 Found 908 hits with Last Name = 'lind' and Initial = 'p'
Found 908 hits with Last Name = 'lind' and Initial = 'p' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590085
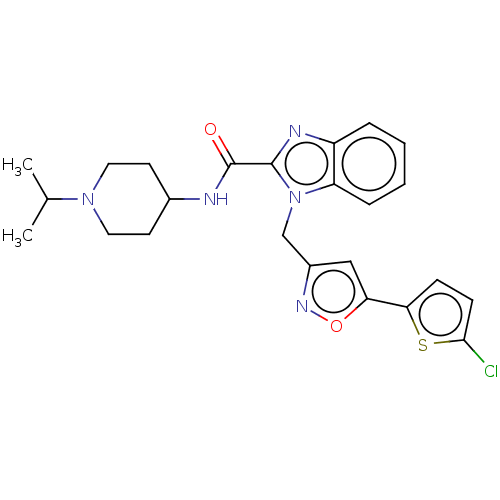
(CHEMBL5187102)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1nc2ccccc2n1Cc1cc(on1)-c1ccc(Cl)s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177012
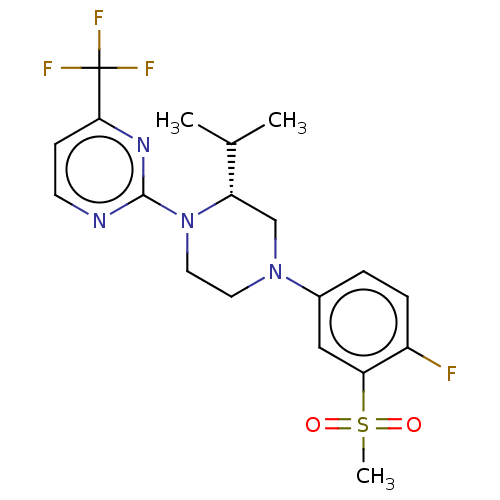
(CHEMBL3814153 | US10144715, Compound 7-32)Show SMILES CC(C)[C@@H]1CN(CCN1c1nccc(n1)C(F)(F)F)c1ccc(F)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C19H22F4N4O2S/c1-12(2)15-11-26(13-4-5-14(20)16(10-13)30(3,28)29)8-9-27(15)18-24-7-6-17(25-18)19(21,22)23/h4-7,10,12,15H,8-9,11H2,1-3H3/t15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177016
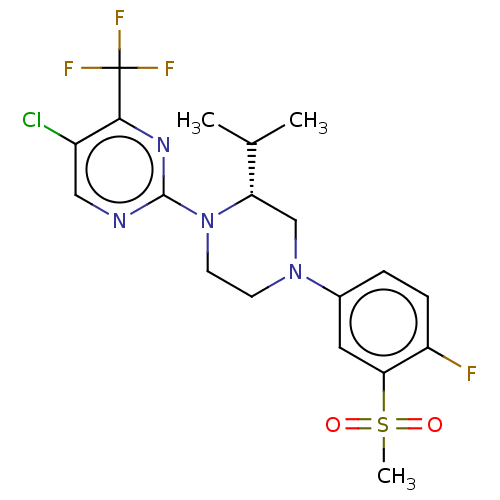
(CHEMBL3814501)Show SMILES CC(C)[C@@H]1CN(CCN1c1ncc(Cl)c(n1)C(F)(F)F)c1ccc(F)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C19H21ClF4N4O2S/c1-11(2)15-10-27(12-4-5-14(21)16(8-12)31(3,29)30)6-7-28(15)18-25-9-13(20)17(26-18)19(22,23)24/h4-5,8-9,11,15H,6-7,10H2,1-3H3/t15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177010
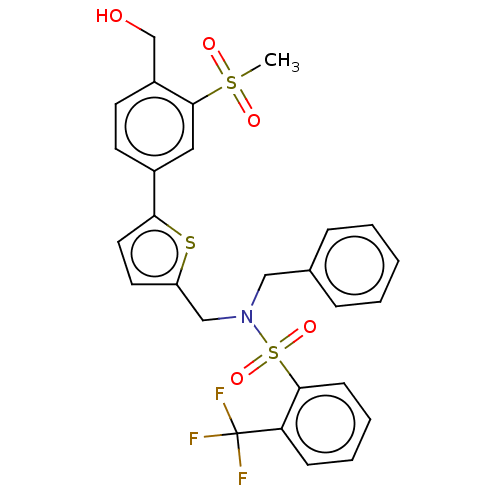
(CHEMBL3814006)Show SMILES CS(=O)(=O)c1cc(ccc1CO)-c1ccc(CN(Cc2ccccc2)S(=O)(=O)c2ccccc2C(F)(F)F)s1 Show InChI InChI=1S/C27H24F3NO5S3/c1-38(33,34)26-15-20(11-12-21(26)18-32)24-14-13-22(37-24)17-31(16-19-7-3-2-4-8-19)39(35,36)25-10-6-5-9-23(25)27(28,29)30/h2-15,32H,16-18H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177015
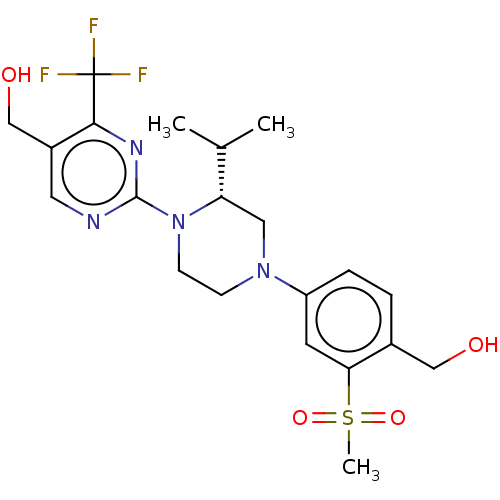
(CHEMBL3814206 | US10144715, Compound 19-1)Show SMILES CC(C)[C@@H]1CN(CCN1c1ncc(CO)c(n1)C(F)(F)F)c1ccc(CO)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C21H27F3N4O4S/c1-13(2)17-10-27(16-5-4-14(11-29)18(8-16)33(3,31)32)6-7-28(17)20-25-9-15(12-30)19(26-20)21(22,23)24/h4-5,8-9,13,17,29-30H,6-7,10-12H2,1-3H3/t17-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50177016

