 Found 170 hits with Last Name = 'shim' and Initial = 'js'
Found 170 hits with Last Name = 'shim' and Initial = 'js' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Tyrosine-protein phosphatase non-receptor type 11
(Homo sapiens (Human)) | BDBM50112356
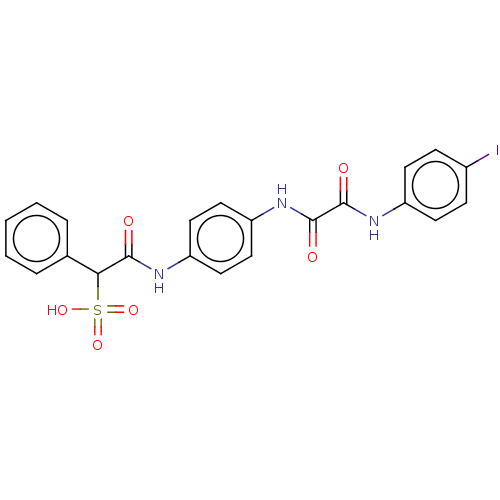
(CHEMBL3609373)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(I)cc2)cc1)c1ccccc1 Show InChI InChI=1S/C22H18IN3O6S/c23-15-6-8-16(9-7-15)25-21(28)22(29)26-18-12-10-17(11-13-18)24-20(27)19(33(30,31)32)14-4-2-1-3-5-14/h1-13,19H,(H,24,27)(H,25,28)(H,26,29)(H,30,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Competitive inhibition of phosphatase activity of SHP2 (unknown origin) using pNPP as a substrate Lineweaver-Burk plot analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Aminopeptidase N
(Homo sapiens (Human)) | BDBM50157325
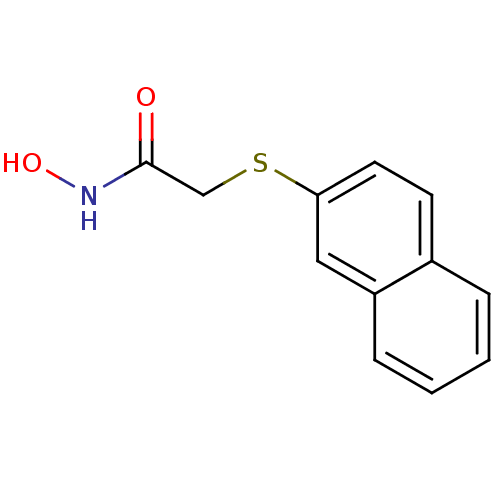
(CHEMBL182879 | N-Hydroxy-2-(naphthalen-2-ylsulfany...)Show InChI InChI=1S/C12H11NO2S/c14-12(13-15)8-16-11-6-5-9-3-1-2-4-10(9)7-11/h1-7,15H,8H2,(H,13,14) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 3.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sejong University
Curated by ChEMBL
| Assay Description
Binding affinity towards aminopeptidase N |
Bioorg Med Chem Lett 15: 181-3 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.010
BindingDB Entry DOI: 10.7270/Q2BG2PRF |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 11
(Homo sapiens (Human)) | BDBM50420258
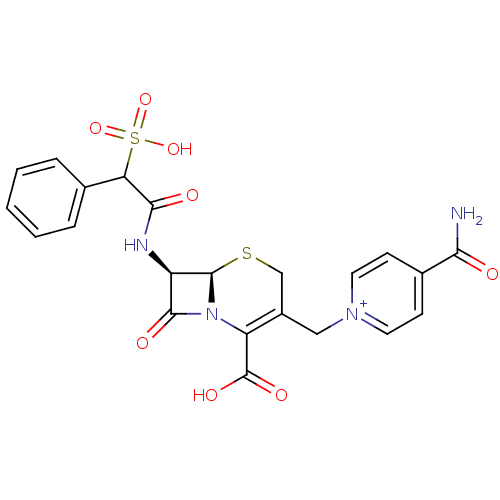
(CEFSULODIN)Show SMILES NC(=O)c1cc[n+](CC2=C(N3[C@H](SC2)[C@H](NC(=O)C(c2ccccc2)S(O)(=O)=O)C3=O)C(O)=O)cc1 |t:8| Show InChI InChI=1S/C22H20N4O8S2/c23-18(27)13-6-8-25(9-7-13)10-14-11-35-21-15(20(29)26(21)16(14)22(30)31)24-19(28)17(36(32,33)34)12-4-2-1-3-5-12/h1-9,15,17,21H,10-11H2,(H4-,23,24,27,28,30,31,32,33,34)/p+1/t15-,17?,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Competitive inhibition of phosphatase activity of SHP2 (unknown origin) using pNPP as a substrate Lineweaver-Burk plot analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50063918
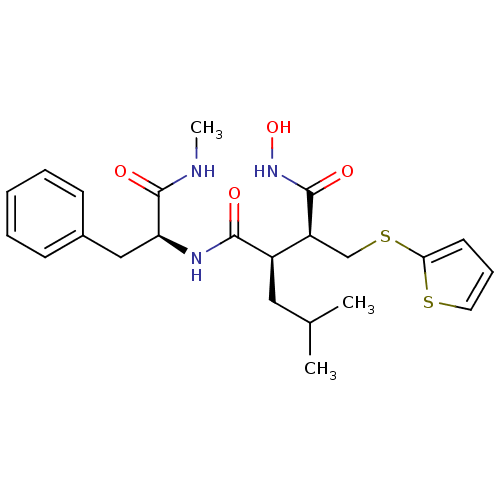
((2R,3S)-N*4*-Hydroxy-2-isobutyl-N*1*-((S)-1-methyl...)Show SMILES CNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(C)C)[C@H](CSc1cccs1)C(=O)NO Show InChI InChI=1S/C23H31N3O4S2/c1-15(2)12-17(18(22(28)26-30)14-32-20-10-7-11-31-20)21(27)25-19(23(29)24-3)13-16-8-5-4-6-9-16/h4-11,15,17-19,30H,12-14H2,1-3H3,(H,24,29)(H,25,27)(H,26,28)/t17-,18+,19+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Sejong University
Curated by ChEMBL
| Assay Description
Inhibition of human matrix metalloprotease-2 expressed in Sf9 insect cells |
Bioorg Med Chem Lett 15: 181-3 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.010
BindingDB Entry DOI: 10.7270/Q2BG2PRF |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50063918

