 Found 67977 hits with Last Name = 'ha' and Initial = 'e'
Found 67977 hits with Last Name = 'ha' and Initial = 'e' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Adenosine deaminase
(Bos taurus (bovine)) | BDBM22925

((8R)-3-[(2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)oxol...)Show SMILES OC[C@H]1O[C@H](C[C@@H]1O)n1cnc2[C@H](O)CNC=Nc12 |c:17| Show InChI InChI=1S/C11H16N4O4/c16-3-8-6(17)1-9(19-8)15-5-14-10-7(18)2-12-4-13-11(10)15/h4-9,16-18H,1-3H2,(H,12,13)/t6-,7+,8+,9+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PubMed
| 0.00250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Rhode Island
Curated by ChEMBL
| Assay Description
Binding affinity (Ki) at calf intestinal adenosine deaminase. |
J Med Chem 35: 4180-4 (1992)
Checked by Author
BindingDB Entry DOI: 10.7270/Q2SQ9118 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM110961

(US8614206, 518)Show SMILES Cn1ncc(NC(=O)c2nc(sc2N)-c2c(F)cccc2F)c1N1CC[C@@](C)(N)CC(F)(F)C1 |r| Show InChI InChI=1S/C21H23F4N7OS/c1-20(27)6-7-32(10-21(24,25)9-20)19-13(8-28-31(19)2)29-17(33)15-16(26)34-18(30-15)14-11(22)4-3-5-12(14)23/h3-5,8H,6-7,9-10,26-27H2,1-2H3,(H,29,33)/t20-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM1 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM50434655
(CHEMBL2387464)Show SMILES F[C@H]1CNCC[C@@H]1CNc1ccn2ncc(C(=O)Nc3c[nH]c4ncc(cc34)-c3ccccc3F)c2n1 |r| Show InChI InChI=1S/C26H24F2N8O/c27-20-4-2-1-3-17(20)16-9-18-22(14-32-24(18)31-11-16)34-26(37)19-12-33-36-8-6-23(35-25(19)36)30-10-15-5-7-29-13-21(15)28/h1-4,6,8-9,11-12,14-15,21,29H,5,7,10,13H2,(H,30,35)(H,31,32)(H,34,37)/t15-,21+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of PIM1 (unknown origin) using FAM-pimtide as substrate after 90 mins by spectrophotometry in presence of ATP |
Bioorg Med Chem Lett 23: 3149-53 (2013)
Article DOI: 10.1016/j.bmcl.2013.04.020
BindingDB Entry DOI: 10.7270/Q2PC33S9 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-3
(Homo sapiens (Human)) | BDBM227170

(US9328106, 118)Show SMILES Cn1ncc(NC(=O)c2nc(sc2N)-c2c(F)cccc2F)c1[C@@]12CC[C@@H](O1)[C@H](N)CC2 |r| Show InChI InChI=1S/C21H22F2N6O2S/c1-29-17(21-7-5-12(24)14(31-21)6-8-21)13(9-26-29)27-19(30)16-18(25)32-20(28-16)15-10(22)3-2-4-11(15)23/h2-4,9,12,14H,5-8,24-25H2,1H3,(H,27,30)/t12-,14-,21-/m1/s1 | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM3 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-3
(Homo sapiens (Human)) | BDBM50505054

(CHEMBL4455188)Show SMILES Cn1ncc(NC(=O)c2nc(sc2N)-c2c(F)cccc2F)c1N1CCC[C@@H](CC1)NCC(F)F |r| Show InChI InChI=1S/C22H25F4N7OS/c1-32-22(33-8-3-4-12(7-9-33)28-11-16(25)26)15(10-29-32)30-20(34)18-19(27)35-21(31-18)17-13(23)5-2-6-14(17)24/h2,5-6,10,12,16,28H,3-4,7-9,11,27H2,1H3,(H,30,34)/t12-/m0/s1 | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM3 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
5'-nucleotidase
(Homo sapiens (Human)) | BDBM50527134
(CHEMBL4471306 | US20230295213, Compound a)Show SMILES C[C@H](Nc1cc(Cl)nc2n(ncc12)[C@@H]1O[C@H](COP(O)(=O)CP(O)(O)=O)[C@@H](O)[C@H]1O)c1ccccc1F |r| Show InChI InChI=1S/C20H24ClFN4O9P2/c1-10(11-4-2-3-5-13(11)22)24-14-6-16(21)25-19-12(14)7-23-26(19)20-18(28)17(27)15(35-20)8-34-37(32,33)9-36(29,30)31/h2-7,10,15,17-18,20,27-28H,8-9H2,1H3,(H,24,25)(H,32,33)(H2,29,30,31)/t10-,15+,17+,18+,20+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Competitive reversible inhibition of human C-terminal His6-tagged CD73 expressed in HEK293 cells using AMP as substrate preincubated with substrate f... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00525
BindingDB Entry DOI: 10.7270/Q29W0K29 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM50505052

(CHEMBL3623150)Show SMILES Cn1ncc(NC(=O)c2nc(sc2N)-c2c(F)cccc2F)c1N1CC[C@@H](N)CC(F)(F)C1 |r| Show InChI InChI=1S/C20H21F4N7OS/c1-30-19(31-6-5-10(25)7-20(23,24)9-31)13(8-27-30)28-17(32)15-16(26)33-18(29-15)14-11(21)3-2-4-12(14)22/h2-4,8,10H,5-7,9,25-26H2,1H3,(H,28,32)/t10-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM1 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM227170

(US9328106, 118)Show SMILES Cn1ncc(NC(=O)c2nc(sc2N)-c2c(F)cccc2F)c1[C@@]12CC[C@@H](O1)[C@H](N)CC2 |r| Show InChI InChI=1S/C21H22F2N6O2S/c1-29-17(21-7-5-12(24)14(31-21)6-8-21)13(9-26-29)27-19(30)16-18(25)32-20(28-16)15-10(22)3-2-4-11(15)23/h2-4,9,12,14H,5-8,24-25H2,1H3,(H,27,30)/t12-,14-,21-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM1 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-3
(Homo sapiens (Human)) | BDBM50505051
(CHEMBL4437940)Show SMILES Cn1ncc(NC(=O)c2nc(sc2N)-c2c(F)cccc2F)c1N1CCC[C@H](N)CC1 |r| Show InChI InChI=1S/C20H23F2N7OS/c1-28-20(29-8-3-4-11(23)7-9-29)14(10-25-28)26-18(30)16-17(24)31-19(27-16)15-12(21)5-2-6-13(15)22/h2,5-6,10-11H,3-4,7-9,23-24H2,1H3,(H,26,30)/t11-/m0/s1 | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM3 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM50505059