(CHEMBL3814501)Show SMILES CC(C)[C@@H]1CN(CCN1c1ncc(Cl)c(n1)C(F)(F)F)c1ccc(F)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C19H21ClF4N4O2S/c1-11(2)15-10-27(12-4-5-14(21)16(8-12)31(3,29)30)6-7-28(15)18-25-9-13(20)17(26-18)19(22,23)24/h4-5,8-9,11,15H,6-7,10H2,1-3H3/t15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12400
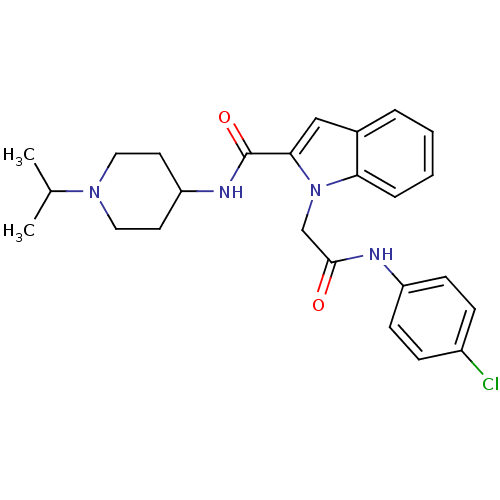
(1-[(4-Chloro-phenylcarbamoyl)-methyl]-1H-indole-2-...)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2ccccc2n1CC(=O)Nc1ccc(Cl)cc1 Show InChI InChI=1S/C25H29ClN4O2/c1-17(2)29-13-11-21(12-14-29)28-25(32)23-15-18-5-3-4-6-22(18)30(23)16-24(31)27-20-9-7-19(26)8-10-20/h3-10,15,17,21H,11-14,16H2,1-2H3,(H,27,31)(H,28,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM12372
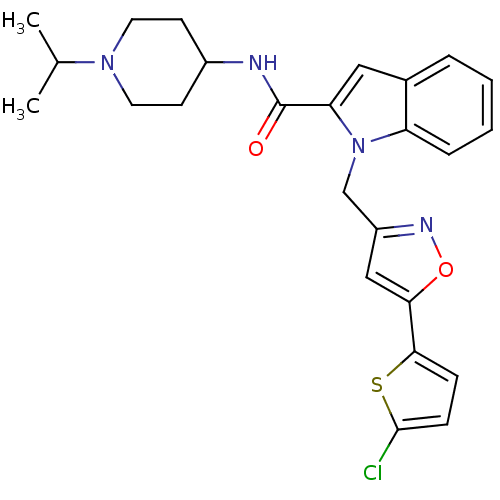
(1-{[5-(5-chlorothiophen-2-yl)-1,2-oxazol-3-yl]meth...)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2ccccc2n1Cc1cc(on1)-c1ccc(Cl)s1 Show InChI InChI=1S/C25H27ClN4O2S/c1-16(2)29-11-9-18(10-12-29)27-25(31)21-13-17-5-3-4-6-20(17)30(21)15-19-14-22(32-28-19)23-7-8-24(26)33-23/h3-8,13-14,16,18H,9-12,15H2,1-2H3,(H,27,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590072