((2R,3S)-N*4*-Hydroxy-2-isobutyl-N*1*-((S)-1-methyl...)Show SMILES CNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(C)C)[C@H](CSc1cccs1)C(=O)NO Show InChI InChI=1S/C23H31N3O4S2/c1-15(2)12-17(18(22(28)26-30)14-32-20-10-7-11-31-20)21(27)25-19(23(29)24-3)13-16-8-5-4-6-9-16/h4-11,15,17-19,30H,12-14H2,1-3H3,(H,24,29)(H,25,27)(H,26,28)/t17-,18+,19+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
Sejong University
Curated by ChEMBL
| Assay Description
Inhibition of human matrix metalloprotease-9 expressed in Sf9 insect cells |
Bioorg Med Chem Lett 15: 181-3 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.010
BindingDB Entry DOI: 10.7270/Q2BG2PRF |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-14
(Homo sapiens (Human)) | BDBM50063918

((2R,3S)-N*4*-Hydroxy-2-isobutyl-N*1*-((S)-1-methyl...)Show SMILES CNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(C)C)[C@H](CSc1cccs1)C(=O)NO Show InChI InChI=1S/C23H31N3O4S2/c1-15(2)12-17(18(22(28)26-30)14-32-20-10-7-11-31-20)21(27)25-19(23(29)24-3)13-16-8-5-4-6-9-16/h4-11,15,17-19,30H,12-14H2,1-3H3,(H,24,29)(H,25,27)(H,26,28)/t17-,18+,19+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 47 | n/a | n/a | n/a | n/a | n/a | n/a |
Sejong University
Curated by ChEMBL
| Assay Description
Inhibition of human matrix metalloprotease-14 expressed in Escherichia coli |
Bioorg Med Chem Lett 15: 181-3 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.010
BindingDB Entry DOI: 10.7270/Q2BG2PRF |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 11
(Homo sapiens (Human)) | BDBM50112358
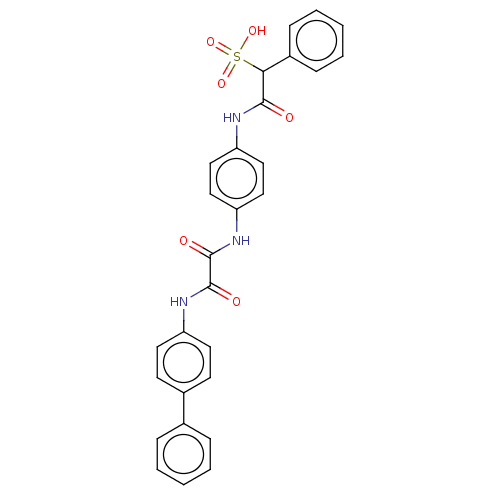
(CHEMBL3609374)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(cc2)-c2ccccc2)cc1)c1ccccc1 Show InChI InChI=1S/C28H23N3O6S/c32-26(25(38(35,36)37)21-9-5-2-6-10-21)29-23-15-17-24(18-16-23)31-28(34)27(33)30-22-13-11-20(12-14-22)19-7-3-1-4-8-19/h1-18,25H,(H,29,32)(H,30,33)(H,31,34)(H,35,36,37) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of SHP2 (unknown origin) using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 11
(Homo sapiens (Human)) | BDBM50112356

