 Found 262 hits with Last Name = 'ganesh' and Initial = 't'
Found 262 hits with Last Name = 'ganesh' and Initial = 't' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM50016952
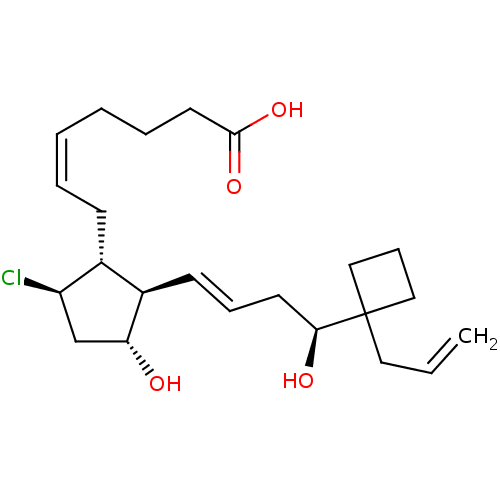
(CHEMBL3286796)Show SMILES O[C@@H](C\C=C\[C@H]1[C@H](O)C[C@@H](Cl)[C@@H]1C\C=C/CCCC(O)=O)C1(CC=C)CCC1 |r| Show InChI InChI=1S/C23H35ClO4/c1-2-13-23(14-8-15-23)21(26)11-7-10-18-17(19(24)16-20(18)25)9-5-3-4-6-12-22(27)28/h2-3,5,7,10,17-21,25-26H,1,4,6,8-9,11-16H2,(H,27,28)/b5-3-,10-7+/t17-,18-,19-,20-,21+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP2 receptor (unknown origin) by competitive binding assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP4 subtype
(Homo sapiens (Human)) | BDBM35847

((15S)-prostaglandin E2 | (5Z,11alpha,13E,15S)-11,1...)Show SMILES CCCCC[C@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C20H32O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h4,7,12-13,15-17,19,21,23H,2-3,5-6,8-11,14H2,1H3,(H,24,25)/b7-4-,13-12+/t15-,16+,17+,19+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 3.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP4 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM50101830
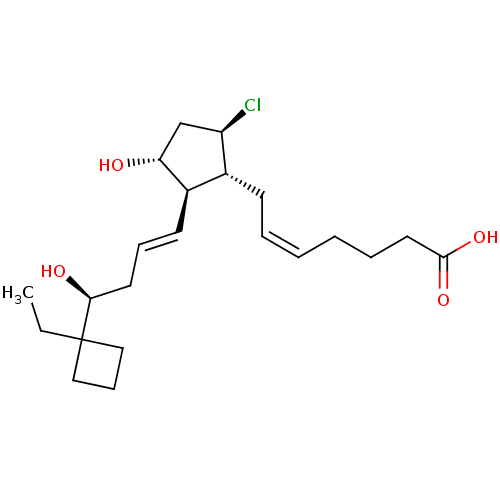
((Z)-7-{(1R,2R,3R,5R)-5-Chloro-2-[(E)-(S)-4-(1-ethy...)Show SMILES CCC1(CCC1)[C@@H](O)C\C=C\[C@H]1[C@H](O)C[C@@H](Cl)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C22H35ClO4/c1-2-22(13-8-14-22)20(25)11-7-10-17-16(18(23)15-19(17)24)9-5-3-4-6-12-21(26)27/h3,5,7,10,16-20,24-25H,2,4,6,8-9,11-15H2,1H3,(H,26,27)/b5-3-,10-7+/t16-,17-,18-,19-,20+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP2 receptor (unknown origin) by competitive binding assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(Homo sapiens (Human)) | BDBM35847