(CHEMBL4459538)Show SMILES CO[C@H]1C[C@H](N)CCN(C1)c1c(NC(=O)c2nc(sc2N)-c2c(F)cccc2F)cnn1C |r| Show InChI InChI=1S/C21H25F2N7O2S/c1-29-21(30-7-6-11(24)8-12(10-30)32-2)15(9-26-29)27-19(31)17-18(25)33-20(28-17)16-13(22)4-3-5-14(16)23/h3-5,9,11-12H,6-8,10,24-25H2,1-2H3,(H,27,31)/t11-,12+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM1 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM50434656
(CHEMBL2387463)Show SMILES Fc1ccccc1-c1cnc2[nH]cc(NC(=O)c3cnn4ccc(NCC5CCNCC5)nc34)c2c1 Show InChI InChI=1S/C26H25FN8O/c27-21-4-2-1-3-18(21)17-11-19-22(15-31-24(19)30-13-17)33-26(36)20-14-32-35-10-7-23(34-25(20)35)29-12-16-5-8-28-9-6-16/h1-4,7,10-11,13-16,28H,5-6,8-9,12H2,(H,29,34)(H,30,31)(H,33,36) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of PIM1 (unknown origin) using FAM-pimtide as substrate after 90 mins by spectrophotometry in presence of ATP |
Bioorg Med Chem Lett 23: 3149-53 (2013)
Article DOI: 10.1016/j.bmcl.2013.04.020
BindingDB Entry DOI: 10.7270/Q2PC33S9 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM50434654
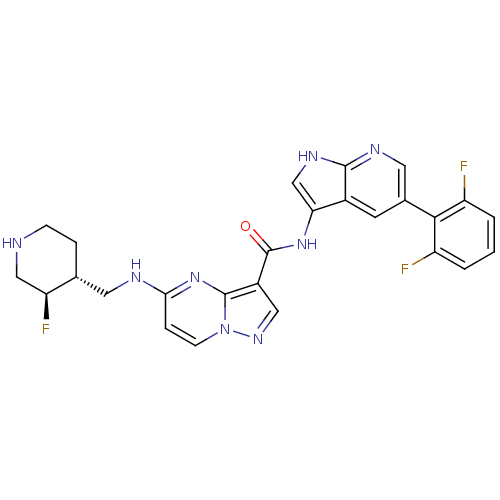
(CHEMBL2387465)Show SMILES F[C@H]1CNCC[C@@H]1CNc1ccn2ncc(C(=O)Nc3c[nH]c4ncc(cc34)-c3c(F)cccc3F)c2n1 |r| Show InChI InChI=1S/C26H23F3N8O/c27-18-2-1-3-19(28)23(18)15-8-16-21(13-33-24(16)32-10-15)35-26(38)17-11-34-37-7-5-22(36-25(17)37)31-9-14-4-6-30-12-20(14)29/h1-3,5,7-8,10-11,13-14,20,30H,4,6,9,12H2,(H,31,36)(H,32,33)(H,35,38)/t14-,20+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of PIM1 (unknown origin) using FAM-pimtide as substrate after 90 mins by spectrophotometry in presence of ATP |
Bioorg Med Chem Lett 23: 3149-53 (2013)
Article DOI: 10.1016/j.bmcl.2013.04.020
BindingDB Entry DOI: 10.7270/Q2PC33S9 |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM209097
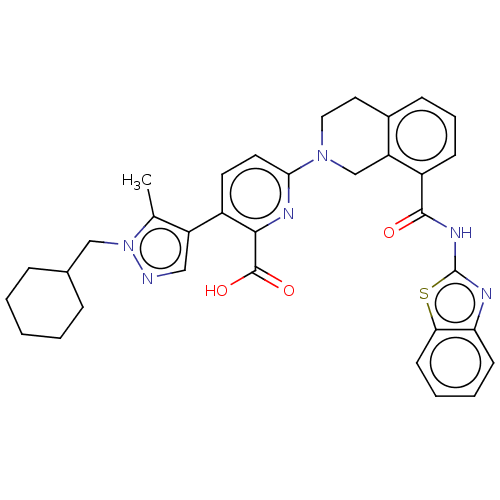
(US9266877, 43)Show SMILES Cc1c(cnn1CC1CCCCC1)-c1ccc(nc1C(O)=O)N1CCc2cccc(C(=O)Nc3nc4ccccc4s3)c2C1 Show InChI InChI=1S/C34H34N6O3S/c1-21-26(18-35-40(21)19-22-8-3-2-4-9-22)24-14-15-30(37-31(24)33(42)43)39-17-16-23-10-7-11-25(27(23)20-39)32(41)38-34-36-28-12-5-6-13-29(28)44-34/h5-7,10-15,18,22H,2-4,8-9,16-17,19-20H2,1H3,(H,42,43)(H,36,38,41) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of F-Bak (GQVGRQLAIIGDK(6-FAM)INR-amide probe binding to BCL-xl (unknown origin) incubated for 1 hr by TR-FRET assay |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.9b00568
BindingDB Entry DOI: 10.7270/Q2542S8D |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-3
(Homo sapiens (Human)) | BDBM248955

(US9434725, 186)Show SMILES COc1ccc(nc1N1CCC[C@H](N)C1)-n1ncc2cnc(cc12)-c1cncc(C)n1 |r| Show InChI InChI=1S/C22H24N8O/c1-14-9-24-12-18(27-14)17-8-19-15(10-25-17)11-26-30(19)21-6-5-20(31-2)22(28-21)29-7-3-4-16(23)13-29/h5-6,8-12,16H,3-4,7,13,23H2,1-2H3/t16-/m0/s1 | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.00800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of PIM3 (unknown origin) using 5FAM-ARKRRRHPSGPPTA as substrate after 90 mins in presence of ATP by caliper microfluidic mobility shift as... |
J Med Chem 60: 4458-4473 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00418
BindingDB Entry DOI: 10.7270/Q2H997PN |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM50434666
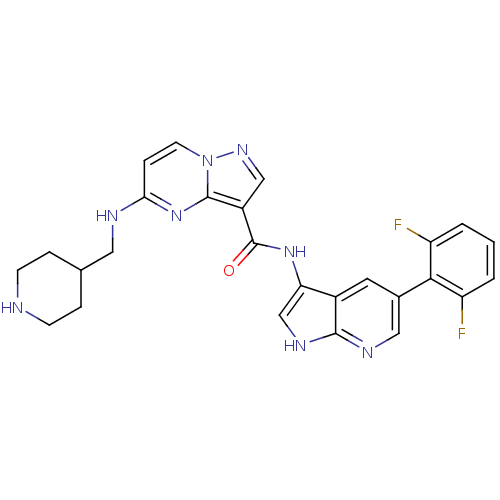
(CHEMBL2387471)Show SMILES Fc1cccc(F)c1-c1cnc2[nH]cc(NC(=O)c3cnn4ccc(NCC5CCNCC5)nc34)c2c1 Show InChI InChI=1S/C26H24F2N8O/c27-19-2-1-3-20(28)23(19)16-10-17-21(14-32-24(17)31-12-16)34-26(37)18-13-33-36-9-6-22(35-25(18)36)30-11-15-4-7-29-8-5-15/h1-3,6,9-10,12-15,29H,4-5,7-8,11H2,(H,30,35)(H,31,32)(H,34,37) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of PIM1 (unknown origin) using FAM-pimtide as substrate after 90 mins by spectrophotometry in presence of ATP |
Bioorg Med Chem Lett 23: 3149-53 (2013)
Article DOI: 10.1016/j.bmcl.2013.04.020
BindingDB Entry DOI: 10.7270/Q2PC33S9 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-3
(Homo sapiens (Human)) | BDBM50434655