(CHEMBL3609373)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(I)cc2)cc1)c1ccccc1 Show InChI InChI=1S/C22H18IN3O6S/c23-15-6-8-16(9-7-15)25-21(28)22(29)26-18-12-10-17(11-13-18)24-20(27)19(33(30,31)32)14-4-2-1-3-5-14/h1-13,19H,(H,24,27)(H,25,28)(H,26,29)(H,30,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of SHP2 (unknown origin) using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 11
(Homo sapiens (Human)) | BDBM50112357

(CHEMBL3609375)Show SMILES CC(C)c1ccc(NC(=O)C(=O)Nc2ccc(NC(=O)C(c3ccccc3)S(O)(=O)=O)cc2)cc1 Show InChI InChI=1S/C25H25N3O6S/c1-16(2)17-8-10-19(11-9-17)27-24(30)25(31)28-21-14-12-20(13-15-21)26-23(29)22(35(32,33)34)18-6-4-3-5-7-18/h3-16,22H,1-2H3,(H,26,29)(H,27,30)(H,28,31)(H,32,33,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of SHP2 (unknown origin) using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50112358

(CHEMBL3609374)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(cc2)-c2ccccc2)cc1)c1ccccc1 Show InChI InChI=1S/C28H23N3O6S/c32-26(25(38(35,36)37)21-9-5-2-6-10-21)29-23-15-17-24(18-16-23)31-28(34)27(33)30-22-13-11-20(12-14-22)19-7-3-1-4-8-19/h1-18,25H,(H,29,32)(H,30,33)(H,31,34)(H,35,36,37) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human PTP1B using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Aminopeptidase N
(Homo sapiens (Human)) | BDBM50157325

(CHEMBL182879 | N-Hydroxy-2-(naphthalen-2-ylsulfany...)Show InChI InChI=1S/C12H11NO2S/c14-12(13-15)8-16-11-6-5-9-3-1-2-4-10(9)7-11/h1-7,15H,8H2,(H,13,14) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Sejong University
Curated by ChEMBL
| Assay Description
Inhibitory concentration against aminopeptidase N |
Bioorg Med Chem Lett 15: 181-3 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.010
BindingDB Entry DOI: 10.7270/Q2BG2PRF |
More data for this
Ligand-Target Pair | |
Aminopeptidase N
(Homo sapiens (Human)) | BDBM23971

((2S)-2-[(2S,3R)-3-amino-2-hydroxy-4-phenylbutanami...)Show SMILES CC(C)C[C@H](NC(=O)[C@@H](O)[C@H](N)Cc1ccccc1)C(O)=O Show InChI InChI=1S/C16H24N2O4/c1-10(2)8-13(16(21)22)18-15(20)14(19)12(17)9-11-6-4-3-5-7-11/h3-7,10,12-14,19H,8-9,17H2,1-2H3,(H,18,20)(H,21,22)/t12-,13+,14+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 3.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Sejong University
Curated by ChEMBL
| Assay Description
Inhibitory concentration against aminopeptidase N |
Bioorg Med Chem Lett 15: 181-3 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.010
BindingDB Entry DOI: 10.7270/Q2BG2PRF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein phosphatase non-receptor type 6
(Homo sapiens (Human)) | BDBM50112358

(CHEMBL3609374)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(cc2)-c2ccccc2)cc1)c1ccccc1 Show InChI InChI=1S/C28H23N3O6S/c32-26(25(38(35,36)37)21-9-5-2-6-10-21)29-23-15-17-24(18-16-23)31-28(34)27(33)30-22-13-11-20(12-14-22)19-7-3-1-4-8-19/h1-18,25H,(H,29,32)(H,30,33)(H,31,34)(H,35,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human SHP1 using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 6
(Homo sapiens (Human)) | BDBM50112357

