 Found 174 hits with Last Name = 'thorn' and Initial = 's'
Found 174 hits with Last Name = 'thorn' and Initial = 's' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Procathepsin L
(Homo sapiens (Human)) | BDBM50090644

(CHEMBL48837 | {(S)-1-[(S)-1-(4-Butoxy-benzyl)-2,3-...)Show SMILES CCCCOc1ccc(C[C@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)OCc2ccccc2)C(=O)C=O)cc1 Show InChI InChI=1S/C31H34N2O6/c1-2-3-18-38-26-16-14-24(15-17-26)19-27(29(35)21-34)32-30(36)28(20-23-10-6-4-7-11-23)33-31(37)39-22-25-12-8-5-9-13-25/h4-17,21,27-28H,2-3,18-20,22H2,1H3,(H,32,36)(H,33,37)/t27-,28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Queen's University Belfast
Curated by ChEMBL
| Assay Description
Ability to block cathepsin L-catalyzed hydrolysis of the fluorogenic substrate Z-Phe-Arg-NHMec |
Bioorg Med Chem Lett 10: 1771-3 (2000)
BindingDB Entry DOI: 10.7270/Q2TQ60SW |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Bos taurus (bovine)) | BDBM50090645

((S)-2-Benzyl-N-[(S)-4-guanidino-1-(2-oxo-acetyl)-b...)Show SMILES NC(=N)NCCC[C@H](NC(=O)[C@H](Cc1ccccc1)C(=O)OCc1ccccc1)C(=O)C=O Show InChI InChI=1S/C24H28N4O5/c25-24(26)27-13-7-12-20(21(30)15-29)28-22(31)19(14-17-8-3-1-4-9-17)23(32)33-16-18-10-5-2-6-11-18/h1-6,8-11,15,19-20H,7,12-14,16H2,(H,28,31)(H4,25,26,27)/t19-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Queen's University Belfast
Curated by ChEMBL
| Assay Description
Ability to block cathepsin B-catalyzed hydrolysis of the fluorogenic substrate Z-Arg-Arg-NHMec |
Bioorg Med Chem Lett 10: 1771-3 (2000)
BindingDB Entry DOI: 10.7270/Q2TQ60SW |
More data for this
Ligand-Target Pair | |
Procathepsin L
(Homo sapiens (Human)) | BDBM50090645

((S)-2-Benzyl-N-[(S)-4-guanidino-1-(2-oxo-acetyl)-b...)Show SMILES NC(=N)NCCC[C@H](NC(=O)[C@H](Cc1ccccc1)C(=O)OCc1ccccc1)C(=O)C=O Show InChI InChI=1S/C24H28N4O5/c25-24(26)27-13-7-12-20(21(30)15-29)28-22(31)19(14-17-8-3-1-4-9-17)23(32)33-16-18-10-5-2-6-11-18/h1-6,8-11,15,19-20H,7,12-14,16H2,(H,28,31)(H4,25,26,27)/t19-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| PubMed
| 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Queen's University Belfast
Curated by ChEMBL
| Assay Description
Ability to block cathepsin L-catalyzed hydrolysis of the fluorogenic substrate Z-Phe-Arg-NHMec |
Bioorg Med Chem Lett 10: 1771-3 (2000)
BindingDB Entry DOI: 10.7270/Q2TQ60SW |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM81670