((15S)-prostaglandin E2 | (5Z,11alpha,13E,15S)-11,1...)Show SMILES CCCCC[C@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C20H32O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h4,7,12-13,15-17,19,21,23H,2-3,5-6,8-11,14H2,1H3,(H,24,25)/b7-4-,13-12+/t15-,16+,17+,19+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP3 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM50016953
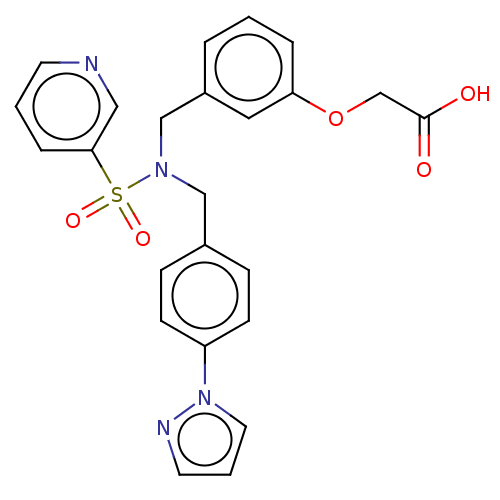
(Taprenepag)Show SMILES OC(=O)COc1cccc(CN(Cc2ccc(cc2)-n2cccn2)S(=O)(=O)c2cccnc2)c1 Show InChI InChI=1S/C24H22N4O5S/c29-24(30)18-33-22-5-1-4-20(14-22)17-27(34(31,32)23-6-2-11-25-15-23)16-19-7-9-21(10-8-19)28-13-3-12-26-28/h1-15H,16-18H2,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP2 receptor (unknown origin) by competitive binding assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E2 receptor EP1 subtype
(Homo sapiens (Human)) | BDBM35847

((15S)-prostaglandin E2 | (5Z,11alpha,13E,15S)-11,1...)Show SMILES CCCCC[C@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C20H32O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h4,7,12-13,15-17,19,21,23H,2-3,5-6,8-11,14H2,1H3,(H,24,25)/b7-4-,13-12+/t15-,16+,17+,19+/m0/s1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP1 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM35847

((15S)-prostaglandin E2 | (5Z,11alpha,13E,15S)-11,1...)Show SMILES CCCCC[C@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C20H32O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h4,7,12-13,15-17,19,21,23H,2-3,5-6,8-11,14H2,1H3,(H,24,25)/b7-4-,13-12+/t15-,16+,17+,19+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 38 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP2 receptor (unknown origin) by competitive binding assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM50293496
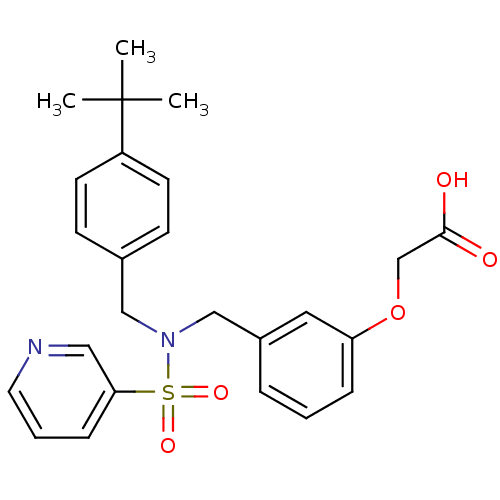
(2-(3-((N-(4-tert-butylbenzyl)pyridine-3-sulfonamid...)Show SMILES CC(C)(C)c1ccc(CN(Cc2cccc(OCC(O)=O)c2)S(=O)(=O)c2cccnc2)cc1 Show InChI InChI=1S/C25H28N2O5S/c1-25(2,3)21-11-9-19(10-12-21)16-27(33(30,31)23-8-5-13-26-15-23)17-20-6-4-7-22(14-20)32-18-24(28)29/h4-15H,16-18H2,1-3H3,(H,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
| PDB
Article
PubMed
| 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP2 receptor (unknown origin) by competitive binding assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM50016954
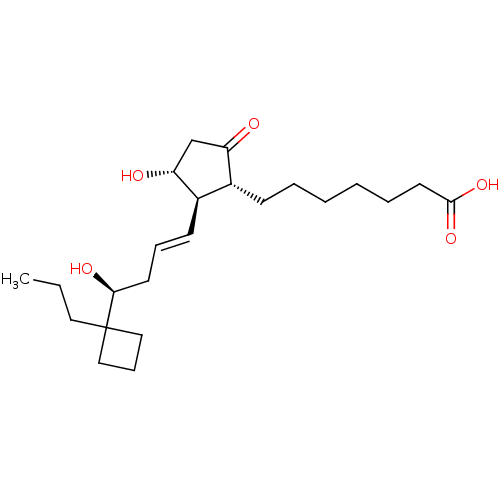
(CHEMBL1628262)Show SMILES CCCC1(CCC1)[C@@H](O)C\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C23H38O5/c1-2-13-23(14-8-15-23)21(26)11-7-10-18-17(19(24)16-20(18)25)9-5-3-4-6-12-22(27)28/h7,10,17-18,20-21,25-26H,2-6,8-9,11-16H2,1H3,(H,27,28)/b10-7+/t17-,18-,20-,21+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 73 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP2 receptor (unknown origin) by competitive binding assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(Homo sapiens (Human)) | BDBM50016981
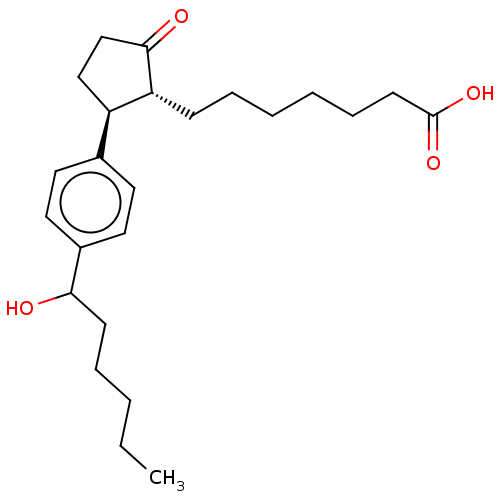
(CHEMBL1722929)Show SMILES CCCCCC(O)c1ccc(cc1)[C@H]1CCC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C24H36O4/c1-2-3-6-10-22(25)19-14-12-18(13-15-19)20-16-17-23(26)21(20)9-7-4-5-8-11-24(27)28/h12-15,20-22,25H,2-11,16-17H2,1H3,(H,27,28)/t20-,21-,22?/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 82 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP3 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM50016981

