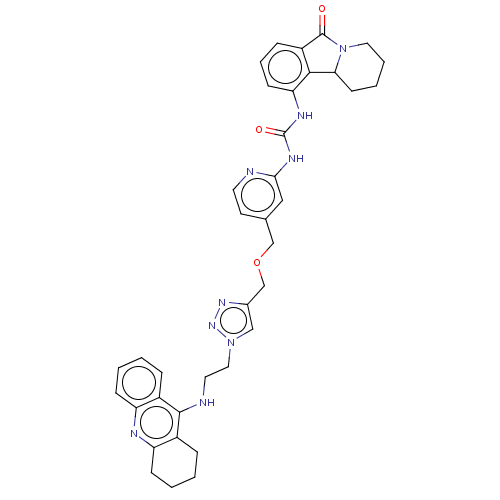
(CHEMBL4516356)Show SMILES O=C(Nc1cc(COCc2cn(CCNc3c4CCCCc4nc4ccccc34)nn2)ccn1)Nc1cccc2C(=O)N3CCCCC3c12 Show InChI InChI=1S/C37H39N9O3/c47-36-28-10-7-13-31(34(28)32-14-5-6-18-46(32)36)41-37(48)42-33-20-24(15-16-38-33)22-49-23-25-21-45(44-43-25)19-17-39-35-26-8-1-3-11-29(26)40-30-12-4-2-9-27(30)35/h1,3,7-8,10-11,13,15-16,20-21,32H,2,4-6,9,12,14,17-19,22-23H2,(H,39,40)(H2,38,41,42,48) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS
Curated by ChEMBL
| Assay Description
Inhibition of native porcine brain GSK-3alpha/beta using GS-1 as substrate incubated for 30 mins in presence of [gamma-33P]ATP by liquid scintillatio... |
Eur J Med Chem 168: 58-77 (2019)
Article DOI: 10.1016/j.ejmech.2018.12.063
BindingDB Entry DOI: 10.7270/Q2J969S0 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50468733

(CHEMBL4294570)Show SMILES CN(C)CCN(CC1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2cccc(O)c2n1 Show InChI InChI=1S/C29H36N4O2/c1-31(2)15-16-33(29(35)26-13-12-22-10-5-11-27(34)28(22)30-26)20-21-7-6-14-32(19-21)25-17-23-8-3-4-9-24(23)18-25/h3-5,8-13,21,25,34H,6-7,14-20H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of human BChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition and measured for 1 min b... |
Eur J Med Chem 156: 598-617 (2018)
Article DOI: 10.1016/j.ejmech.2018.07.033
BindingDB Entry DOI: 10.7270/Q27W6FW2 |
More data for this
Ligand-Target Pair | |
Glycogen synthase kinase-3 beta/[Tau protein] kinase
(Sus scrofa) | BDBM50523266

(CHEMBL4444585)Show SMILES CN(CC#C)Cc1ccnc(NC(=O)Nc2cccc3C(=O)N4CCCCC4c23)c1 Show InChI InChI=1S/C23H25N5O2/c1-3-12-27(2)15-16-10-11-24-20(14-16)26-23(30)25-18-8-6-7-17-21(18)19-9-4-5-13-28(19)22(17)29/h1,6-8,10-11,14,19H,4-5,9,12-13,15H2,2H3,(H2,24,25,26,30) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS
Curated by ChEMBL
| Assay Description
Inhibition of native porcine brain GSK-3alpha/beta using GS-1 as substrate incubated for 30 mins in presence of [gamma-33P]ATP by liquid scintillatio... |
Eur J Med Chem 168: 58-77 (2019)
Article DOI: 10.1016/j.ejmech.2018.12.063
BindingDB Entry DOI: 10.7270/Q2J969S0 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50523267