(CHEMBL2387464)Show SMILES F[C@H]1CNCC[C@@H]1CNc1ccn2ncc(C(=O)Nc3c[nH]c4ncc(cc34)-c3ccccc3F)c2n1 |r| Show InChI InChI=1S/C26H24F2N8O/c27-20-4-2-1-3-17(20)16-9-18-22(14-32-24(18)31-11-16)34-26(37)19-12-33-36-8-6-23(35-25(19)36)30-10-15-5-7-29-13-21(15)28/h1-4,6,8-9,11-12,14-15,21,29H,5,7,10,13H2,(H,30,35)(H,31,32)(H,34,37)/t15-,21+/m1/s1 | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of PIM3 (unknown origin) using FAM-pimtide as substrate after 90 mins by spectrophotometry in presence of ATP |
Bioorg Med Chem Lett 23: 3149-53 (2013)
Article DOI: 10.1016/j.bmcl.2013.04.020
BindingDB Entry DOI: 10.7270/Q2PC33S9 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-3
(Homo sapiens (Human)) | BDBM110700

(US8614206, 120 | US8614206, 125 | US8614206, 400)Show SMILES Cn1ncc(NC(=O)c2nc(sc2N)-c2c(F)cccc2F)c1N1CCCC(N)C(F)C1 Show InChI InChI=1S/C20H22F3N7OS/c1-29-20(30-7-3-6-13(24)12(23)9-30)14(8-26-29)27-18(31)16-17(25)32-19(28-16)15-10(21)4-2-5-11(15)22/h2,4-5,8,12-13H,3,6-7,9,24-25H2,1H3,(H,27,31) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM3 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-3
(Homo sapiens (Human)) | BDBM50505052

(CHEMBL3623150)Show SMILES Cn1ncc(NC(=O)c2nc(sc2N)-c2c(F)cccc2F)c1N1CC[C@@H](N)CC(F)(F)C1 |r| Show InChI InChI=1S/C20H21F4N7OS/c1-30-19(31-6-5-10(25)7-20(23,24)9-31)13(8-27-30)28-17(32)15-16(26)33-18(29-15)14-11(21)3-2-4-12(14)22/h2-4,8,10H,5-7,9,25-26H2,1H3,(H,28,32)/t10-/m1/s1 | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM3 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50030754
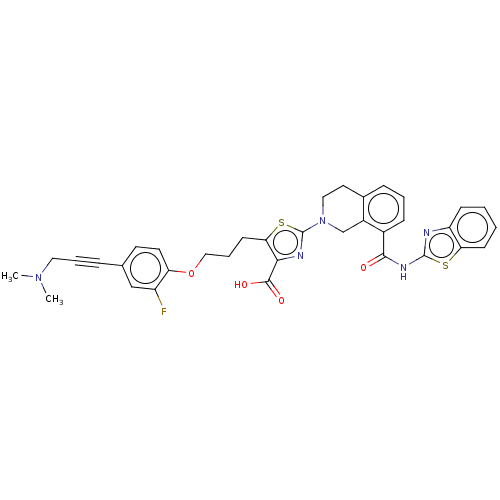
(CHEMBL3342332)Show SMILES CN(C)CC#Cc1ccc(OCCCc2sc(nc2C(O)=O)N2CCc3cccc(C(=O)Nc4nc5ccccc5s4)c3C2)c(F)c1 Show InChI InChI=1S/C35H32FN5O4S2/c1-40(2)17-6-8-22-14-15-28(26(36)20-22)45-19-7-13-30-31(33(43)44)38-35(47-30)41-18-16-23-9-5-10-24(25(23)21-41)32(42)39-34-37-27-11-3-4-12-29(27)46-34/h3-5,9-12,14-15,20H,7,13,16-19,21H2,1-2H3,(H,43,44)(H,37,39,42) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of BCL-XL (unknown origin) incubated for 1 hr by TR-FRET assay |
ACS Med Chem Lett 5: 1088-93 (2014)
Article DOI: 10.1021/ml5001867
BindingDB Entry DOI: 10.7270/Q2VX0J43 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-3
(Homo sapiens (Human)) | BDBM50505053