(CHEMBL3609375)Show SMILES CC(C)c1ccc(NC(=O)C(=O)Nc2ccc(NC(=O)C(c3ccccc3)S(O)(=O)=O)cc2)cc1 Show InChI InChI=1S/C25H25N3O6S/c1-16(2)17-8-10-19(11-9-17)27-24(30)25(31)28-21-14-12-20(13-15-21)26-23(29)22(35(32,33)34)18-6-4-3-5-7-18/h3-16,22H,1-2H3,(H,26,29)(H,27,30)(H,28,31)(H,32,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human SHP1 using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50112357

(CHEMBL3609375)Show SMILES CC(C)c1ccc(NC(=O)C(=O)Nc2ccc(NC(=O)C(c3ccccc3)S(O)(=O)=O)cc2)cc1 Show InChI InChI=1S/C25H25N3O6S/c1-16(2)17-8-10-19(11-9-17)27-24(30)25(31)28-21-14-12-20(13-15-21)26-23(29)22(35(32,33)34)18-6-4-3-5-7-18/h3-16,22H,1-2H3,(H,26,29)(H,27,30)(H,28,31)(H,32,33,34) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human PTP1B using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 6
(Homo sapiens (Human)) | BDBM50112356

(CHEMBL3609373)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(I)cc2)cc1)c1ccccc1 Show InChI InChI=1S/C22H18IN3O6S/c23-15-6-8-16(9-7-15)25-21(28)22(29)26-18-12-10-17(11-13-18)24-20(27)19(33(30,31)32)14-4-2-1-3-5-14/h1-13,19H,(H,24,27)(H,25,28)(H,26,29)(H,30,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human SHP1 using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50112356

(CHEMBL3609373)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(I)cc2)cc1)c1ccccc1 Show InChI InChI=1S/C22H18IN3O6S/c23-15-6-8-16(9-7-15)25-21(28)22(29)26-18-12-10-17(11-13-18)24-20(27)19(33(30,31)32)14-4-2-1-3-5-14/h1-13,19H,(H,24,27)(H,25,28)(H,26,29)(H,30,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human PTP1B using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Cytosol aminopeptidase
(Homo sapiens (Human)) | BDBM23971

((2S)-2-[(2S,3R)-3-amino-2-hydroxy-4-phenylbutanami...)Show SMILES CC(C)C[C@H](NC(=O)[C@@H](O)[C@H](N)Cc1ccccc1)C(O)=O Show InChI InChI=1S/C16H24N2O4/c1-10(2)8-13(16(21)22)18-15(20)14(19)12(17)9-11-6-4-3-5-7-11/h3-7,10,12-14,19H,8-9,17H2,1-2H3,(H,18,20)(H,21,22)/t12-,13+,14+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.12E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Sejong University
Curated by ChEMBL
| Assay Description
Inhibitory concentration against adipocyte-derived leucine aminopeptidase |
Bioorg Med Chem Lett 15: 181-3 (2004)
Article DOI: 10.1016/j.bmcl.2004.10.010
BindingDB Entry DOI: 10.7270/Q2BG2PRF |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 11
(Homo sapiens (Human)) | BDBM50420258

(CEFSULODIN)Show SMILES NC(=O)c1cc[n+](CC2=C(N3[C@H](SC2)[C@H](NC(=O)C(c2ccccc2)S(O)(=O)=O)C3=O)C(O)=O)cc1 |t:8| Show InChI InChI=1S/C22H20N4O8S2/c23-18(27)13-6-8-25(9-7-13)10-14-11-35-21-15(20(29)26(21)16(14)22(30)31)24-19(28)17(36(32,33)34)12-4-2-1-3-5-12/h1-9,15,17,21H,10-11H2,(H4-,23,24,27,28,30,31,32,33,34)/p+1/t15-,17?,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.68E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of SHP2 (unknown origin) using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 6
(Homo sapiens (Human)) | BDBM50420258

(CEFSULODIN)Show SMILES NC(=O)c1cc[n+](CC2=C(N3[C@H](SC2)[C@H](NC(=O)C(c2ccccc2)S(O)(=O)=O)C3=O)C(O)=O)cc1 |t:8| Show InChI InChI=1S/C22H20N4O8S2/c23-18(27)13-6-8-25(9-7-13)10-14-11-35-21-15(20(29)26(21)16(14)22(30)31)24-19(28)17(36(32,33)34)12-4-2-1-3-5-12/h1-9,15,17,21H,10-11H2,(H4-,23,24,27,28,30,31,32,33,34)/p+1/t15-,17?,21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human SHP1 using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase CDC14A
(Homo sapiens (Human)) | BDBM50112358