(CHEMBL1722929)Show SMILES CCCCCC(O)c1ccc(cc1)[C@H]1CCC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C24H36O4/c1-2-3-6-10-22(25)19-14-12-18(13-15-19)20-16-17-23(26)21(20)9-7-4-5-8-11-24(27)28/h12-15,20-22,25H,2-11,16-17H2,1H3,(H,27,28)/t20-,21-,22?/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP2 receptor (unknown origin) by competitive binding assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50016954

(CHEMBL1628262)Show SMILES CCCC1(CCC1)[C@@H](O)C\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C23H38O5/c1-2-13-23(14-8-15-23)21(26)11-7-10-18-17(19(24)16-20(18)25)9-5-3-4-6-12-22(27)28/h7,10,17-18,20-21,25-26H,2-6,8-9,11-16H2,1H3,(H,27,28)/b10-7+/t17-,18-,20-,21+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to prostanoid IP receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM85184

(AH-6809 | CAS_33458-93-4 | NSC_119461)Show InChI InChI=1S/C17H14O5/c1-9(2)21-11-4-5-12-15(8-11)22-14-6-3-10(17(19)20)7-13(14)16(12)18/h3-9H,1-2H3,(H,19,20) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 1.15E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of EP2 receptor (unknown origin) by competitive binding assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM85184

(AH-6809 | CAS_33458-93-4 | NSC_119461)Show InChI InChI=1S/C17H14O5/c1-9(2)21-11-4-5-12-15(8-11)22-14-6-3-10(17(19)20)7-13(14)16(12)18/h3-9H,1-2H3,(H,19,20) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 1.15E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Antagonist activity at EP2 receptor (unknown origin) by functional cAMP assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP1 subtype
(Homo sapiens (Human)) | BDBM85184

(AH-6809 | CAS_33458-93-4 | NSC_119461)Show InChI InChI=1S/C17H14O5/c1-9(2)21-11-4-5-12-15(8-11)22-14-6-3-10(17(19)20)7-13(14)16(12)18/h3-9H,1-2H3,(H,19,20) | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Antagonist activity at EP1 receptor (unknown origin) by functional cAMP assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP1 subtype
(Homo sapiens (Human)) | BDBM50016952

(CHEMBL3286796)Show SMILES O[C@@H](C\C=C\[C@H]1[C@H](O)C[C@@H](Cl)[C@@H]1C\C=C/CCCC(O)=O)C1(CC=C)CCC1 |r| Show InChI InChI=1S/C23H35ClO4/c1-2-13-23(14-8-15-23)21(26)11-7-10-18-17(19(24)16-20(18)25)9-5-3-4-6-12-22(27)28/h2-3,5,7,10,17-21,25-26H,1,4,6,8-9,11-16H2,(H,27,28)/b5-3-,10-7+/t17-,18-,19-,20-,21+/m1/s1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP1 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin D2 receptor
(Homo sapiens (Human)) | BDBM85184

(AH-6809 | CAS_33458-93-4 | NSC_119461)Show InChI InChI=1S/C17H14O5/c1-9(2)21-11-4-5-12-15(8-11)22-14-6-3-10(17(19)20)7-13(14)16(12)18/h3-9H,1-2H3,(H,19,20) | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Antagonist activity at DP1 receptor (unknown origin) by functional cAMP assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(Homo sapiens (Human)) | BDBM85184