(CHEMBL4469964)Show SMILES Cn1ncc(NC(=O)c2nc(sc2N)-c2c(F)cccc2F)c1C1CCC(N)C(F)CC1 Show InChI InChI=1S/C21H23F3N6OS/c1-30-18(10-5-7-11(22)14(25)8-6-10)15(9-27-30)28-20(31)17-19(26)32-21(29-17)16-12(23)3-2-4-13(16)24/h2-4,9-11,14H,5-8,25-26H2,1H3,(H,28,31) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM3 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50561528
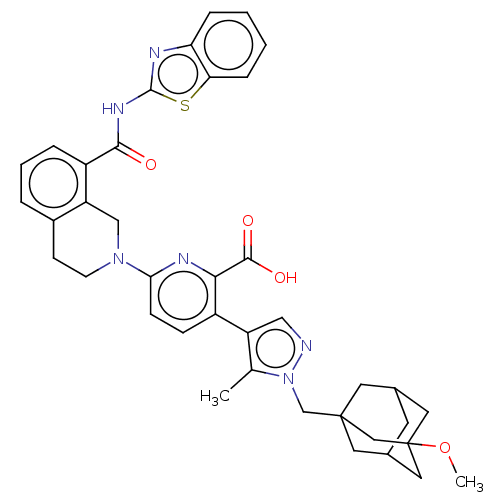
(CHEMBL4762875)Show SMILES COC12CC3CC(CC(Cn4ncc(c4C)-c4ccc(nc4C(O)=O)N4CCc5cccc(C(=O)Nc6nc7ccccc7s6)c5C4)(C3)C1)C2 |TLB:1:2:5.4.47:7,5:6:4.3.47:48,THB:5:4:6.49.7:48,3:4:7:49.2.48,3:2:5.4.47:7| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of F-Bak (GQVGRQLAIIGDK(6-FAM)INR-amide probe binding to BCL-xl (unknown origin) incubated for 1 hr by TR-FRET assay |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.9b00568
BindingDB Entry DOI: 10.7270/Q2542S8D |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50162797
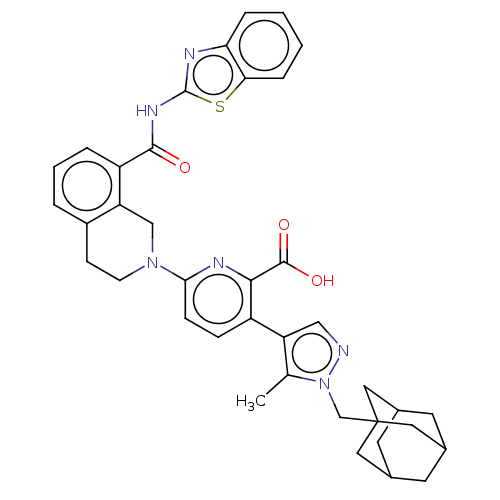
(CHEMBL3793424)Show SMILES Cc1c(cnn1CC12CC3CC(CC(C3)C1)C2)-c1ccc(nc1C(O)=O)N1CCc2cccc(C(=O)Nc3nc4ccccc4s3)c2C1 |TLB:14:9:16:12.13.15,14:13:16:10.9.8,THB:12:11:8:14.13.15,12:13:10.11.16:8| Show InChI InChI=1S/C38H38N6O3S/c1-22-29(19-39-44(22)21-38-16-23-13-24(17-38)15-25(14-23)18-38)27-9-10-33(41-34(27)36(46)47)43-12-11-26-5-4-6-28(30(26)20-43)35(45)42-37-40-31-7-2-3-8-32(31)48-37/h2-10,19,23-25H,11-18,20-21H2,1H3,(H,46,47)(H,40,42,45) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of F-Bak (GQVGRQLAIIGDK(6-FAM)INR-amide probe binding to BCL-xl (unknown origin) incubated for 1 hr by TR-FRET assay |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.9b00568
BindingDB Entry DOI: 10.7270/Q2542S8D |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Rattus norvegicus (rat)) | BDBM50109062
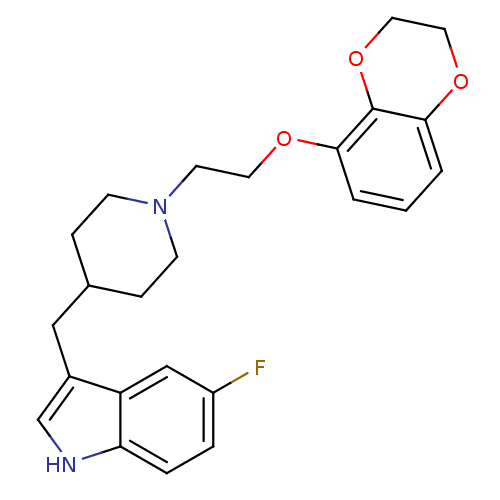
(3-{1-[2-(2,3-Dihydro-benzo[1,4]dioxin-5-yloxy)-eth...)Show SMILES Fc1ccc2[nH]cc(CC3CCN(CCOc4cccc5OCCOc45)CC3)c2c1 Show InChI InChI=1S/C24H27FN2O3/c25-19-4-5-21-20(15-19)18(16-26-21)14-17-6-8-27(9-7-17)10-11-28-22-2-1-3-23-24(22)30-13-12-29-23/h1-5,15-17,26H,6-14H2 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research Laboratories
Curated by ChEMBL
| Assay Description
Affinity of the compound for RB Serotonin transporter was determined in vitro by incubating compound |
Bioorg Med Chem Lett 12: 307-10 (2002)
BindingDB Entry DOI: 10.7270/Q2FJ2G2M |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-3
(Homo sapiens (Human)) | BDBM50505061
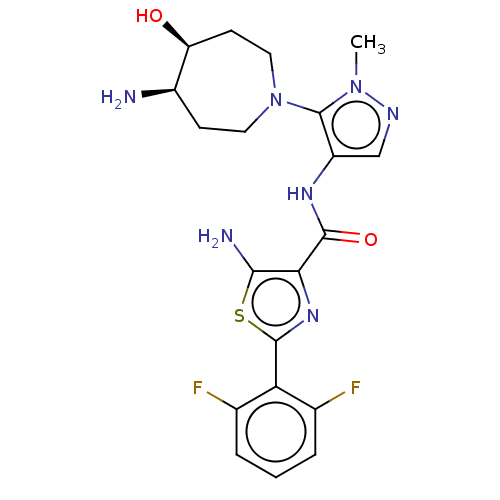
(CHEMBL4453890)Show SMILES Cn1ncc(NC(=O)c2nc(sc2N)-c2c(F)cccc2F)c1N1CC[C@@H](N)[C@@H](O)CC1 |r| Show InChI InChI=1S/C20H23F2N7O2S/c1-28-20(29-7-5-12(23)14(30)6-8-29)13(9-25-28)26-18(31)16-17(24)32-19(27-16)15-10(21)3-2-4-11(15)22/h2-4,9,12,14,30H,5-8,23-24H2,1H3,(H,26,31)/t12-,14+/m1/s1 | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM3 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50030757
(CHEMBL3342196)Show SMILES CN1CCN(CCCNc2ncnc3n(ncc23)-c2ccc(OCCCc3sc(nc3C(O)=O)N3CCc4cccc(C(=O)Nc5nc6ccccc6s5)c4C3)cc2)CC1 Show InChI InChI=1S/C43H45N11O4S2/c1-51-20-22-52(23-21-51)18-6-17-44-38-32-25-47-54(39(32)46-27-45-38)29-12-14-30(15-13-29)58-24-5-11-36-37(41(56)57)49-43(60-36)53-19-16-28-7-4-8-31(33(28)26-53)40(55)50-42-48-34-9-2-3-10-35(34)59-42/h2-4,7-10,12-15,25,27H,5-6,11,16-24,26H2,1H3,(H,56,57)(H,44,45,46)(H,48,50,55) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of BCL-XL (unknown origin) incubated for 1 hr by TR-FRET assay |
ACS Med Chem Lett 5: 1088-93 (2014)
Article DOI: 10.1021/ml5001867
BindingDB Entry DOI: 10.7270/Q2VX0J43 |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50030758
(CHEMBL3342195)Show SMILES CN(C)CCCNc1ncnc2n(ncc12)-c1ccc(OCCCc2sc(nc2C(O)=O)N2CCc3cccc(C(=O)Nc4nc5ccccc5s4)c3C2)cc1 Show InChI InChI=1S/C40H40N10O4S2/c1-48(2)19-7-18-41-35-29-22-44-50(36(29)43-24-42-35)26-13-15-27(16-14-26)54-21-6-12-33-34(38(52)53)46-40(56-33)49-20-17-25-8-5-9-28(30(25)23-49)37(51)47-39-45-31-10-3-4-11-32(31)55-39/h3-5,8-11,13-16,22,24H,6-7,12,17-21,23H2,1-2H3,(H,52,53)(H,41,42,43)(H,45,47,51) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of BCL-XL (unknown origin) incubated for 1 hr by TR-FRET assay |
ACS Med Chem Lett 5: 1088-93 (2014)
Article DOI: 10.1021/ml5001867
BindingDB Entry DOI: 10.7270/Q2VX0J43 |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50030759
(CHEMBL3342194)Show SMILES OC(=O)c1nc(sc1CCCOc1ccc(cc1)-n1cccn1)N1CCc2cccc(C(=O)Nc3nc4ccccc4s3)c2C1 Show InChI InChI=1S/C33H28N6O4S2/c40-30(37-32-35-26-8-1-2-9-27(26)44-32)24-7-3-6-21-15-18-38(20-25(21)24)33-36-29(31(41)42)28(45-33)10-4-19-43-23-13-11-22(12-14-23)39-17-5-16-34-39/h1-3,5-9,11-14,16-17H,4,10,15,18-20H2,(H,41,42)(H,35,37,40) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of BCL-XL (unknown origin) incubated for 1 hr by TR-FRET assay |
ACS Med Chem Lett 5: 1088-93 (2014)
Article DOI: 10.1021/ml5001867
BindingDB Entry DOI: 10.7270/Q2VX0J43 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50334988
((S)-2-amino-N-((R)-1-(2-((S)-3-(4-fluorophenyl)-1-...)Show SMILES CCC(=O)N(C1CCN(CC1)C(=O)[C@H](Cc1ccc(F)cc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C)c1ccccc1 |r| Show InChI InChI=1S/C39H49FN6O6/c1-5-36(49)46(29-9-7-6-8-10-29)30-15-17-45(18-16-30)39(52)34(21-27-11-13-28(40)14-12-27)44-35(48)23-42-37(50)26(4)43-38(51)33(41)22-32-24(2)19-31(47)20-25(32)3/h6-14,19-20,26,30,33-34,47H,5,15-18,21-23,41H2,1-4H3,(H,42,50)(H,43,51)(H,44,48)/t26-,33+,34+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat mu opioid receptor expressed in mouse HN9.10 cells |
J Med Chem 54: 382-6 (2011)
Article DOI: 10.1021/jm100982d
BindingDB Entry DOI: 10.7270/Q2N29XWF |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50030752
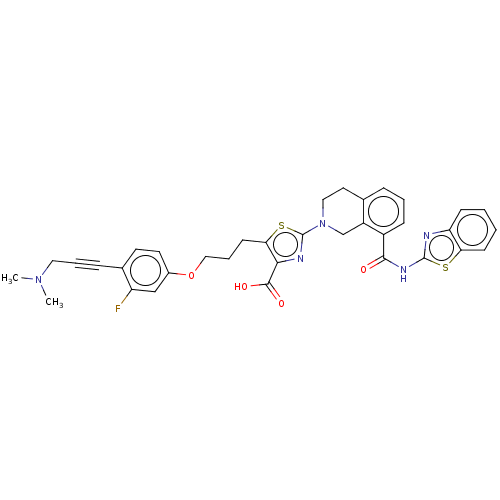
(CHEMBL3342333)Show SMILES CN(C)CC#Cc1ccc(OCCCc2sc(nc2C(O)=O)N2CCc3cccc(C(=O)Nc4nc5ccccc5s4)c3C2)cc1F Show InChI InChI=1S/C35H32FN5O4S2/c1-40(2)17-6-9-23-14-15-24(20-27(23)36)45-19-7-13-30-31(33(43)44)38-35(47-30)41-18-16-22-8-5-10-25(26(22)21-41)32(42)39-34-37-28-11-3-4-12-29(28)46-34/h3-5,8,10-12,14-15,20H,7,13,16-19,21H2,1-2H3,(H,43,44)(H,37,39,42) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of BCL-XL (unknown origin) incubated for 1 hr by TR-FRET assay |
ACS Med Chem Lett 5: 1088-93 (2014)
Article DOI: 10.1021/ml5001867
BindingDB Entry DOI: 10.7270/Q2VX0J43 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-3
(Homo sapiens (Human)) | BDBM110961