(CHEMBL3904655)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2ccccc2n1CC(=O)Nc1ccc(Br)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177011
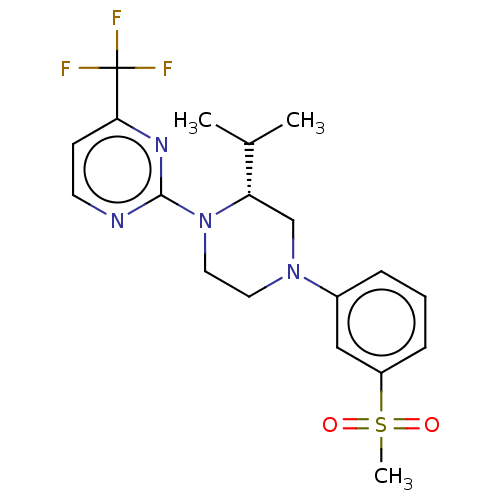
(CHEMBL3815014 | US10144715, Compound 7-13)Show SMILES CC(C)[C@@H]1CN(CCN1c1nccc(n1)C(F)(F)F)c1cccc(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C19H23F3N4O2S/c1-13(2)16-12-25(14-5-4-6-15(11-14)29(3,27)28)9-10-26(16)18-23-8-7-17(24-18)19(20,21)22/h4-8,11,13,16H,9-10,12H2,1-3H3/t16-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590086
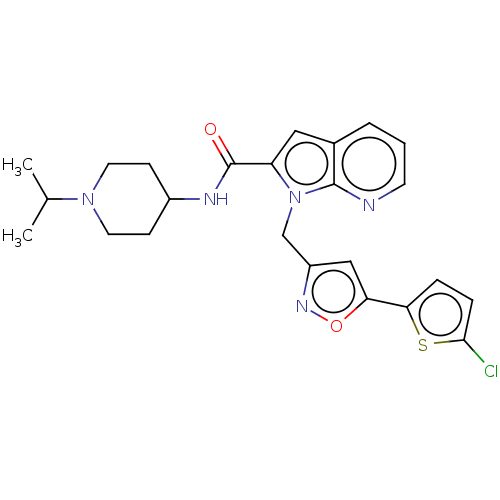
(CHEMBL5192536)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2cccnc2n1Cc1cc(on1)-c1ccc(Cl)s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590082
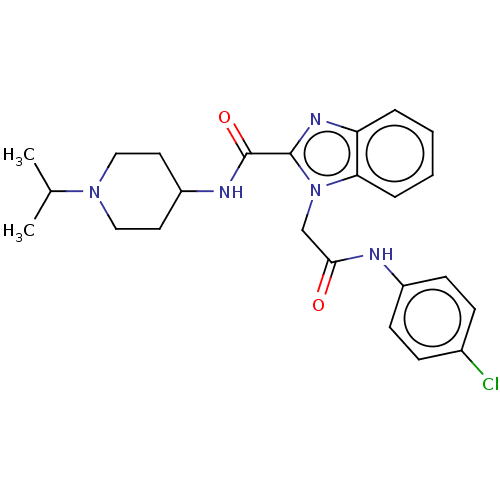
(CHEMBL5175184)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1nc2ccccc2n1CC(=O)Nc1ccc(Cl)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590073
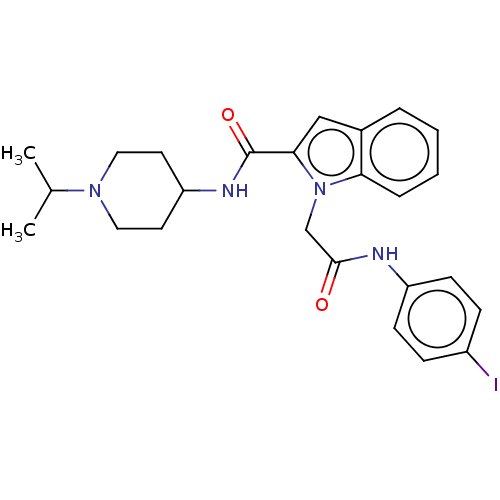
(CHEMBL5175600)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2ccccc2n1CC(=O)Nc1ccc(I)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177018
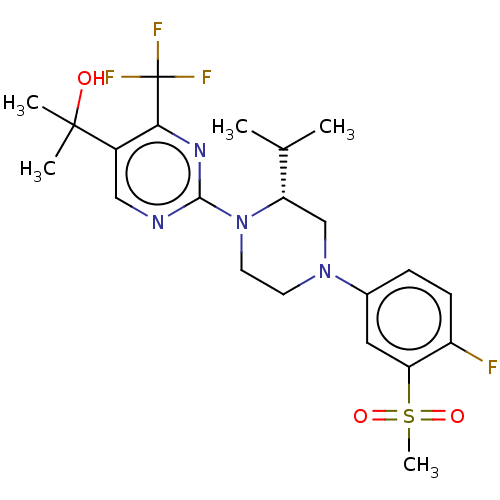
(CHEMBL3814478 | US10144715, Compound 14-1)Show SMILES CC(C)[C@@H]1CN(CCN1c1ncc(c(n1)C(F)(F)F)C(C)(C)O)c1ccc(F)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C22H28F4N4O3S/c1-13(2)17-12-29(14-6-7-16(23)18(10-14)34(5,32)33)8-9-30(17)20-27-11-15(21(3,4)31)19(28-20)22(24,25)26/h6-7,10-11,13,17,31H,8-9,12H2,1-5H3/t17-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590087
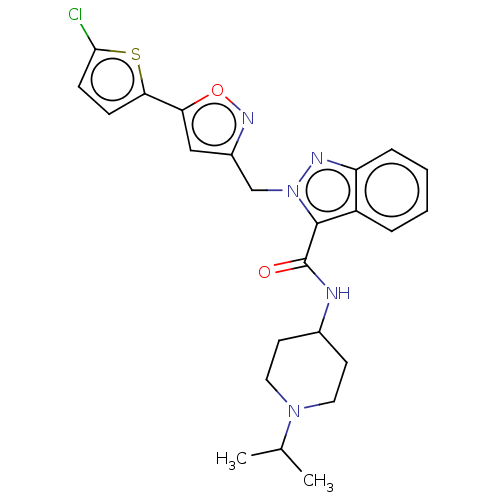
(CHEMBL5179837)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1n(Cc2cc(on2)-c2ccc(Cl)s2)nc2ccccc12 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177017
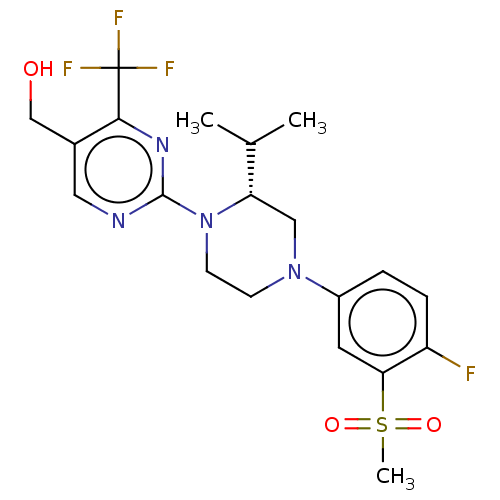
(CHEMBL3815001 | US10144715, Compound 11-1)Show SMILES CC(C)[C@@H]1CN(CCN1c1ncc(CO)c(n1)C(F)(F)F)c1ccc(F)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C20H24F4N4O3S/c1-12(2)16-10-27(14-4-5-15(21)17(8-14)32(3,30)31)6-7-28(16)19-25-9-13(11-29)18(26-19)20(22,23)24/h4-5,8-9,12,16,29H,6-7,10-11H2,1-3H3/t16-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590097