(CHEMBL3609374)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(cc2)-c2ccccc2)cc1)c1ccccc1 Show InChI InChI=1S/C28H23N3O6S/c32-26(25(38(35,36)37)21-9-5-2-6-10-21)29-23-15-17-24(18-16-23)31-28(34)27(33)30-22-13-11-20(12-14-22)19-7-3-1-4-8-19/h1-18,25H,(H,29,32)(H,30,33)(H,31,34)(H,35,36,37) | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human CDC14A using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase CDC14A
(Homo sapiens (Human)) | BDBM50112356

(CHEMBL3609373)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(I)cc2)cc1)c1ccccc1 Show InChI InChI=1S/C22H18IN3O6S/c23-15-6-8-16(9-7-15)25-21(28)22(29)26-18-12-10-17(11-13-18)24-20(27)19(33(30,31)32)14-4-2-1-3-5-14/h1-13,19H,(H,24,27)(H,25,28)(H,26,29)(H,30,31,32) | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human CDC14A using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50112357

(CHEMBL3609375)Show SMILES CC(C)c1ccc(NC(=O)C(=O)Nc2ccc(NC(=O)C(c3ccccc3)S(O)(=O)=O)cc2)cc1 Show InChI InChI=1S/C25H25N3O6S/c1-16(2)17-8-10-19(11-9-17)27-24(30)25(31)28-21-14-12-20(13-15-21)26-23(29)22(35(32,33)34)18-6-4-3-5-7-18/h3-16,22H,1-2H3,(H,26,29)(H,27,30)(H,28,31)(H,32,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human VHR using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50112358

(CHEMBL3609374)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(cc2)-c2ccccc2)cc1)c1ccccc1 Show InChI InChI=1S/C28H23N3O6S/c32-26(25(38(35,36)37)21-9-5-2-6-10-21)29-23-15-17-24(18-16-23)31-28(34)27(33)30-22-13-11-20(12-14-22)19-7-3-1-4-8-19/h1-18,25H,(H,29,32)(H,30,33)(H,31,34)(H,35,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human VHR using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase 3
(Homo sapiens (Human)) | BDBM50112356

(CHEMBL3609373)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(I)cc2)cc1)c1ccccc1 Show InChI InChI=1S/C22H18IN3O6S/c23-15-6-8-16(9-7-15)25-21(28)22(29)26-18-12-10-17(11-13-18)24-20(27)19(33(30,31)32)14-4-2-1-3-5-14/h1-13,19H,(H,24,27)(H,25,28)(H,26,29)(H,30,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human VHR using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Dual specificity protein phosphatase CDC14A
(Homo sapiens (Human)) | BDBM50112357

(CHEMBL3609375)Show SMILES CC(C)c1ccc(NC(=O)C(=O)Nc2ccc(NC(=O)C(c3ccccc3)S(O)(=O)=O)cc2)cc1 Show InChI InChI=1S/C25H25N3O6S/c1-16(2)17-8-10-19(11-9-17)27-24(30)25(31)28-21-14-12-20(13-15-21)26-23(29)22(35(32,33)34)18-6-4-3-5-7-18/h3-16,22H,1-2H3,(H,26,29)(H,27,30)(H,28,31)(H,32,33,34) | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human CDC14A using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Low molecular weight phosphotyrosine protein phosphatase
(Homo sapiens (Human)) | BDBM50112356

(CHEMBL3609373)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(I)cc2)cc1)c1ccccc1 Show InChI InChI=1S/C22H18IN3O6S/c23-15-6-8-16(9-7-15)25-21(28)22(29)26-18-12-10-17(11-13-18)24-20(27)19(33(30,31)32)14-4-2-1-3-5-14/h1-13,19H,(H,24,27)(H,25,28)(H,26,29)(H,30,31,32) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human LMWPTP using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Low molecular weight phosphotyrosine protein phosphatase
(Homo sapiens (Human)) | BDBM50112358