(US8614206, 518)Show SMILES Cn1ncc(NC(=O)c2nc(sc2N)-c2c(F)cccc2F)c1N1CC[C@@](C)(N)CC(F)(F)C1 |r| Show InChI InChI=1S/C21H23F4N7OS/c1-20(27)6-7-32(10-21(24,25)9-20)19-13(8-28-31(19)2)29-17(33)15-16(26)34-18(30-15)14-11(22)4-3-5-12(14)23/h3-5,8H,6-7,9-10,26-27H2,1-2H3,(H,29,33)/t20-/m1/s1 | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM3 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM50505057

(CHEMBL3676285)Show SMILES N[C@H]1CCCN(C1)c1ccncc1NC(=O)c1nc(sc1N)-c1c(F)cccc1F Show InChI InChI=1S/C20H20F2N6OS/c21-12-4-1-5-13(22)16(12)20-27-17(18(24)30-20)19(29)26-14-9-25-7-6-15(14)28-8-2-3-11(23)10-28/h1,4-7,9,11H,2-3,8,10,23-24H2,(H,26,29)/t11-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM1 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM50505050

(CHEMBL4439756)Show SMILES Cn1ncc(NC(=O)c2nc(sc2N)-c2c(F)cccc2F)c1N1CCC(N)CC(F)C1 Show InChI InChI=1S/C20H22F3N7OS/c1-29-20(30-6-5-11(24)7-10(21)9-30)14(8-26-29)27-18(31)16-17(25)32-19(28-16)15-12(22)3-2-4-13(15)23/h2-4,8,10-11H,5-7,9,24-25H2,1H3,(H,27,31) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM1 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-3
(Homo sapiens (Human)) | BDBM50505050

(CHEMBL4439756)Show SMILES Cn1ncc(NC(=O)c2nc(sc2N)-c2c(F)cccc2F)c1N1CCC(N)CC(F)C1 Show InChI InChI=1S/C20H22F3N7OS/c1-29-20(30-6-5-11(24)7-10(21)9-30)14(8-26-29)27-18(31)16-17(25)32-19(28-16)15-12(22)3-2-4-13(15)23/h2-4,8,10-11H,5-7,9,24-25H2,1H3,(H,27,31) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human PIM3 using FAM-pimtide as substrate after 90 mins by Z-LYTE assay |
J Med Chem 62: 2140-2153 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01857
BindingDB Entry DOI: 10.7270/Q2Q52SVB |
More data for this
Ligand-Target Pair | |
Calcitonin gene-related peptide type 1 receptor
(Homo sapiens (Human)) | BDBM50184069
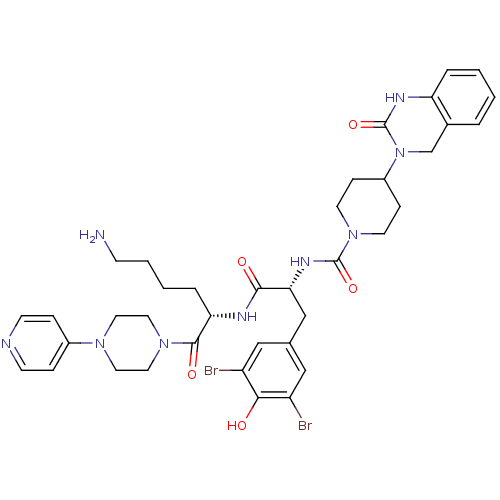
(CHEMBL207197 | N-((R)-1-((S)-6-amino-1-oxo-1-(4-(p...)Show SMILES NCCCC[C@H](NC(=O)[C@@H](Cc1cc(Br)c(O)c(Br)c1)NC(=O)N1CCC(CC1)N1Cc2ccccc2NC1=O)C(=O)N1CCN(CC1)c1ccncc1 Show InChI InChI=1S/C38H47Br2N9O5/c39-29-21-25(22-30(40)34(29)50)23-33(45-37(53)48-15-10-28(11-16-48)49-24-26-5-1-2-6-31(26)44-38(49)54)35(51)43-32(7-3-4-12-41)36(52)47-19-17-46(18-20-47)27-8-13-42-14-9-27/h1-2,5-6,8-9,13-14,21-22,28,32-33,50H,3-4,7,10-12,15-20,23-24,41H2,(H,43,51)(H,44,54)(H,45,53)/t32-,33+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Displacement of [125I]CGRP from CGRP receptor in human SK-N-MC cell membranes preincubated for 30 mins followed by radioligand addition measured afte... |
Bioorg Med Chem Lett 24: 845-9 (2014)
Article DOI: 10.1016/j.bmcl.2013.12.089
BindingDB Entry DOI: 10.7270/Q2FJ2J84 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Somatostatin receptor type 2
(Homo sapiens (Human)) | BDBM85357
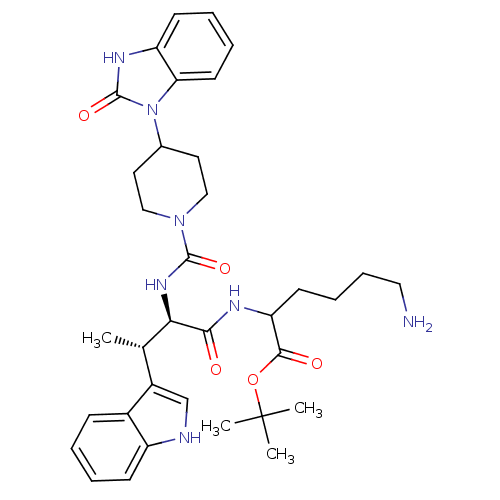
(2-[[(2R,3S)-2-[[4-[(2-Oxo-2,3-dihydro-1H-benzimida...)Show SMILES C[C@H]([C@@H](NC(=O)N1CCC(CC1)n1c2ccccc2[nH]c1=O)C(=O)NC(CCCCN)C(=O)OC(C)(C)C)c1c[nH]c2ccccc12 Show InChI InChI=1S/C35H47N7O5/c1-22(25-21-37-26-12-6-5-11-24(25)26)30(31(43)38-28(14-9-10-18-36)32(44)47-35(2,3)4)40-33(45)41-19-16-23(17-20-41)42-29-15-8-7-13-27(29)39-34(42)46/h5-8,11-13,15,21-23,28,30,37H,9-10,14,16-20,36H2,1-4H3,(H,38,43)(H,39,46)(H,40,45)/t22-,28?,30+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by PDSP Ki Database
| |
Proc Natl Acad Sci U S A 95: 10836-41 (1998)
Article DOI: 10.1073/pnas.95.18.10836
BindingDB Entry DOI: 10.7270/Q2XW4HCM |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50130563