(CHEMBL5187449)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1nn(Cc2cc(on2)-c2ccc(Cl)s2)c2ccccc12 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590083
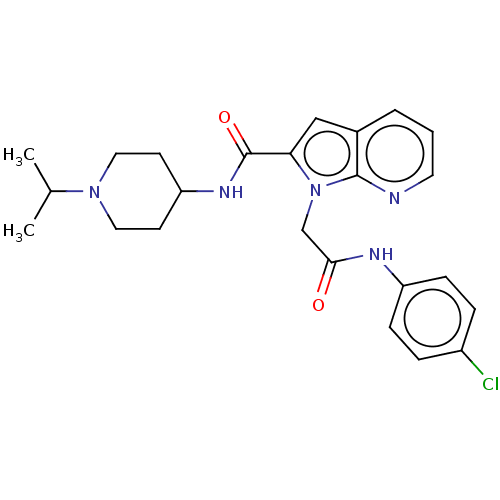
(CHEMBL5192808)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2cccnc2n1CC(=O)Nc1ccc(Cl)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590078
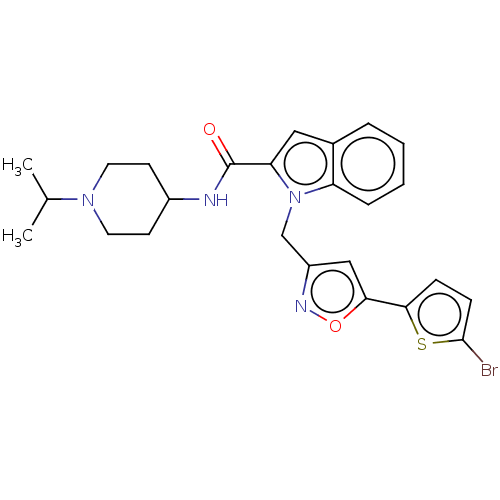
(CHEMBL5175799)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2ccccc2n1Cc1cc(on1)-c1ccc(Br)s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590081
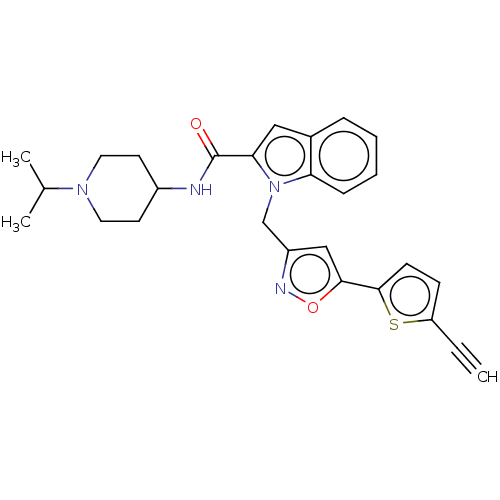
(CHEMBL5180386)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2ccccc2n1Cc1cc(on1)-c1ccc(s1)C#C | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50177010

(CHEMBL3814006)Show SMILES CS(=O)(=O)c1cc(ccc1CO)-c1ccc(CN(Cc2ccccc2)S(=O)(=O)c2ccccc2C(F)(F)F)s1 Show InChI InChI=1S/C27H24F3NO5S3/c1-38(33,34)26-15-20(11-12-21(26)18-32)24-14-13-22(37-24)17-31(16-19-7-3-2-4-8-19)39(35,36)25-10-6-5-9-23(25)27(28,29)30/h2-15,32H,16-18H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50177012