(AH-6809 | CAS_33458-93-4 | NSC_119461)Show InChI InChI=1S/C17H14O5/c1-9(2)21-11-4-5-12-15(8-11)22-14-6-3-10(17(19)20)7-13(14)16(12)18/h3-9H,1-2H3,(H,19,20) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Antagonist activity at EP3 receptor (unknown origin) by functional cAMP assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP4 subtype
(Homo sapiens (Human)) | BDBM50016952

(CHEMBL3286796)Show SMILES O[C@@H](C\C=C\[C@H]1[C@H](O)C[C@@H](Cl)[C@@H]1C\C=C/CCCC(O)=O)C1(CC=C)CCC1 |r| Show InChI InChI=1S/C23H35ClO4/c1-2-13-23(14-8-15-23)21(26)11-7-10-18-17(19(24)16-20(18)25)9-5-3-4-6-12-22(27)28/h2-3,5,7,10,17-21,25-26H,1,4,6,8-9,11-16H2,(H,27,28)/b5-3-,10-7+/t17-,18-,19-,20-,21+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP4 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM85602
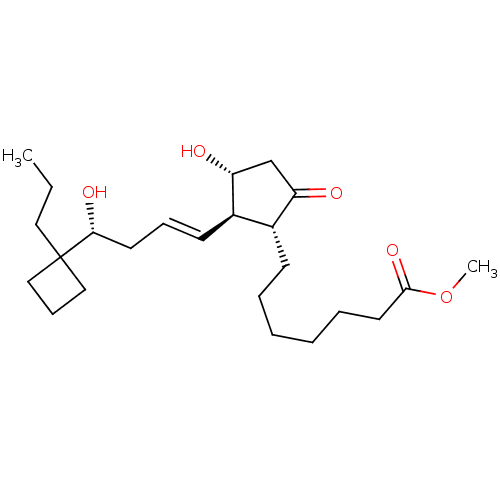
((R)-BUTAPROST | Butaprost (Free Acid) | CAS_69648-...)Show SMILES CCCC1(CCC1)[C@H](O)C\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(=O)OC |r| Show InChI InChI=1S/C24H40O5/c1-3-14-24(15-9-16-24)22(27)12-8-11-19-18(20(25)17-21(19)26)10-6-4-5-7-13-23(28)29-2/h8,11,18-19,21-22,26-27H,3-7,9-10,12-17H2,1-2H3/b11-8+/t18-,19-,21-,22-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP2 receptor (unknown origin) by competitive binding assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(Homo sapiens (Human)) | BDBM50016952

(CHEMBL3286796)Show SMILES O[C@@H](C\C=C\[C@H]1[C@H](O)C[C@@H](Cl)[C@@H]1C\C=C/CCCC(O)=O)C1(CC=C)CCC1 |r| Show InChI InChI=1S/C23H35ClO4/c1-2-13-23(14-8-15-23)21(26)11-7-10-18-17(19(24)16-20(18)25)9-5-3-4-6-12-22(27)28/h2-3,5,7,10,17-21,25-26H,1,4,6,8-9,11-16H2,(H,27,28)/b5-3-,10-7+/t17-,18-,19-,20-,21+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP3 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP1 subtype
(Homo sapiens (Human)) | BDBM50293496

(2-(3-((N-(4-tert-butylbenzyl)pyridine-3-sulfonamid...)Show SMILES CC(C)(C)c1ccc(CN(Cc2cccc(OCC(O)=O)c2)S(=O)(=O)c2cccnc2)cc1 Show InChI InChI=1S/C25H28N2O5S/c1-25(2,3)21-11-9-19(10-12-21)16-27(33(30,31)23-8-5-13-26-15-23)17-20-6-4-7-22(14-20)32-18-24(28)29/h4-15H,16-18H2,1-3H3,(H,28,29) | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
| Article
PubMed
| >2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP1 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(Homo sapiens (Human)) | BDBM50293496

(2-(3-((N-(4-tert-butylbenzyl)pyridine-3-sulfonamid...)Show SMILES CC(C)(C)c1ccc(CN(Cc2cccc(OCC(O)=O)c2)S(=O)(=O)c2cccnc2)cc1 Show InChI InChI=1S/C25H28N2O5S/c1-25(2,3)21-11-9-19(10-12-21)16-27(33(30,31)23-8-5-13-26-15-23)17-20-6-4-7-22(14-20)32-18-24(28)29/h4-15H,16-18H2,1-3H3,(H,28,29) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
| Article
PubMed
| >2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP3 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(Homo sapiens (Human)) | BDBM50016953