(Peptidyl ene diones, COOEt)Show SMILES CCOC(=O)C=CC(=O)[C@H](C)NC(=O)[C@H](Cc1ccccc1)NC(=O)OCc1ccccc1 |r,w:6.6| Show InChI InChI=1S/C25H28N2O6/c1-3-32-23(29)15-14-22(28)18(2)26-24(30)21(16-19-10-6-4-7-11-19)27-25(31)33-17-20-12-8-5-9-13-20/h4-15,18,21H,3,16-17H2,1-2H3,(H,26,30)(H,27,31)/t18-,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 94 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Queen's University of Belfast,
| Assay Description
Inhibition assay using peptidyl ene diones to inactivate human cathepsins B and L. |
Chem Biol Drug Des 69: 170-9 (2007)
Article DOI: 10.1111/j.1747-0285.2007.00490.x
BindingDB Entry DOI: 10.7270/Q2348HVC |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM81671
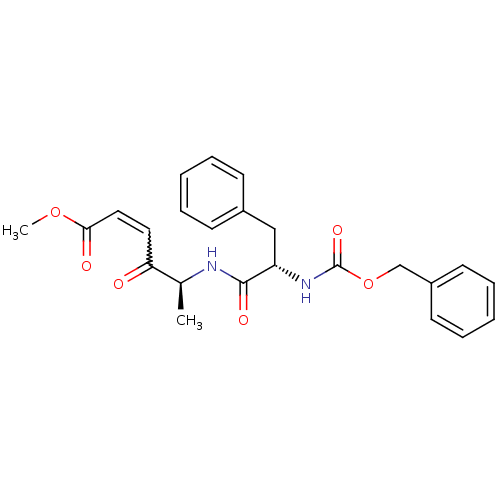
(Peptidyl ene diones, COOMe)Show SMILES COC(=O)C=CC(=O)[C@H](C)NC(=O)[C@H](Cc1ccccc1)NC(=O)OCc1ccccc1 |r,w:5.5| Show InChI InChI=1S/C24H26N2O6/c1-17(21(27)13-14-22(28)31-2)25-23(29)20(15-18-9-5-3-6-10-18)26-24(30)32-16-19-11-7-4-8-12-19/h3-14,17,20H,15-16H2,1-2H3,(H,25,29)(H,26,30)/t17-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 125 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Queen's University of Belfast,
| Assay Description
Inhibition assay using peptidyl ene diones to inactivate human cathepsins B and L. |
Chem Biol Drug Des 69: 170-9 (2007)
Article DOI: 10.1111/j.1747-0285.2007.00490.x
BindingDB Entry DOI: 10.7270/Q2348HVC |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Bos taurus (bovine)) | BDBM50090644

(CHEMBL48837 | {(S)-1-[(S)-1-(4-Butoxy-benzyl)-2,3-...)Show SMILES CCCCOc1ccc(C[C@H](NC(=O)[C@H](Cc2ccccc2)NC(=O)OCc2ccccc2)C(=O)C=O)cc1 Show InChI InChI=1S/C31H34N2O6/c1-2-3-18-38-26-16-14-24(15-17-26)19-27(29(35)21-34)32-30(36)28(20-23-10-6-4-7-11-23)33-31(37)39-22-25-12-8-5-9-13-25/h4-17,21,27-28H,2-3,18-20,22H2,1H3,(H,32,36)(H,33,37)/t27-,28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 214 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Queen's University Belfast
Curated by ChEMBL
| Assay Description
Ability to block cathepsin B-catalyzed hydrolysis of the fluorogenic substrate Z-Arg-Arg-NHMec |
Bioorg Med Chem Lett 10: 1771-3 (2000)
BindingDB Entry DOI: 10.7270/Q2TQ60SW |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Bos taurus (bovine)) | BDBM50090646
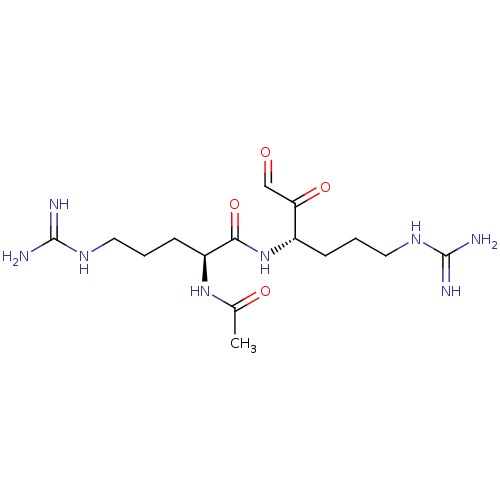
((S)-2-Acetylamino-5-guanidino-pentanoic acid [(S)-...)Show SMILES CC(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)C=O Show InChI InChI=1S/C15H28N8O4/c1-9(25)22-11(5-3-7-21-15(18)19)13(27)23-10(12(26)8-24)4-2-6-20-14(16)17/h8,10-11H,2-7H2,1H3,(H,22,25)(H,23,27)(H4,16,17,20)(H4,18,19,21)/t10-,11-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 640 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Queen's University Belfast
Curated by ChEMBL
| Assay Description
Ability to block cathepsin B-catalyzed hydrolysis of the fluorogenic substrate Z-Arg-Arg-NHMec |
Bioorg Med Chem Lett 10: 1771-3 (2000)
BindingDB Entry DOI: 10.7270/Q2TQ60SW |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM81670