(CHEMBL3814153 | US10144715, Compound 7-32)Show SMILES CC(C)[C@@H]1CN(CCN1c1nccc(n1)C(F)(F)F)c1ccc(F)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C19H22F4N4O2S/c1-12(2)15-11-26(13-4-5-14(20)16(10-13)30(3,28)29)8-9-27(15)18-24-7-6-17(25-18)19(21,22)23/h4-7,10,12,15H,8-9,11H2,1-3H3/t15-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590099
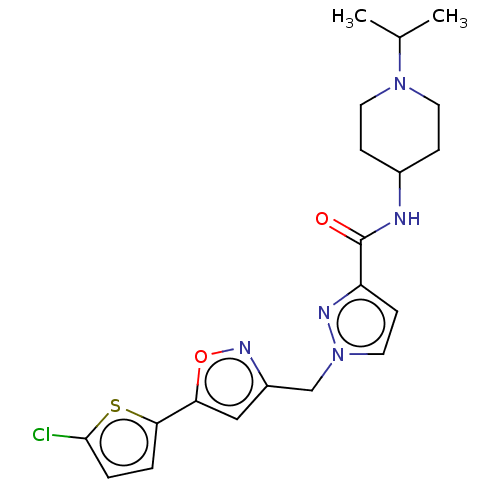
(CHEMBL5193338)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1ccn(Cc2cc(on2)-c2ccc(Cl)s2)n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50177011

(CHEMBL3815014 | US10144715, Compound 7-13)Show SMILES CC(C)[C@@H]1CN(CCN1c1nccc(n1)C(F)(F)F)c1cccc(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C19H23F3N4O2S/c1-13(2)16-12-25(14-5-4-6-15(11-14)29(3,27)28)9-10-26(16)18-23-8-7-17(24-18)19(20,21)22/h4-8,11,13,16H,9-10,12H2,1-3H3/t16-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 43 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590075
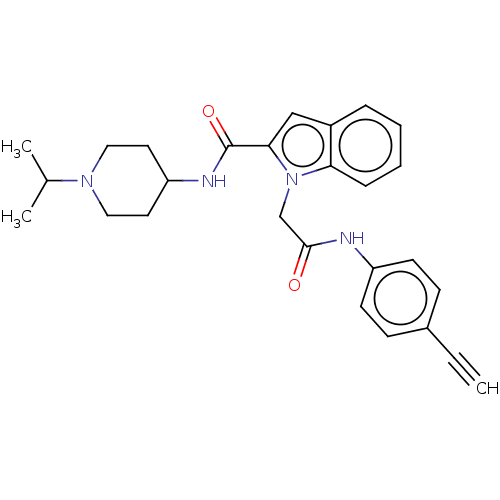
(CHEMBL5190215)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2ccccc2n1CC(=O)Nc1ccc(cc1)C#C | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 49 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590084
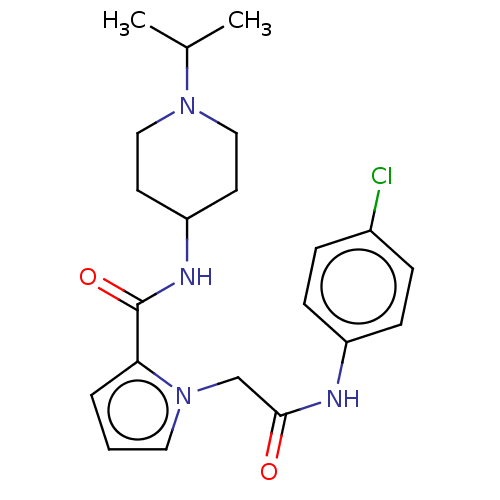
(CHEMBL5208176)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cccn1CC(=O)Nc1ccc(Cl)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 51 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590089

(CHEMBL5181980)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1ccnn1Cc1cc(on1)-c1ccc(Cl)s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 52 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590080
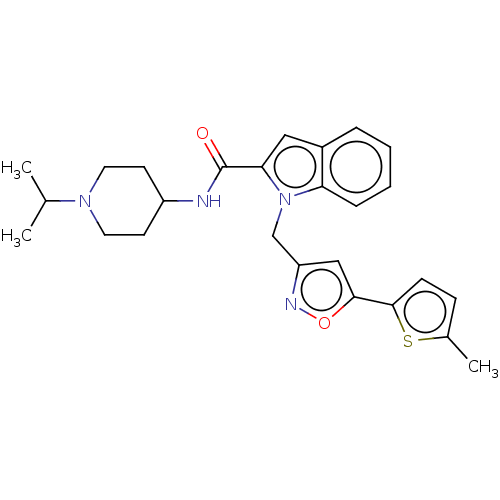
(CHEMBL5185736)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2ccccc2n1Cc1cc(on1)-c1ccc(C)s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590074
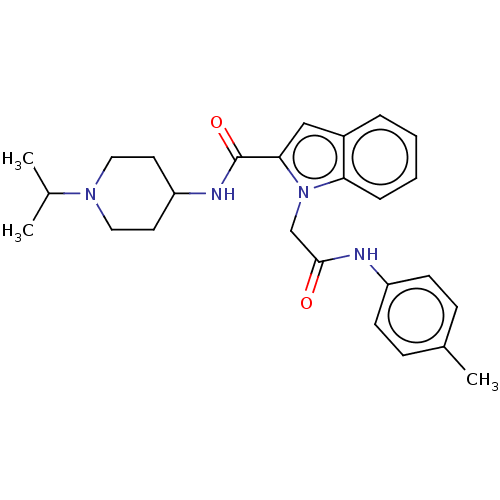
(CHEMBL5187415)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2ccccc2n1CC(=O)Nc1ccc(C)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 56 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590088