(Taprenepag)Show SMILES OC(=O)COc1cccc(CN(Cc2ccc(cc2)-n2cccn2)S(=O)(=O)c2cccnc2)c1 Show InChI InChI=1S/C24H22N4O5S/c29-24(30)18-33-22-5-1-4-20(14-22)17-27(34(31,32)23-6-2-11-25-15-23)16-19-7-9-21(10-8-19)28-13-3-12-26-28/h1-15H,16-18H2,(H,29,30) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| >3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP3 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP4 subtype
(Homo sapiens (Human)) | BDBM50293496

(2-(3-((N-(4-tert-butylbenzyl)pyridine-3-sulfonamid...)Show SMILES CC(C)(C)c1ccc(CN(Cc2cccc(OCC(O)=O)c2)S(=O)(=O)c2cccnc2)cc1 Show InChI InChI=1S/C25H28N2O5S/c1-25(2,3)21-11-9-19(10-12-21)16-27(33(30,31)23-8-5-13-26-15-23)17-20-6-4-7-22(14-20)32-18-24(28)29/h4-15H,16-18H2,1-3H3,(H,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
| Article
PubMed
| >3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP4 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50293496

(2-(3-((N-(4-tert-butylbenzyl)pyridine-3-sulfonamid...)Show SMILES CC(C)(C)c1ccc(CN(Cc2cccc(OCC(O)=O)c2)S(=O)(=O)c2cccnc2)cc1 Show InChI InChI=1S/C25H28N2O5S/c1-25(2,3)21-11-9-19(10-12-21)16-27(33(30,31)23-8-5-13-26-15-23)17-20-6-4-7-22(14-20)32-18-24(28)29/h4-15H,16-18H2,1-3H3,(H,28,29) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
| Article
PubMed
| >3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to prostanoid IP receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP4 subtype
(Homo sapiens (Human)) | BDBM50016953

(Taprenepag)Show SMILES OC(=O)COc1cccc(CN(Cc2ccc(cc2)-n2cccn2)S(=O)(=O)c2cccnc2)c1 Show InChI InChI=1S/C24H22N4O5S/c29-24(30)18-33-22-5-1-4-20(14-22)17-27(34(31,32)23-6-2-11-25-15-23)16-19-7-9-21(10-8-19)28-13-3-12-26-28/h1-15H,16-18H2,(H,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| >3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP4 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP1 subtype
(Homo sapiens (Human)) | BDBM50016953

(Taprenepag)Show SMILES OC(=O)COc1cccc(CN(Cc2ccc(cc2)-n2cccn2)S(=O)(=O)c2cccnc2)c1 Show InChI InChI=1S/C24H22N4O5S/c29-24(30)18-33-22-5-1-4-20(14-22)17-27(34(31,32)23-6-2-11-25-15-23)16-19-7-9-21(10-8-19)28-13-3-12-26-28/h1-15H,16-18H2,(H,29,30) | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| >3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP1 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(Homo sapiens (Human)) | BDBM50101830

((Z)-7-{(1R,2R,3R,5R)-5-Chloro-2-[(E)-(S)-4-(1-ethy...)Show SMILES CCC1(CCC1)[C@@H](O)C\C=C\[C@H]1[C@H](O)C[C@@H](Cl)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C22H35ClO4/c1-2-22(13-8-14-22)20(25)11-7-10-17-16(18(23)15-19(17)24)9-5-3-4-6-12-21(26)27/h3,5,7,10,16-20,24-25H,2,4,6,8-9,11-15H2,1H3,(H,26,27)/b5-3-,10-7+/t16-,17-,18-,19-,20+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP3 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(Homo sapiens (Human)) | BDBM50016954

(CHEMBL1628262)Show SMILES CCCC1(CCC1)[C@@H](O)C\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C23H38O5/c1-2-13-23(14-8-15-23)21(26)11-7-10-18-17(19(24)16-20(18)25)9-5-3-4-6-12-22(27)28/h7,10,17-18,20-21,25-26H,2-6,8-9,11-16H2,1H3,(H,27,28)/b10-7+/t17-,18-,20-,21+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP3 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP3 subtype
(Homo sapiens (Human)) | BDBM85602