((3R)-7-Hydroxy-N-[(1S)-1-{[(3R,4R)-4-(3-hydroxyphe...)Show SMILES CC(C)[C@@H](CN1CC[C@](C)([C@@H](C)C1)c1cccc(O)c1)NC(=O)[C@H]1Cc2ccc(O)cc2CN1 |r| Show InChI InChI=1S/C28H39N3O3/c1-18(2)26(30-27(34)25-13-20-8-9-24(33)12-21(20)15-29-25)17-31-11-10-28(4,19(3)16-31)22-6-5-7-23(32)14-22/h5-9,12,14,18-19,25-26,29,32-33H,10-11,13,15-17H2,1-4H3,(H,30,34)/t19-,25+,26+,28+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Science and Technology
Curated by ChEMBL
| Assay Description
Antagonist activity at kappa opioid receptor (unknown origin) assessed as stimulation of [35S]GTPgammaS |
Eur J Med Chem 141: 632-647 (2017)
Article DOI: 10.1016/j.ejmech.2017.10.012
BindingDB Entry DOI: 10.7270/Q2V98BQ2 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM50434658

(CHEMBL2387479)Show SMILES F[C@@H]1CNCC[C@H]1CNc1ccn2ncc(C(=O)Nc3c[nH]c4ncccc34)c2n1 |r| Show InChI InChI=1S/C20H21FN8O/c21-15-10-22-6-3-12(15)8-24-17-4-7-29-19(28-17)14(9-26-29)20(30)27-16-11-25-18-13(16)2-1-5-23-18/h1-2,4-5,7,9,11-12,15,22H,3,6,8,10H2,(H,23,25)(H,24,28)(H,27,30)/t12-,15+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of PIM1 (unknown origin) using FAM-pimtide as substrate after 90 mins by spectrophotometry in presence of ATP |
Bioorg Med Chem Lett 23: 3149-53 (2013)
Article DOI: 10.1016/j.bmcl.2013.04.020
BindingDB Entry DOI: 10.7270/Q2PC33S9 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM50434663
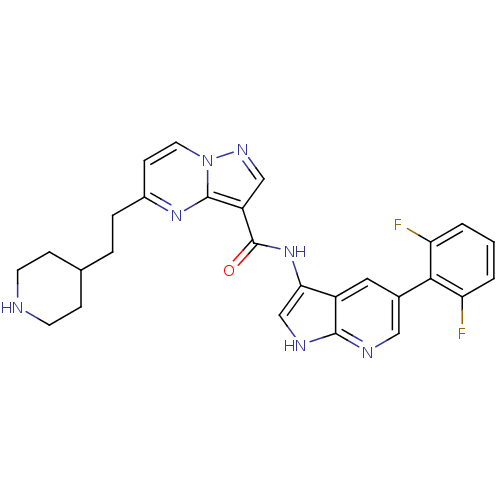
(CHEMBL2387474)Show SMILES Fc1cccc(F)c1-c1cnc2[nH]cc(NC(=O)c3cnn4ccc(CCC5CCNCC5)nc34)c2c1 Show InChI InChI=1S/C27H25F2N7O/c28-21-2-1-3-22(29)24(21)17-12-19-23(15-32-25(19)31-13-17)35-27(37)20-14-33-36-11-8-18(34-26(20)36)5-4-16-6-9-30-10-7-16/h1-3,8,11-16,30H,4-7,9-10H2,(H,31,32)(H,35,37) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of PIM1 (unknown origin) using FAM-pimtide as substrate after 90 mins by spectrophotometry in presence of ATP |
Bioorg Med Chem Lett 23: 3149-53 (2013)
Article DOI: 10.1016/j.bmcl.2013.04.020
BindingDB Entry DOI: 10.7270/Q2PC33S9 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50149477
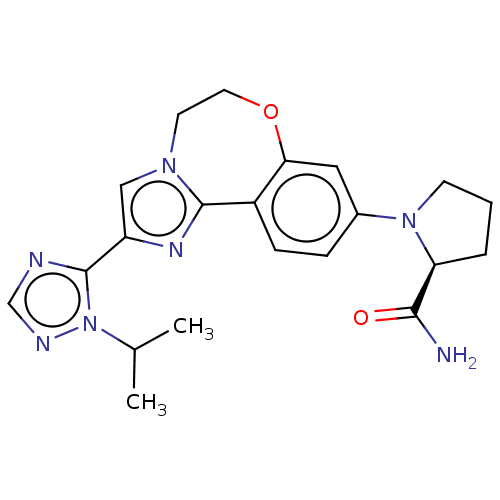
(CHEMBL3770993 | US10851091, U.S. Pat. No. 8,242,10...)Show SMILES CC(C)n1ncnc1-c1cn2CCOc3cc(ccc3-c2n1)N1CCC[C@H]1C(N)=O |r| Show InChI InChI=1S/C21H25N7O2/c1-13(2)28-21(23-12-24-28)16-11-26-8-9-30-18-10-14(5-6-15(18)20(26)25-16)27-7-3-4-17(27)19(22)29/h5-6,10-13,17H,3-4,7-9H2,1-2H3,(H2,22,29)/t17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech, Inc.
US Patent
| Assay Description
The biochemical inhibition of four PI3K isoforms by the Formula I compounds of Table 1. In addition, two clinically tested PI3K compounds, taselisib ... |
US Patent US10851091 (2020)
BindingDB Entry DOI: 10.7270/Q2WS8X9V |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha
(Homo sapiens (Human)) | BDBM50149477