(CHEMBL5192209)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cccn1Cc1cc(on1)-c1ccc(Cl)s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 57 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590071
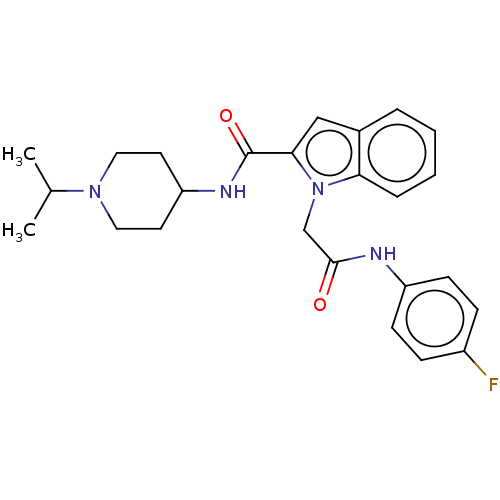
(CHEMBL5178354)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2ccccc2n1CC(=O)Nc1ccc(F)cc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 63 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590096
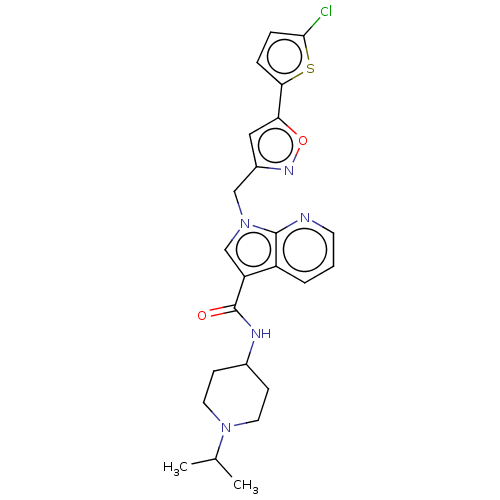
(CHEMBL5173515)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cn(Cc2cc(on2)-c2ccc(Cl)s2)c2ncccc12 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 65 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590077
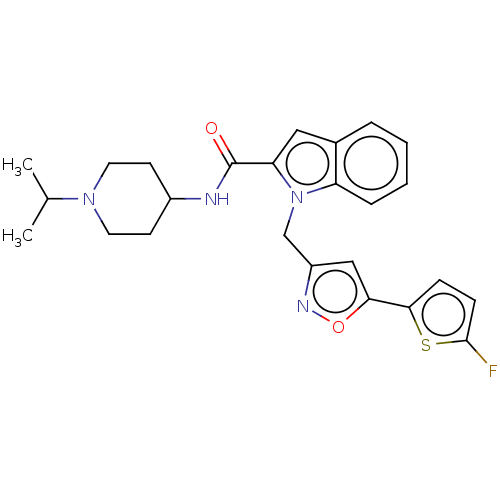
(CHEMBL5188689)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2ccccc2n1Cc1cc(on1)-c1ccc(F)s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50177015

(CHEMBL3814206 | US10144715, Compound 19-1)Show SMILES CC(C)[C@@H]1CN(CCN1c1ncc(CO)c(n1)C(F)(F)F)c1ccc(CO)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C21H27F3N4O4S/c1-13(2)17-10-27(16-5-4-14(11-29)18(8-16)33(3,31)32)6-7-28(17)20-25-9-15(12-30)19(26-20)21(22,23)24/h4-5,8-9,13,17,29-30H,6-7,10-12H2,1-3H3/t17-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 81 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177014
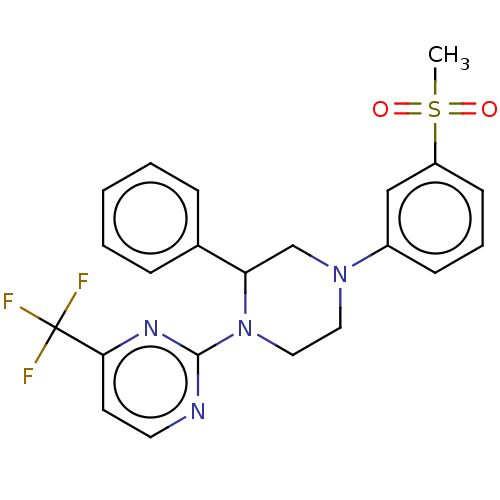
(CHEMBL3813875)Show SMILES CS(=O)(=O)c1cccc(c1)N1CCN(C(C1)c1ccccc1)c1nccc(n1)C(F)(F)F Show InChI InChI=1S/C22H21F3N4O2S/c1-32(30,31)18-9-5-8-17(14-18)28-12-13-29(19(15-28)16-6-3-2-4-7-16)21-26-11-10-20(27-21)22(23,24)25/h2-11,14,19H,12-13,15H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 94 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177014

(CHEMBL3813875)Show SMILES CS(=O)(=O)c1cccc(c1)N1CCN(C(C1)c1ccccc1)c1nccc(n1)C(F)(F)F Show InChI InChI=1S/C22H21F3N4O2S/c1-32(30,31)18-9-5-8-17(14-18)28-12-13-29(19(15-28)16-6-3-2-4-7-16)21-26-11-10-20(27-21)22(23,24)25/h2-11,14,19H,12-13,15H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 94 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50177017

(CHEMBL3815001 | US10144715, Compound 11-1)Show SMILES CC(C)[C@@H]1CN(CCN1c1ncc(CO)c(n1)C(F)(F)F)c1ccc(F)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C20H24F4N4O3S/c1-12(2)16-10-27(14-4-5-15(21)17(8-14)32(3,30)31)6-7-28(16)19-25-9-13(11-29)18(26-19)20(22,23)24/h4-5,8-9,12,16,29H,6-7,10-11H2,1-3H3/t16-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 146 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50177018