((R)-BUTAPROST | Butaprost (Free Acid) | CAS_69648-...)Show SMILES CCCC1(CCC1)[C@H](O)C\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(=O)OC |r| Show InChI InChI=1S/C24H40O5/c1-3-14-24(15-9-16-24)22(27)12-8-11-19-18(20(25)17-21(19)26)10-6-4-5-7-13-23(28)29-2/h8,11,18-19,21-22,26-27H,3-7,9-10,12-17H2,1-2H3/b11-8+/t18-,19-,21-,22-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP3 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP1 subtype
(Homo sapiens (Human)) | BDBM50101830

((Z)-7-{(1R,2R,3R,5R)-5-Chloro-2-[(E)-(S)-4-(1-ethy...)Show SMILES CCC1(CCC1)[C@@H](O)C\C=C\[C@H]1[C@H](O)C[C@@H](Cl)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C22H35ClO4/c1-2-22(13-8-14-22)20(25)11-7-10-17-16(18(23)15-19(17)24)9-5-3-4-6-12-21(26)27/h3,5,7,10,16-20,24-25H,2,4,6,8-9,11-15H2,1H3,(H,26,27)/b5-3-,10-7+/t16-,17-,18-,19-,20+/m1/s1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP1 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP1 subtype
(Homo sapiens (Human)) | BDBM50016954

(CHEMBL1628262)Show SMILES CCCC1(CCC1)[C@@H](O)C\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C23H38O5/c1-2-13-23(14-8-15-23)21(26)11-7-10-18-17(19(24)16-20(18)25)9-5-3-4-6-12-22(27)28/h7,10,17-18,20-21,25-26H,2-6,8-9,11-16H2,1H3,(H,27,28)/b10-7+/t17-,18-,20-,21+/m1/s1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP1 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP1 subtype
(Homo sapiens (Human)) | BDBM85602

((R)-BUTAPROST | Butaprost (Free Acid) | CAS_69648-...)Show SMILES CCCC1(CCC1)[C@H](O)C\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(=O)OC |r| Show InChI InChI=1S/C24H40O5/c1-3-14-24(15-9-16-24)22(27)12-8-11-19-18(20(25)17-21(19)26)10-6-4-5-7-13-23(28)29-2/h8,11,18-19,21-22,26-27H,3-7,9-10,12-17H2,1-2H3/b11-8+/t18-,19-,21-,22-/m1/s1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP1 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50016952

(CHEMBL3286796)Show SMILES O[C@@H](C\C=C\[C@H]1[C@H](O)C[C@@H](Cl)[C@@H]1C\C=C/CCCC(O)=O)C1(CC=C)CCC1 |r| Show InChI InChI=1S/C23H35ClO4/c1-2-13-23(14-8-15-23)21(26)11-7-10-18-17(19(24)16-20(18)25)9-5-3-4-6-12-22(27)28/h2-3,5,7,10,17-21,25-26H,1,4,6,8-9,11-16H2,(H,27,28)/b5-3-,10-7+/t17-,18-,19-,20-,21+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to prostanoid IP receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50101830

((Z)-7-{(1R,2R,3R,5R)-5-Chloro-2-[(E)-(S)-4-(1-ethy...)Show SMILES CCC1(CCC1)[C@@H](O)C\C=C\[C@H]1[C@H](O)C[C@@H](Cl)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C22H35ClO4/c1-2-22(13-8-14-22)20(25)11-7-10-17-16(18(23)15-19(17)24)9-5-3-4-6-12-21(26)27/h3,5,7,10,16-20,24-25H,2,4,6,8-9,11-15H2,1H3,(H,26,27)/b5-3-,10-7+/t16-,17-,18-,19-,20+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to prostanoid IP receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM85602

((R)-BUTAPROST | Butaprost (Free Acid) | CAS_69648-...)Show SMILES CCCC1(CCC1)[C@H](O)C\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(=O)OC |r| Show InChI InChI=1S/C24H40O5/c1-3-14-24(15-9-16-24)22(27)12-8-11-19-18(20(25)17-21(19)26)10-6-4-5-7-13-23(28)29-2/h8,11,18-19,21-22,26-27H,3-7,9-10,12-17H2,1-2H3/b11-8+/t18-,19-,21-,22-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to prostanoid IP receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM35847

((15S)-prostaglandin E2 | (5Z,11alpha,13E,15S)-11,1...)Show SMILES CCCCC[C@H](O)\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C20H32O5/c1-2-3-6-9-15(21)12-13-17-16(18(22)14-19(17)23)10-7-4-5-8-11-20(24)25/h4,7,12-13,15-17,19,21,23H,2-3,5-6,8-11,14H2,1H3,(H,24,25)/b7-4-,13-12+/t15-,16+,17+,19+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to prostanoid IP receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP4 subtype
(Homo sapiens (Human)) | BDBM50101830