(CHEMBL4559936)Show SMILES O=C(Nc1cc(ccn1)-c1cn(CCNc2c3CCCCc3nc3ccccc23)nn1)Nc1cccc2C(=O)N3CCCCC3c12 Show InChI InChI=1S/C35H35N9O2/c45-34-25-10-7-13-28(32(25)30-14-5-6-18-44(30)34)39-35(46)40-31-20-22(15-16-36-31)29-21-43(42-41-29)19-17-37-33-23-8-1-3-11-26(23)38-27-12-4-2-9-24(27)33/h1,3,7-8,10-11,13,15-16,20-21,30H,2,4-6,9,12,14,17-19H2,(H,37,38)(H2,36,39,40,46) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant AChE using ATCh as substrate preincubated for 20 mins followed by substrate addition and measured for 3 mins by Ellma... |
Eur J Med Chem 168: 58-77 (2019)
Article DOI: 10.1016/j.ejmech.2018.12.063
BindingDB Entry DOI: 10.7270/Q2J969S0 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM8961

(1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...)Show InChI InChI=1S/C13H14N2/c14-13-9-5-1-3-7-11(9)15-12-8-4-2-6-10(12)13/h1,3,5,7H,2,4,6,8H2,(H2,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human BChE by Ellman method |
J Med Chem 57: 8167-79 (2014)
Article DOI: 10.1021/jm501195e
BindingDB Entry DOI: 10.7270/Q22V2HQ7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cholinesterase
(Homo sapiens (Human)) | BDBM8961

(1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...)Show InChI InChI=1S/C13H14N2/c14-13-9-5-1-3-7-11(9)15-12-8-4-2-6-10(12)13/h1,3,5,7H,2,4,6,8H2,(H2,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BChE using butyrylthiocholine iodide as substrate after 5 mins by Ellman's method |
Bioorg Med Chem 23: 4442-52 (2015)
Article DOI: 10.1016/j.bmc.2015.06.010
BindingDB Entry DOI: 10.7270/Q2W097PZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cholinesterase
(Homo sapiens (Human)) | BDBM50027375

(CHEMBL3338394)Show SMILES COCCN(C[C@@H]1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2ccccc2c1 |r| Show InChI InChI=1S/C29H34N2O2/c1-33-16-15-31(29(32)27-13-12-23-8-2-3-9-24(23)17-27)21-22-7-6-14-30(20-22)28-18-25-10-4-5-11-26(25)19-28/h2-5,8-13,17,22,28H,6-7,14-16,18-21H2,1H3/t22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human BChE by Ellman method |
J Med Chem 57: 8167-79 (2014)
Article DOI: 10.1021/jm501195e
BindingDB Entry DOI: 10.7270/Q22V2HQ7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cholinesterase
(Homo sapiens (Human)) | BDBM50468736

(CHEMBL4283585)Show SMILES CN(C)CCN(CC1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2cccnc2c1O Show InChI InChI=1S/C29H36N4O2/c1-31(2)15-16-33(29(35)26-12-11-22-10-5-13-30-27(22)28(26)34)20-21-7-6-14-32(19-21)25-17-23-8-3-4-9-24(23)18-25/h3-5,8-13,21,25,34H,6-7,14-20H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of human BChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition and measured for 1 min b... |
Eur J Med Chem 156: 598-617 (2018)
Article DOI: 10.1016/j.ejmech.2018.07.033
BindingDB Entry DOI: 10.7270/Q27W6FW2 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50523262

(CHEMBL4443468)Show SMILES [H][C@@]12CCCCN1C(=O)c1cccc(NC(=O)Nc3cc(ccn3)-c3cn(CCNc4c5CCCCc5nc5ccccc45)nn3)c21 |r| Show InChI InChI=1S/C35H35N9O2/c45-34-25-10-7-13-28(32(25)30-14-5-6-18-44(30)34)39-35(46)40-31-20-22(15-16-36-31)29-21-43(42-41-29)19-17-37-33-23-8-1-3-11-26(23)38-27-12-4-2-9-24(27)33/h1,3,7-8,10-11,13,15-16,20-21,30H,2,4-6,9,12,14,17-19H2,(H,37,38)(H2,36,39,40,46)/t30-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant AChE using ATCh as substrate preincubated for 20 mins followed by substrate addition and measured for 3 mins by Ellma... |
Eur J Med Chem 168: 58-77 (2019)
Article DOI: 10.1016/j.ejmech.2018.12.063
BindingDB Entry DOI: 10.7270/Q2J969S0 |
More data for this
Ligand-Target Pair | |
Glycogen synthase kinase-3 beta/[Tau protein] kinase
(Sus scrofa) | BDBM50523282