(CHEMBL3814478 | US10144715, Compound 14-1)Show SMILES CC(C)[C@@H]1CN(CCN1c1ncc(c(n1)C(F)(F)F)C(C)(C)O)c1ccc(F)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C22H28F4N4O3S/c1-13(2)17-12-29(14-6-7-16(23)18(10-14)34(5,32)33)8-9-30(17)20-27-11-15(21(3,4)31)19(28-20)22(24,25)26/h6-7,10-11,13,17,31H,8-9,12H2,1-5H3/t17-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 157 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177019
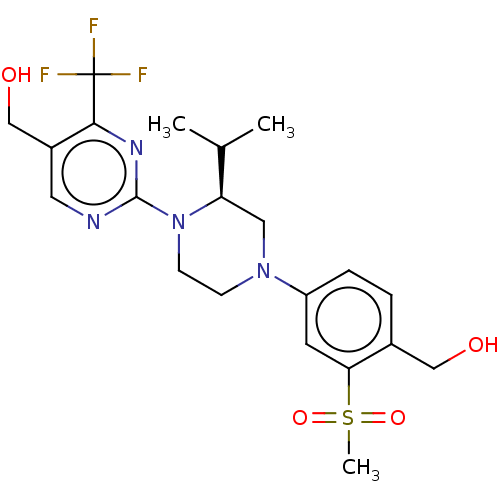
(CHEMBL3813863 | US10144715, Compound 19-2)Show SMILES CC(C)[C@H]1CN(CCN1c1ncc(CO)c(n1)C(F)(F)F)c1ccc(CO)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C21H27F3N4O4S/c1-13(2)17-10-27(16-5-4-14(11-29)18(8-16)33(3,31)32)6-7-28(17)20-25-9-15(12-30)19(26-20)21(22,23)24/h4-5,8-9,13,17,29-30H,6-7,10-12H2,1-3H3/t17-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590076
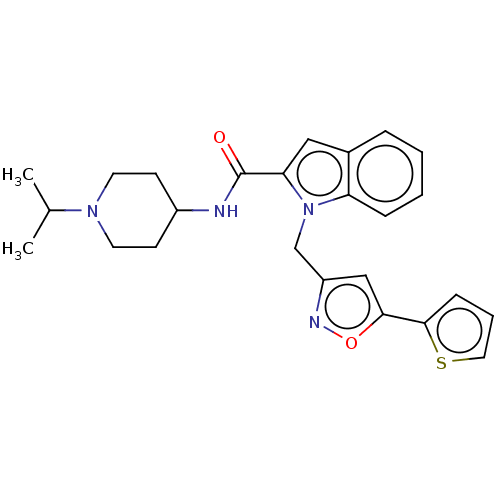
(CHEMBL5204725)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2ccccc2n1Cc1cc(on1)-c1cccs1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 162 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590079
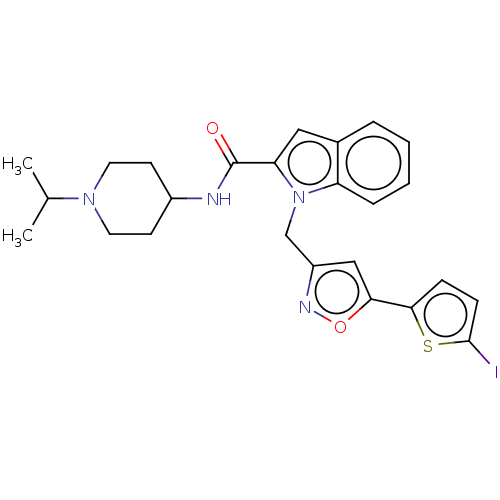
(CHEMBL5193560)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2ccccc2n1Cc1cc(on1)-c1ccc(I)s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 188 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590070
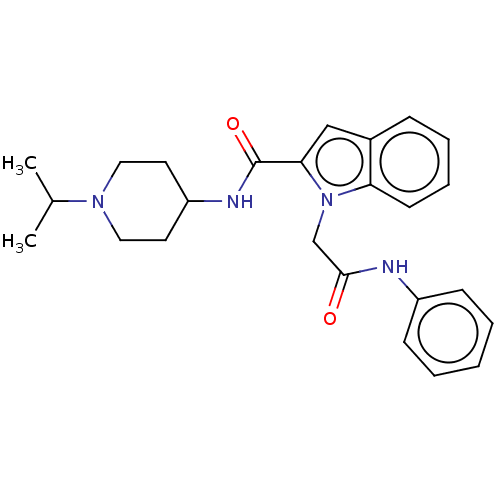
(CHEMBL5206688)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cc2ccccc2n1CC(=O)Nc1ccccc1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 204 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590093