(CHEMBL3770993 | US10851091, U.S. Pat. No. 8,242,10...)Show SMILES CC(C)n1ncnc1-c1cn2CCOc3cc(ccc3-c2n1)N1CCC[C@H]1C(N)=O |r| Show InChI InChI=1S/C21H25N7O2/c1-13(2)28-21(23-12-24-28)16-11-26-8-9-30-18-10-14(5-6-15(18)20(26)25-16)27-7-3-4-17(27)19(22)29/h5-6,10-13,17H,3-4,7-9H2,1-2H3,(H2,22,29)/t17-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech, Inc.
US Patent
| Assay Description
PI3K Binding assays are intended for determining the biochemical potency of small molecule PI3K inhibitors. The PI3K lipid kinase reaction is perform... |
US Patent US10112932 (2018)
BindingDB Entry DOI: 10.7270/Q2JQ132B |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM50434657

(CHEMBL2387462)Show SMILES FC1(CNc2ccn3ncc(C(=O)Nc4c[nH]c5ncccc45)c3n2)CCNCC1 Show InChI InChI=1S/C20H21FN8O/c21-20(4-7-22-8-5-20)12-25-16-3-9-29-18(28-16)14(10-26-29)19(30)27-15-11-24-17-13(15)2-1-6-23-17/h1-3,6,9-11,22H,4-5,7-8,12H2,(H,23,24)(H,25,28)(H,27,30) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of PIM1 (unknown origin) using FAM-pimtide as substrate after 90 mins by spectrophotometry in presence of ATP |
Bioorg Med Chem Lett 23: 3149-53 (2013)
Article DOI: 10.1016/j.bmcl.2013.04.020
BindingDB Entry DOI: 10.7270/Q2PC33S9 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM207378
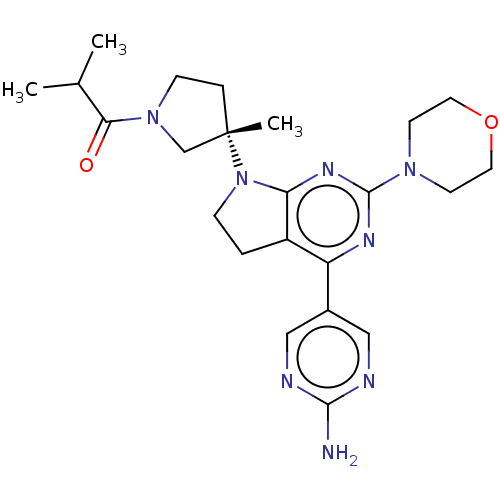
(US9260439, 262)Show SMILES CC(C)C(=O)N1CC[C@@](C)(C1)N1CCc2c1nc(nc2-c1cnc(N)nc1)N1CCOCC1 Show InChI InChI=1S/C23H32N8O2/c1-15(2)20(32)30-7-5-23(3,14-30)31-6-4-17-18(16-12-25-21(24)26-13-16)27-22(28-19(17)31)29-8-10-33-11-9-29/h12-13,15H,4-11,14H2,1-3H3,(H2,24,25,26)/t23-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length PI3K p110alpha/p85alpha (322 to 600) expressed in baculovirus infected Sf21 cells using phosphatidylinosi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01652
BindingDB Entry DOI: 10.7270/Q2TH8RC9 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM207196
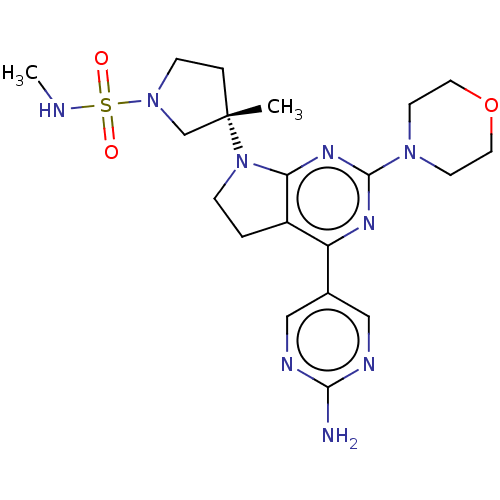
(US9260439, 173)Show SMILES CNS(=O)(=O)N1CC[C@@](C)(C1)N1CCc2c1nc(nc2-c1cnc(N)nc1)N1CCOCC1 |r| Show InChI InChI=1S/C20H29N9O3S/c1-20(4-6-28(13-20)33(30,31)22-2)29-5-3-15-16(14-11-23-18(21)24-12-14)25-19(26-17(15)29)27-7-9-32-10-8-27/h11-12,22H,3-10,13H2,1-2H3,(H2,21,23,24)/t20-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length PI3K p110alpha/p85alpha (322 to 600) expressed in baculovirus infected Sf21 cells using phosphatidylinosi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01652
BindingDB Entry DOI: 10.7270/Q2TH8RC9 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM207217
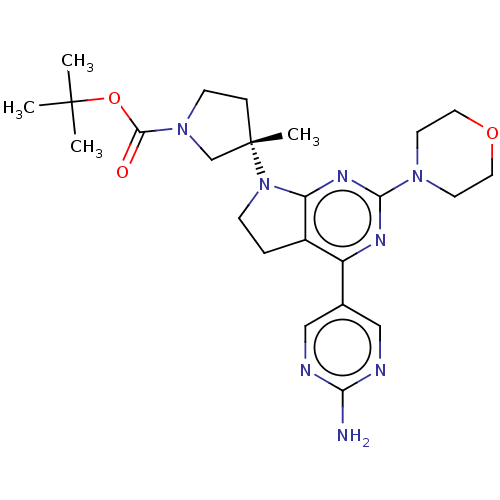
(US9260439, 194 | US9260439, 238 | US9260439, 239)Show SMILES CC(C)(C)OC(=O)N1CC[C@@](C)(C1)N1CCc2c1nc(nc2-c1cnc(N)nc1)N1CCOCC1 |r| Show InChI InChI=1S/C24H34N8O3/c1-23(2,3)35-22(33)31-8-6-24(4,15-31)32-7-5-17-18(16-13-26-20(25)27-14-16)28-21(29-19(17)32)30-9-11-34-12-10-30/h13-14H,5-12,15H2,1-4H3,(H2,25,26,27)/t24-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| <0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length PI3K p110alpha/p85alpha (322 to 600) expressed in baculovirus infected Sf21 cells using phosphatidylinosi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01652
BindingDB Entry DOI: 10.7270/Q2TH8RC9 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM340384
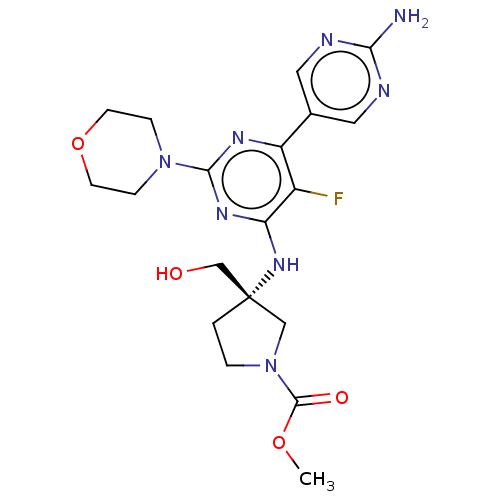
(US9758538, Example 72)Show SMILES COC(=O)N1CC[C@@](CO)(C1)Nc1nc(nc(c1F)-c1cnc(N)nc1)N1CCOCC1 |r| Show InChI InChI=1S/C19H25FN8O4/c1-31-18(30)28-3-2-19(10-28,11-29)26-15-13(20)14(12-8-22-16(21)23-9-12)24-17(25-15)27-4-6-32-7-5-27/h8-9,29H,2-7,10-11H2,1H3,(H2,21,22,23)(H,24,25,26)/t19-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length PI3K p110alpha/p85alpha (322 to 600) expressed in baculovirus infected Sf21 cells using phosphatidylinosi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01652
BindingDB Entry DOI: 10.7270/Q2TH8RC9 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM248955