(CHEMBL3609374)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(cc2)-c2ccccc2)cc1)c1ccccc1 Show InChI InChI=1S/C28H23N3O6S/c32-26(25(38(35,36)37)21-9-5-2-6-10-21)29-23-15-17-24(18-16-23)31-28(34)27(33)30-22-13-11-20(12-14-22)19-7-3-1-4-8-19/h1-18,25H,(H,29,32)(H,30,33)(H,31,34)(H,35,36,37) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human LMWPTP using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Low molecular weight phosphotyrosine protein phosphatase
(Homo sapiens (Human)) | BDBM50112357

(CHEMBL3609375)Show SMILES CC(C)c1ccc(NC(=O)C(=O)Nc2ccc(NC(=O)C(c3ccccc3)S(O)(=O)=O)cc2)cc1 Show InChI InChI=1S/C25H25N3O6S/c1-16(2)17-8-10-19(11-9-17)27-24(30)25(31)28-21-14-12-20(13-15-21)26-23(29)22(35(32,33)34)18-6-4-3-5-7-18/h3-16,22H,1-2H3,(H,26,29)(H,27,30)(H,28,31)(H,32,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human LMWPTP using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein phosphatase 5
(Homo sapiens (Human)) | BDBM50112358

(CHEMBL3609374)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(cc2)-c2ccccc2)cc1)c1ccccc1 Show InChI InChI=1S/C28H23N3O6S/c32-26(25(38(35,36)37)21-9-5-2-6-10-21)29-23-15-17-24(18-16-23)31-28(34)27(33)30-22-13-11-20(12-14-22)19-7-3-1-4-8-19/h1-18,25H,(H,29,32)(H,30,33)(H,31,34)(H,35,36,37) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human PP5 using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein phosphatase 5
(Homo sapiens (Human)) | BDBM50112357

(CHEMBL3609375)Show SMILES CC(C)c1ccc(NC(=O)C(=O)Nc2ccc(NC(=O)C(c3ccccc3)S(O)(=O)=O)cc2)cc1 Show InChI InChI=1S/C25H25N3O6S/c1-16(2)17-8-10-19(11-9-17)27-24(30)25(31)28-21-14-12-20(13-15-21)26-23(29)22(35(32,33)34)18-6-4-3-5-7-18/h3-16,22H,1-2H3,(H,26,29)(H,27,30)(H,28,31)(H,32,33,34) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human PP5 using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein phosphatase 5
(Homo sapiens (Human)) | BDBM50112356

(CHEMBL3609373)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(I)cc2)cc1)c1ccccc1 Show InChI InChI=1S/C22H18IN3O6S/c23-15-6-8-16(9-7-15)25-21(28)22(29)26-18-12-10-17(11-13-18)24-20(27)19(33(30,31)32)14-4-2-1-3-5-14/h1-13,19H,(H,24,27)(H,25,28)(H,26,29)(H,30,31,32) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human PP5 using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 22
(Homo sapiens (Human)) | BDBM50112356

(CHEMBL3609373)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(I)cc2)cc1)c1ccccc1 Show InChI InChI=1S/C22H18IN3O6S/c23-15-6-8-16(9-7-15)25-21(28)22(29)26-18-12-10-17(11-13-18)24-20(27)19(33(30,31)32)14-4-2-1-3-5-14/h1-13,19H,(H,24,27)(H,25,28)(H,26,29)(H,30,31,32) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human LYP using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 22
(Homo sapiens (Human)) | BDBM50112358

(CHEMBL3609374)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(cc2)-c2ccccc2)cc1)c1ccccc1 Show InChI InChI=1S/C28H23N3O6S/c32-26(25(38(35,36)37)21-9-5-2-6-10-21)29-23-15-17-24(18-16-23)31-28(34)27(33)30-22-13-11-20(12-14-22)19-7-3-1-4-8-19/h1-18,25H,(H,29,32)(H,30,33)(H,31,34)(H,35,36,37) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human LYP using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 22
(Homo sapiens (Human)) | BDBM50112357

(CHEMBL3609375)Show SMILES CC(C)c1ccc(NC(=O)C(=O)Nc2ccc(NC(=O)C(c3ccccc3)S(O)(=O)=O)cc2)cc1 Show InChI InChI=1S/C25H25N3O6S/c1-16(2)17-8-10-19(11-9-17)27-24(30)25(31)28-21-14-12-20(13-15-21)26-23(29)22(35(32,33)34)18-6-4-3-5-7-18/h3-16,22H,1-2H3,(H,26,29)(H,27,30)(H,28,31)(H,32,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human LYP using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 7
(Homo sapiens (Human)) | BDBM50112356