(CHEMBL4515295)Show SMILES O=C(Nc1cc(CCC#C)ccn1)Nc1cccc2C(=O)N3CCCCC3c12 Show InChI InChI=1S/C22H22N4O2/c1-2-3-7-15-11-12-23-19(14-15)25-22(28)24-17-9-6-8-16-20(17)18-10-4-5-13-26(18)21(16)27/h1,6,8-9,11-12,14,18H,3-5,7,10,13H2,(H2,23,24,25,28) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS
Curated by ChEMBL
| Assay Description
Inhibition of native porcine brain GSK-3alpha/beta using GS-1 as substrate incubated for 30 mins in presence of [gamma-33P]ATP by liquid scintillatio... |
Eur J Med Chem 168: 58-77 (2019)
Article DOI: 10.1016/j.ejmech.2018.12.063
BindingDB Entry DOI: 10.7270/Q2J969S0 |
More data for this
Ligand-Target Pair | |
Glycogen synthase kinase-3 beta/[Tau protein] kinase
(Sus scrofa) | BDBM50523267

(CHEMBL4559936)Show SMILES O=C(Nc1cc(ccn1)-c1cn(CCNc2c3CCCCc3nc3ccccc23)nn1)Nc1cccc2C(=O)N3CCCCC3c12 Show InChI InChI=1S/C35H35N9O2/c45-34-25-10-7-13-28(32(25)30-14-5-6-18-44(30)34)39-35(46)40-31-20-22(15-16-36-31)29-21-43(42-41-29)19-17-37-33-23-8-1-3-11-26(23)38-27-12-4-2-9-24(27)33/h1,3,7-8,10-11,13,15-16,20-21,30H,2,4-6,9,12,14,17-19H2,(H,37,38)(H2,36,39,40,46) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS
Curated by ChEMBL
| Assay Description
Inhibition of native porcine brain GSK-3alpha/beta using GS-1 as substrate incubated for 30 mins in presence of [gamma-33P]ATP by liquid scintillatio... |
Eur J Med Chem 168: 58-77 (2019)
Article DOI: 10.1016/j.ejmech.2018.12.063
BindingDB Entry DOI: 10.7270/Q2J969S0 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8960

((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...)Show SMILES COc1cc2CC(CC3CCN(Cc4ccccc4)CC3)C(=O)c2cc1OC Show InChI InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bari Aldo Moro
Curated by ChEMBL
| Assay Description
Inhibition of human AChE using acetylthiocholineiodide as substrate measured for every 30 sec for 5 mins by Ellman's method |
ACS Med Chem Lett 11: 869-876 (2020)
Article DOI: 10.1021/acsmedchemlett.9b00656
BindingDB Entry DOI: 10.7270/Q2WS8XS4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cholinesterase
(Homo sapiens (Human)) | BDBM50247010