((Z)-7-{(1R,2R,3R,5R)-5-Chloro-2-[(E)-(S)-4-(1-ethy...)Show SMILES CCC1(CCC1)[C@@H](O)C\C=C\[C@H]1[C@H](O)C[C@@H](Cl)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C22H35ClO4/c1-2-22(13-8-14-22)20(25)11-7-10-17-16(18(23)15-19(17)24)9-5-3-4-6-12-21(26)27/h3,5,7,10,16-20,24-25H,2,4,6,8-9,11-15H2,1H3,(H,26,27)/b5-3-,10-7+/t16-,17-,18-,19-,20+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP4 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP4 subtype
(Homo sapiens (Human)) | BDBM85602

((R)-BUTAPROST | Butaprost (Free Acid) | CAS_69648-...)Show SMILES CCCC1(CCC1)[C@H](O)C\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(=O)OC |r| Show InChI InChI=1S/C24H40O5/c1-3-14-24(15-9-16-24)22(27)12-8-11-19-18(20(25)17-21(19)26)10-6-4-5-7-13-23(28)29-2/h8,11,18-19,21-22,26-27H,3-7,9-10,12-17H2,1-2H3/b11-8+/t18-,19-,21-,22-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP4 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP4 subtype
(Homo sapiens (Human)) | BDBM50016954

(CHEMBL1628262)Show SMILES CCCC1(CCC1)[C@@H](O)C\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C23H38O5/c1-2-13-23(14-8-15-23)21(26)11-7-10-18-17(19(24)16-20(18)25)9-5-3-4-6-12-22(27)28/h7,10,17-18,20-21,25-26H,2-6,8-9,11-16H2,1H3,(H,27,28)/b10-7+/t17-,18-,20-,21+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Binding affinity to EP4 receptor (unknown origin) |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP4 subtype
(Homo sapiens (Human)) | BDBM85184

(AH-6809 | CAS_33458-93-4 | NSC_119461)Show InChI InChI=1S/C17H14O5/c1-9(2)21-11-4-5-12-15(8-11)22-14-6-3-10(17(19)20)7-13(14)16(12)18/h3-9H,1-2H3,(H,19,20) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Antagonist activity at EP4 receptor (unknown origin) by functional cAMP assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM85184

(AH-6809 | CAS_33458-93-4 | NSC_119461)Show InChI InChI=1S/C17H14O5/c1-9(2)21-11-4-5-12-15(8-11)22-14-6-3-10(17(19)20)7-13(14)16(12)18/h3-9H,1-2H3,(H,19,20) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Antagonist activity at prostanoid IP receptor (unknown origin) by functional cAMP assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50016954

(CHEMBL1628262)Show SMILES CCCC1(CCC1)[C@@H](O)C\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(O)=O Show InChI InChI=1S/C23H38O5/c1-2-13-23(14-8-15-23)21(26)11-7-10-18-17(19(24)16-20(18)25)9-5-3-4-6-12-22(27)28/h7,10,17-18,20-21,25-26H,2-6,8-9,11-16H2,1H3,(H,27,28)/b10-7+/t17-,18-,20-,21+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Agonist activity at prostanoid IP receptor (unknown origin) by functional assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM85602