(CHEMBL3609373)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(I)cc2)cc1)c1ccccc1 Show InChI InChI=1S/C22H18IN3O6S/c23-15-6-8-16(9-7-15)25-21(28)22(29)26-18-12-10-17(11-13-18)24-20(27)19(33(30,31)32)14-4-2-1-3-5-14/h1-13,19H,(H,24,27)(H,25,28)(H,26,29)(H,30,31,32) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human HePTP using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 7
(Homo sapiens (Human)) | BDBM50112358

(CHEMBL3609374)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(cc2)-c2ccccc2)cc1)c1ccccc1 Show InChI InChI=1S/C28H23N3O6S/c32-26(25(38(35,36)37)21-9-5-2-6-10-21)29-23-15-17-24(18-16-23)31-28(34)27(33)30-22-13-11-20(12-14-22)19-7-3-1-4-8-19/h1-18,25H,(H,29,32)(H,30,33)(H,31,34)(H,35,36,37) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human HePTP using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 7
(Homo sapiens (Human)) | BDBM50112357

(CHEMBL3609375)Show SMILES CC(C)c1ccc(NC(=O)C(=O)Nc2ccc(NC(=O)C(c3ccccc3)S(O)(=O)=O)cc2)cc1 Show InChI InChI=1S/C25H25N3O6S/c1-16(2)17-8-10-19(11-9-17)27-24(30)25(31)28-21-14-12-20(13-15-21)26-23(29)22(35(32,33)34)18-6-4-3-5-7-18/h3-16,22H,1-2H3,(H,26,29)(H,27,30)(H,28,31)(H,32,33,34) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human HePTP using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 9
(Homo sapiens (Human)) | BDBM50112356

(CHEMBL3609373)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(I)cc2)cc1)c1ccccc1 Show InChI InChI=1S/C22H18IN3O6S/c23-15-6-8-16(9-7-15)25-21(28)22(29)26-18-12-10-17(11-13-18)24-20(27)19(33(30,31)32)14-4-2-1-3-5-14/h1-13,19H,(H,24,27)(H,25,28)(H,26,29)(H,30,31,32) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human Meg2 using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 9
(Homo sapiens (Human)) | BDBM50112358

(CHEMBL3609374)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(cc2)-c2ccccc2)cc1)c1ccccc1 Show InChI InChI=1S/C28H23N3O6S/c32-26(25(38(35,36)37)21-9-5-2-6-10-21)29-23-15-17-24(18-16-23)31-28(34)27(33)30-22-13-11-20(12-14-22)19-7-3-1-4-8-19/h1-18,25H,(H,29,32)(H,30,33)(H,31,34)(H,35,36,37) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human Meg2 using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 9
(Homo sapiens (Human)) | BDBM50112357

(CHEMBL3609375)Show SMILES CC(C)c1ccc(NC(=O)C(=O)Nc2ccc(NC(=O)C(c3ccccc3)S(O)(=O)=O)cc2)cc1 Show InChI InChI=1S/C25H25N3O6S/c1-16(2)17-8-10-19(11-9-17)27-24(30)25(31)28-21-14-12-20(13-15-21)26-23(29)22(35(32,33)34)18-6-4-3-5-7-18/h3-16,22H,1-2H3,(H,26,29)(H,27,30)(H,28,31)(H,32,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human Meg2 using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase alpha
(Homo sapiens (Human)) | BDBM50112356

(CHEMBL3609373)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(I)cc2)cc1)c1ccccc1 Show InChI InChI=1S/C22H18IN3O6S/c23-15-6-8-16(9-7-15)25-21(28)22(29)26-18-12-10-17(11-13-18)24-20(27)19(33(30,31)32)14-4-2-1-3-5-14/h1-13,19H,(H,24,27)(H,25,28)(H,26,29)(H,30,31,32) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human PTPalpha using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase alpha
(Homo sapiens (Human)) | BDBM50112358

(CHEMBL3609374)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(cc2)-c2ccccc2)cc1)c1ccccc1 Show InChI InChI=1S/C28H23N3O6S/c32-26(25(38(35,36)37)21-9-5-2-6-10-21)29-23-15-17-24(18-16-23)31-28(34)27(33)30-22-13-11-20(12-14-22)19-7-3-1-4-8-19/h1-18,25H,(H,29,32)(H,30,33)(H,31,34)(H,35,36,37) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human PTPalpha using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase alpha
(Homo sapiens (Human)) | BDBM50112357