(CHEMBL5186260)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1ccn(CC(=O)Nc2ccc(Cl)cc2)n1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 204 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590095
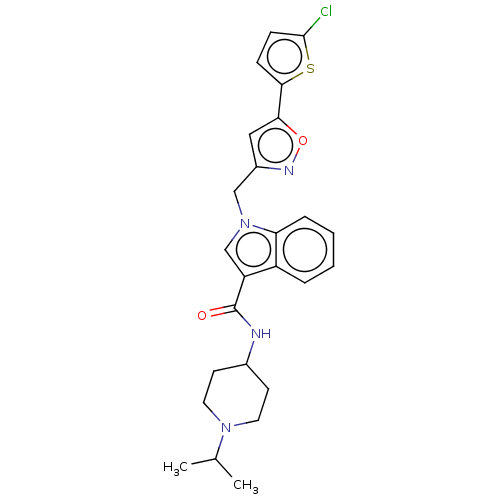
(CHEMBL5199539)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1cn(Cc2cc(on2)-c2ccc(Cl)s2)c2ccccc12 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 241 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50177019

(CHEMBL3813863 | US10144715, Compound 19-2)Show SMILES CC(C)[C@H]1CN(CCN1c1ncc(CO)c(n1)C(F)(F)F)c1ccc(CO)c(c1)S(C)(=O)=O |r| Show InChI InChI=1S/C21H27F3N4O4S/c1-13(2)17-10-27(16-5-4-14(11-29)18(8-16)33(3,31)32)6-7-28(17)20-25-9-15(12-30)19(26-20)21(22,23)24/h4-5,8-9,13,17,29-30H,6-7,10-12H2,1-3H3/t17-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.07E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50177014

(CHEMBL3813875)Show SMILES CS(=O)(=O)c1cccc(c1)N1CCN(C(C1)c1ccccc1)c1nccc(n1)C(F)(F)F Show InChI InChI=1S/C22H21F3N4O2S/c1-32(30,31)18-9-5-8-17(14-18)28-12-13-29(19(15-28)16-6-3-2-4-7-16)21-26-11-10-20(27-21)22(23,24)25/h2-11,14,19H,12-13,15H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-alpha
(Homo sapiens (Human)) | BDBM50177014

(CHEMBL3813875)Show SMILES CS(=O)(=O)c1cccc(c1)N1CCN(C(C1)c1ccccc1)c1nccc(n1)C(F)(F)F Show InChI InChI=1S/C22H21F3N4O2S/c1-32(30,31)18-9-5-8-17(14-18)28-12-13-29(19(15-28)16-6-3-2-4-7-16)21-26-11-10-20(27-21)22(23,24)25/h2-11,14,19H,12-13,15H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50590098
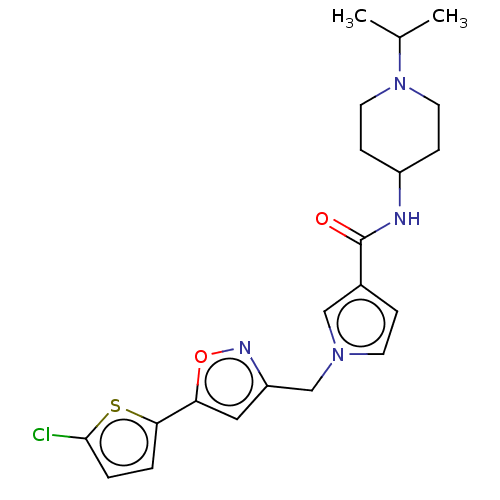
(CHEMBL5194849)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1ccn(Cc2cc(on2)-c2ccc(Cl)s2)c1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.24E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00865
BindingDB Entry DOI: 10.7270/Q2BP06RZ |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177013
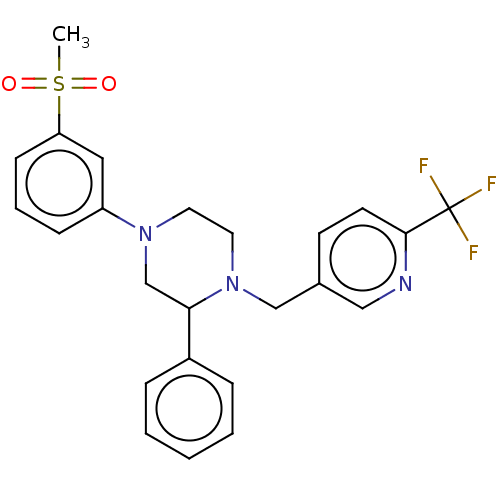
(CHEMBL3814372)Show SMILES CS(=O)(=O)c1cccc(c1)N1CCN(Cc2ccc(nc2)C(F)(F)F)C(C1)c1ccccc1 Show InChI InChI=1S/C24H24F3N3O2S/c1-33(31,32)21-9-5-8-20(14-21)29-12-13-30(22(17-29)19-6-3-2-4-7-19)16-18-10-11-23(28-15-18)24(25,26)27/h2-11,14-15,22H,12-13,16-17H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.27E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |
Oxysterols receptor LXR-beta
(Homo sapiens (Human)) | BDBM50177013

(CHEMBL3814372)Show SMILES CS(=O)(=O)c1cccc(c1)N1CCN(Cc2ccc(nc2)C(F)(F)F)C(C1)c1ccccc1 Show InChI InChI=1S/C24H24F3N3O2S/c1-33(31,32)21-9-5-8-20(14-21)29-12-13-30(22(17-29)19-6-3-2-4-7-19)16-18-10-11-23(28-15-18)24(25,26)27/h2-11,14-15,22H,12-13,16-17H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.28E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vitae Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation counting |
J Med Chem 59: 3264-71 (2016)
Article DOI: 10.1021/acs.jmedchem.5b02029
BindingDB Entry DOI: 10.7270/Q2XP76V7 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data