(Peptidyl ene diones, COOEt)Show SMILES CCOC(=O)C=CC(=O)[C@H](C)NC(=O)[C@H](Cc1ccccc1)NC(=O)OCc1ccccc1 |r,w:6.6| Show InChI InChI=1S/C25H28N2O6/c1-3-32-23(29)15-14-22(28)18(2)26-24(30)21(16-19-10-6-4-7-11-19)27-25(31)33-17-20-12-8-5-9-13-20/h4-15,18,21H,3,16-17H2,1-2H3,(H,26,30)(H,27,31)/t18-,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Queen's University of Belfast,
| Assay Description
Inhibition assay using peptidyl ene diones to inactivate human cathepsins B and L. |
Chem Biol Drug Des 69: 170-9 (2007)
Article DOI: 10.1111/j.1747-0285.2007.00490.x
BindingDB Entry DOI: 10.7270/Q2348HVC |
More data for this
Ligand-Target Pair | |
Procathepsin L
(Homo sapiens (Human)) | BDBM50090643
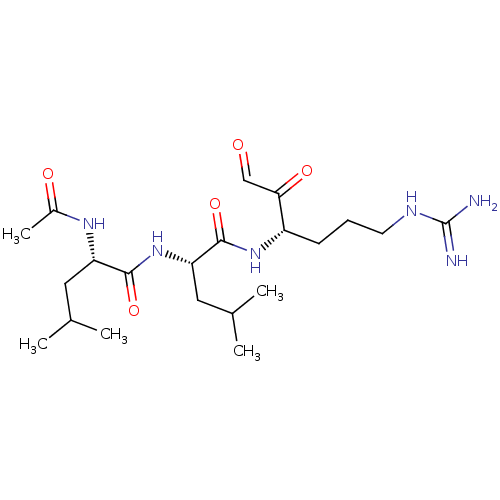
((S)-2-Acetylamino-4-methyl-pentanoic acid {(S)-1-[...)Show SMILES CC(C)C[C@H](NC(C)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)C=O Show InChI InChI=1S/C21H38N6O5/c1-12(2)9-16(25-14(5)29)19(31)27-17(10-13(3)4)20(32)26-15(18(30)11-28)7-6-8-24-21(22)23/h11-13,15-17H,6-10H2,1-5H3,(H,25,29)(H,26,32)(H,27,31)(H4,22,23,24)/t15-,16-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 970 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Queen's University Belfast
Curated by ChEMBL
| Assay Description
Ability to block cathepsin L-catalyzed hydrolysis of the fluorogenic substrate Z-Phe-Arg-NHMec |
Bioorg Med Chem Lett 10: 1771-3 (2000)
BindingDB Entry DOI: 10.7270/Q2TQ60SW |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Bos taurus (bovine)) | BDBM50090643