(CHEMBL3609375)Show SMILES CC(C)c1ccc(NC(=O)C(=O)Nc2ccc(NC(=O)C(c3ccccc3)S(O)(=O)=O)cc2)cc1 Show InChI InChI=1S/C25H25N3O6S/c1-16(2)17-8-10-19(11-9-17)27-24(30)25(31)28-21-14-12-20(13-15-21)26-23(29)22(35(32,33)34)18-6-4-3-5-7-18/h3-16,22H,1-2H3,(H,26,29)(H,27,30)(H,28,31)(H,32,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human PTPalpha using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase beta
(Homo sapiens (Human)) | BDBM50112356

(CHEMBL3609373)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(I)cc2)cc1)c1ccccc1 Show InChI InChI=1S/C22H18IN3O6S/c23-15-6-8-16(9-7-15)25-21(28)22(29)26-18-12-10-17(11-13-18)24-20(27)19(33(30,31)32)14-4-2-1-3-5-14/h1-13,19H,(H,24,27)(H,25,28)(H,26,29)(H,30,31,32) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human PTPbeta using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase F
(Homo sapiens (Human)) | BDBM50112357

(CHEMBL3609375)Show SMILES CC(C)c1ccc(NC(=O)C(=O)Nc2ccc(NC(=O)C(c3ccccc3)S(O)(=O)=O)cc2)cc1 Show InChI InChI=1S/C25H25N3O6S/c1-16(2)17-8-10-19(11-9-17)27-24(30)25(31)28-21-14-12-20(13-15-21)26-23(29)22(35(32,33)34)18-6-4-3-5-7-18/h3-16,22H,1-2H3,(H,26,29)(H,27,30)(H,28,31)(H,32,33,34) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human LAR using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase F
(Homo sapiens (Human)) | BDBM50112358

(CHEMBL3609374)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(cc2)-c2ccccc2)cc1)c1ccccc1 Show InChI InChI=1S/C28H23N3O6S/c32-26(25(38(35,36)37)21-9-5-2-6-10-21)29-23-15-17-24(18-16-23)31-28(34)27(33)30-22-13-11-20(12-14-22)19-7-3-1-4-8-19/h1-18,25H,(H,29,32)(H,30,33)(H,31,34)(H,35,36,37) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human LAR using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase F
(Homo sapiens (Human)) | BDBM50112356

(CHEMBL3609373)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(I)cc2)cc1)c1ccccc1 Show InChI InChI=1S/C22H18IN3O6S/c23-15-6-8-16(9-7-15)25-21(28)22(29)26-18-12-10-17(11-13-18)24-20(27)19(33(30,31)32)14-4-2-1-3-5-14/h1-13,19H,(H,24,27)(H,25,28)(H,26,29)(H,30,31,32) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human LAR using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase mu
(Homo sapiens (Human)) | BDBM50112357

(CHEMBL3609375)Show SMILES CC(C)c1ccc(NC(=O)C(=O)Nc2ccc(NC(=O)C(c3ccccc3)S(O)(=O)=O)cc2)cc1 Show InChI InChI=1S/C25H25N3O6S/c1-16(2)17-8-10-19(11-9-17)27-24(30)25(31)28-21-14-12-20(13-15-21)26-23(29)22(35(32,33)34)18-6-4-3-5-7-18/h3-16,22H,1-2H3,(H,26,29)(H,27,30)(H,28,31)(H,32,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human PTPmu using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase mu
(Homo sapiens (Human)) | BDBM50112358

(CHEMBL3609374)Show SMILES OS(=O)(=O)C(C(=O)Nc1ccc(NC(=O)C(=O)Nc2ccc(cc2)-c2ccccc2)cc1)c1ccccc1 Show InChI InChI=1S/C28H23N3O6S/c32-26(25(38(35,36)37)21-9-5-2-6-10-21)29-23-15-17-24(18-16-23)31-28(34)27(33)30-22-13-11-20(12-14-22)19-7-3-1-4-8-19/h1-18,25H,(H,29,32)(H,30,33)(H,31,34)(H,35,36,37) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Indiana University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of phosphatase activity of human PTPmu using pNPP as a substrate after 10 mins by spectrophotometer analysis |
ACS Med Chem Lett 6: 782-6 (2015)
Article DOI: 10.1021/acsmedchemlett.5b00118
BindingDB Entry DOI: 10.7270/Q2251M0S |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data