(CHEMBL4068039)Show SMILES Cl.Cl.CNCCN(CC1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2ccccc2c1 Show InChI InChI=1S/C29H35N3O.2ClH/c1-30-14-16-32(29(33)27-13-12-23-8-2-3-9-24(23)17-27)21-22-7-6-15-31(20-22)28-18-25-10-4-5-11-26(25)19-28;;/h2-5,8-13,17,22,28,30H,6-7,14-16,18-21H2,1H3;2*1H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate preincubated for 300 secs followed by substrate addition measured f... |
J Med Chem 61: 119-139 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01086
BindingDB Entry DOI: 10.7270/Q2S75JRJ |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 5 activator 1
(Homo sapiens (Human)) | BDBM50523278
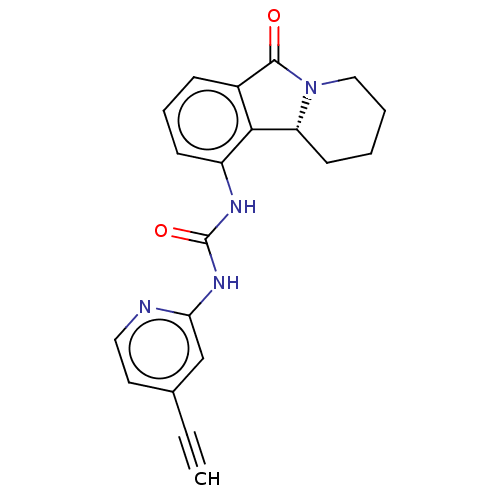
(CHEMBL4463060)Show SMILES [H][C@]12CCCCN1C(=O)c1cccc(NC(=O)Nc3cc(ccn3)C#C)c21 |r| Show InChI InChI=1S/C20H18N4O2/c1-2-13-9-10-21-17(12-13)23-20(26)22-15-7-5-6-14-18(15)16-8-3-4-11-24(16)19(14)25/h1,5-7,9-10,12,16H,3-4,8,11H2,(H2,21,22,23,26)/t16-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CDK5/p25 using histone H1 as substrate in presence of [gamma-33P]ATP incubated for 30 mins by liquid scintillation co... |
Eur J Med Chem 168: 58-77 (2019)
Article DOI: 10.1016/j.ejmech.2018.12.063
BindingDB Entry DOI: 10.7270/Q2J969S0 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50468741

(CHEMBL4291165)Show SMILES CN(C)CCN(CC1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1cc(O)c2cccc(O)c2n1 Show InChI InChI=1S/C29H36N4O3/c1-31(2)13-14-33(29(36)25-17-27(35)24-10-5-11-26(34)28(24)30-25)19-20-7-6-12-32(18-20)23-15-21-8-3-4-9-22(21)16-23/h3-5,8-11,17,20,23,34H,6-7,12-16,18-19H2,1-2H3,(H,30,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of human BChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition and measured for 1 min b... |
Eur J Med Chem 156: 598-617 (2018)
Article DOI: 10.1016/j.ejmech.2018.07.033
BindingDB Entry DOI: 10.7270/Q27W6FW2 |
More data for this
Ligand-Target Pair | |
Glycogen synthase kinase-3 beta/[Tau protein] kinase
(Sus scrofa) | BDBM50523262

(CHEMBL4443468)Show SMILES [H][C@@]12CCCCN1C(=O)c1cccc(NC(=O)Nc3cc(ccn3)-c3cn(CCNc4c5CCCCc5nc5ccccc45)nn3)c21 |r| Show InChI InChI=1S/C35H35N9O2/c45-34-25-10-7-13-28(32(25)30-14-5-6-18-44(30)34)39-35(46)40-31-20-22(15-16-36-31)29-21-43(42-41-29)19-17-37-33-23-8-1-3-11-26(23)38-27-12-4-2-9-24(27)33/h1,3,7-8,10-11,13,15-16,20-21,30H,2,4-6,9,12,14,17-19H2,(H,37,38)(H2,36,39,40,46)/t30-/m0/s1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS
Curated by ChEMBL
| Assay Description
Inhibition of native porcine brain GSK-3alpha/beta using GS-1 as substrate incubated for 30 mins in presence of [gamma-33P]ATP by liquid scintillatio... |
Eur J Med Chem 168: 58-77 (2019)
Article DOI: 10.1016/j.ejmech.2018.12.063
BindingDB Entry DOI: 10.7270/Q2J969S0 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data