((R)-BUTAPROST | Butaprost (Free Acid) | CAS_69648-...)Show SMILES CCCC1(CCC1)[C@H](O)C\C=C\[C@H]1[C@H](O)CC(=O)[C@@H]1CCCCCCC(=O)OC |r| Show InChI InChI=1S/C24H40O5/c1-3-14-24(15-9-16-24)22(27)12-8-11-19-18(20(25)17-21(19)26)10-6-4-5-7-13-23(28)29-2/h8,11,18-19,21-22,26-27H,3-7,9-10,12-17H2,1-2H3/b11-8+/t18-,19-,21-,22-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University School of Medicine
Curated by ChEMBL
| Assay Description
Agonist activity at prostanoid IP receptor (unknown origin) by functional assay |
J Med Chem 57: 4454-65 (2014)
Article DOI: 10.1021/jm401431x
BindingDB Entry DOI: 10.7270/Q2CR5VXZ |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50180302
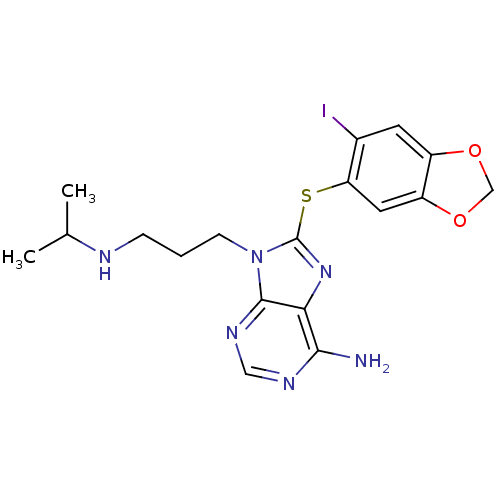
(8-(6-iodo-benzo[1,3]dioxol-5-ylsulfanyl)-9-(3-isop...)Show InChI InChI=1S/C18H21IN6O2S/c1-10(2)21-4-3-5-25-17-15(16(20)22-8-23-17)24-18(25)28-14-7-13-12(6-11(14)19)26-9-27-13/h6-8,10,21H,3-5,9H2,1-2H3,(H2,20,22,23) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 43 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University
Curated by ChEMBL
| Assay Description
Inhibition of Hsp90 in human MCF7 cell lysates assessed as interaction with Cy3b-conjugated geldanamycin by FP assay |
Bioorg Med Chem 16: 6903-10 (2008)
Article DOI: 10.1016/j.bmc.2008.05.047
BindingDB Entry DOI: 10.7270/Q2WS8X18 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50180302

(8-(6-iodo-benzo[1,3]dioxol-5-ylsulfanyl)-9-(3-isop...)Show InChI InChI=1S/C18H21IN6O2S/c1-10(2)21-4-3-5-25-17-15(16(20)22-8-23-17)24-18(25)28-14-7-13-12(6-11(14)19)26-9-27-13/h6-8,10,21H,3-5,9H2,1-2H3,(H2,20,22,23) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University
Curated by ChEMBL
| Assay Description
Inhibition of Hsp90 in human MCF7 cells assessed as Her2 level after 24 hrs by Western blot |
Bioorg Med Chem 16: 6903-10 (2008)
Article DOI: 10.1016/j.bmc.2008.05.047
BindingDB Entry DOI: 10.7270/Q2WS8X18 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Myeloperoxidase
(Homo sapiens (Human)) | BDBM50499557
(CHEMBL3739661)Show SMILES Fc1ccc2[nH]c(=S)n(NC(=O)c3ccc(cc3)C(F)(F)F)c(=O)c2c1 Show InChI InChI=1S/C16H9F4N3O2S/c17-10-5-6-12-11(7-10)14(25)23(15(26)21-12)22-13(24)8-1-3-9(4-2-8)16(18,19)20/h1-7H,(H,21,26)(H,22,24) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant myeloperoxidase by amplex red assay |
ACS Med Chem Lett 6: 1047-52 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00287
BindingDB Entry DOI: 10.7270/Q26W9F24 |
More data for this
Ligand-Target Pair | |
Myeloperoxidase
(Homo sapiens (Human)) | BDBM50499533

(CHEMBL3740322)Show SMILES [O-][N+](=O)c1ccc(cc1)C(=O)Nn1c(=S)[nH]c2ccc(F)cc2c1=O Show InChI InChI=1S/C15H9FN4O4S/c16-9-3-6-12-11(7-9)14(22)19(15(25)17-12)18-13(21)8-1-4-10(5-2-8)20(23)24/h1-7H,(H,17,25)(H,18,21) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant myeloperoxidase by amplex red assay |
ACS Med Chem Lett 6: 1047-52 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00287
BindingDB Entry DOI: 10.7270/Q26W9F24 |
More data for this
Ligand-Target Pair | |
Myeloperoxidase
(Homo sapiens (Human)) | BDBM50499544
(CHEMBL3740127)Show SMILES Fc1ccc2[nH]c(=S)n(Nc3ncc(cc3Cl)C(F)(F)F)c(=O)c2c1 Show InChI InChI=1S/C14H7ClF4N4OS/c15-9-3-6(14(17,18)19)5-20-11(9)22-23-12(24)8-4-7(16)1-2-10(8)21-13(23)25/h1-5H,(H,20,22)(H,21,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant myeloperoxidase by amplex red assay |
ACS Med Chem Lett 6: 1047-52 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00287
BindingDB Entry DOI: 10.7270/Q26W9F24 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data