(US9434725, 186)Show SMILES COc1ccc(nc1N1CCC[C@H](N)C1)-n1ncc2cnc(cc12)-c1cncc(C)n1 |r| Show InChI InChI=1S/C22H24N8O/c1-14-9-24-12-18(27-14)17-8-19-15(10-25-17)11-26-30(19)21-6-5-20(31-2)22(28-21)29-7-3-4-16(23)13-29/h5-6,8-12,16H,3-4,7,13,23H2,1-2H3/t16-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of PIM1 (unknown origin) using 5FAM-ARKRRRHPSGPPTA as substrate after 90 mins in presence of ATP by caliper microfluidic mobility shift as... |
J Med Chem 60: 4458-4473 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00418
BindingDB Entry DOI: 10.7270/Q2H997PN |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM340336
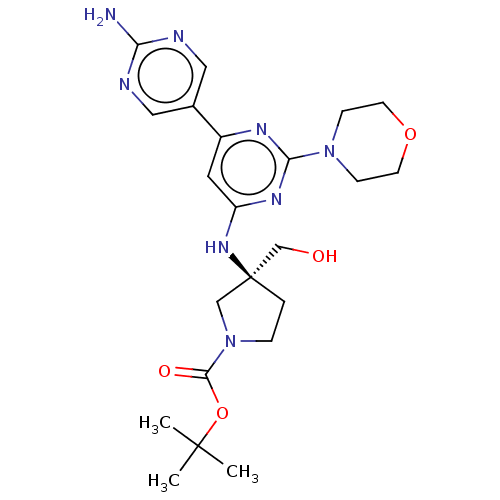
(US9758538, Example 24)Show SMILES CC(C)(C)OC(=O)N1CC[C@@](CO)(C1)Nc1cc(nc(n1)N1CCOCC1)-c1cnc(N)nc1 |r| Show InChI InChI=1S/C22H32N8O4/c1-21(2,3)34-20(32)30-5-4-22(13-30,14-31)28-17-10-16(15-11-24-18(23)25-12-15)26-19(27-17)29-6-8-33-9-7-29/h10-12,31H,4-9,13-14H2,1-3H3,(H2,23,24,25)(H,26,27,28)/t22-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| <0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length PI3K p110alpha/p85alpha (322 to 600) expressed in baculovirus infected Sf21 cells using phosphatidylinosi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01652
BindingDB Entry DOI: 10.7270/Q2TH8RC9 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM340314
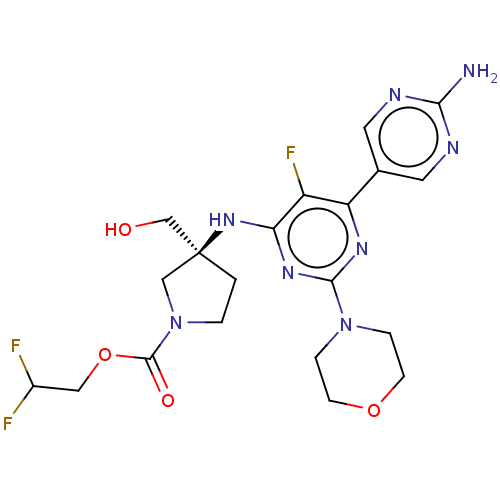
((Scheme A): Preparation of 2,2-difluoroethyl (3S)-...)Show SMILES Nc1ncc(cn1)-c1nc(nc(N[C@@]2(CO)CCN(C2)C(=O)OCC(F)F)c1F)N1CCOCC1 |r| Show InChI InChI=1S/C20H25F3N8O4/c21-13(22)9-35-19(33)31-2-1-20(10-31,11-32)29-16-14(23)15(12-7-25-17(24)26-8-12)27-18(28-16)30-3-5-34-6-4-30/h7-8,13,32H,1-6,9-11H2,(H2,24,25,26)(H,27,28,29)/t20-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| <0.0180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length PI3K p110alpha/p85alpha (322 to 600) expressed in baculovirus infected Sf21 cells using phosphatidylinosi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01652
BindingDB Entry DOI: 10.7270/Q2TH8RC9 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase ABL1
(Homo sapiens (Human)) | BDBM112421

(US8623889, 420)Show SMILES Cc1c(F)cncc1-c1ccc2cc(NC(=O)[C@@H]3C[C@@H]3F)ncc2c1 |r| Show InChI InChI=1S/C19H15F2N3O/c1-10-15(8-22-9-17(10)21)12-3-2-11-5-18(23-7-13(11)4-12)24-19(25)14-6-16(14)20/h2-5,7-9,14,16H,6H2,1H3,(H,23,24,25)/t14-,16+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| 0.0186 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech, Inc.
US Patent
| Assay Description
Using the following procedure, varying concentration of compounds of the invention were assessed for their ability to inhibit c-Abl enzyme's phos... |
US Patent US8623889 (2014)
BindingDB Entry DOI: 10.7270/Q2JQ0ZNX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 3-kinase regulatory subunit alpha/4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM340346

(US9758538, Example 34)Show SMILES COC(=O)N1CC[C@@](CO)(C1)Nc1cc(nc(n1)N1CCOCC1)-c1cnc(N)nc1 |r| Show InChI InChI=1S/C19H26N8O4/c1-30-18(29)27-3-2-19(11-27,12-28)25-15-8-14(13-9-21-16(20)22-10-13)23-17(24-15)26-4-6-31-7-5-26/h8-10,28H,2-7,11-12H2,1H3,(H2,20,21,22)(H,23,24,25)/t19-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of recombinant human full-length PI3K p110alpha/p85alpha (322 to 600) expressed in baculovirus infected Sf21 cells using phosphatidylinosi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01652
BindingDB Entry DOI: 10.7270/Q2TH8RC9 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data