((S)-2-Acetylamino-4-methyl-pentanoic acid {(S)-1-[...)Show SMILES CC(C)C[C@H](NC(C)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)C=O Show InChI InChI=1S/C21H38N6O5/c1-12(2)9-16(25-14(5)29)19(31)27-17(10-13(3)4)20(32)26-15(18(30)11-28)7-6-8-24-21(22)23/h11-13,15-17H,6-10H2,1-5H3,(H,25,29)(H,26,32)(H,27,31)(H4,22,23,24)/t15-,16-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.25E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Queen's University Belfast
Curated by ChEMBL
| Assay Description
Ability to block cathepsin B-catalyzed hydrolysis of the fluorogenic substrate Z-Arg-Arg-NHMec |
Bioorg Med Chem Lett 10: 1771-3 (2000)
BindingDB Entry DOI: 10.7270/Q2TQ60SW |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50421506
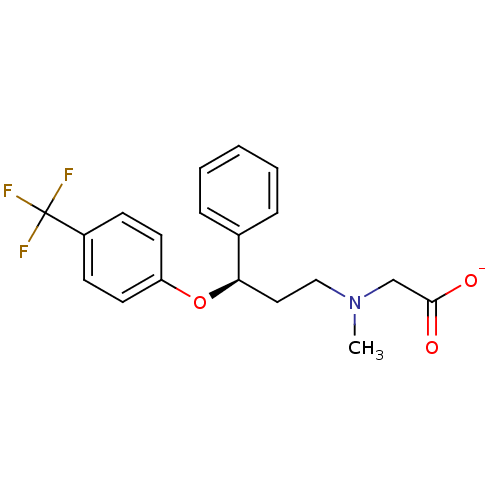
(CHEMBL61861 | ORG-24598)Show SMILES CN(CC[C@@H](Oc1ccc(cc1)C(F)(F)F)c1ccccc1)CC([O-])=O Show InChI InChI=1S/C19H20F3NO3/c1-23(13-18(24)25)12-11-17(14-5-3-2-4-6-14)26-16-9-7-15(8-10-16)19(20,21)22/h2-10,17H,11-13H2,1H3,(H,24,25)/p-1/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organon Research and Development Group
Curated by ChEMBL
| Assay Description
Binding affinity of compound towards Beta-2 adrenergic receptor was determined |
Bioorg Med Chem Lett 11: 2007-9 (2001)
BindingDB Entry DOI: 10.7270/Q2X0687Q |
More data for this
Ligand-Target Pair | |
Alpha-1B adrenergic receptor
(Homo sapiens (Human)) | BDBM50421506

(CHEMBL61861 | ORG-24598)Show SMILES CN(CC[C@@H](Oc1ccc(cc1)C(F)(F)F)c1ccccc1)CC([O-])=O Show InChI InChI=1S/C19H20F3NO3/c1-23(13-18(24)25)12-11-17(14-5-3-2-4-6-14)26-16-9-7-15(8-10-16)19(20,21)22/h2-10,17H,11-13H2,1H3,(H,24,25)/p-1/t17-/m1/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organon Research and Development Group
Curated by ChEMBL
| Assay Description
The compound was tested for the binding affinity towards Alpha-1B adrenergic receptor |
Bioorg Med Chem Lett 11: 2007-9 (2001)
BindingDB Entry DOI: 10.7270/Q2X0687Q |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50421506

(CHEMBL61861 | ORG-24598)Show SMILES CN(CC[C@@H](Oc1ccc(cc1)C(F)(F)F)c1ccccc1)CC([O-])=O Show InChI InChI=1S/C19H20F3NO3/c1-23(13-18(24)25)12-11-17(14-5-3-2-4-6-14)26-16-9-7-15(8-10-16)19(20,21)22/h2-10,17H,11-13H2,1H3,(H,24,25)/p-1/t17-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organon Research and Development Group
Curated by ChEMBL
| Assay Description
Binding affinity of compound towards Beta-1 adrenergic receptor was determined |
Bioorg Med Chem Lett 11: 2007-9 (2001)
BindingDB Entry DOI: 10.7270/Q2X0687Q |
More data for this
Ligand-Target Pair | |
Alpha-2C adrenergic receptor
(Homo sapiens (Human)) | BDBM50421506

(CHEMBL61861 | ORG-24598)Show SMILES CN(CC[C@@H](Oc1ccc(cc1)C(F)(F)F)c1ccccc1)CC([O-])=O Show InChI InChI=1S/C19H20F3NO3/c1-23(13-18(24)25)12-11-17(14-5-3-2-4-6-14)26-16-9-7-15(8-10-16)19(20,21)22/h2-10,17H,11-13H2,1H3,(H,24,25)/p-1/t17-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organon Research and Development Group
Curated by ChEMBL
| Assay Description
Binding affinity of compound towards Alpha-2C adrenergic receptor was determined |
Bioorg Med Chem Lett 11: 2007-9 (2001)
BindingDB Entry DOI: 10.7270/Q2X0687Q |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM50421506

(CHEMBL61861 | ORG-24598)Show SMILES CN(CC[C@@H](Oc1ccc(cc1)C(F)(F)F)c1ccccc1)CC([O-])=O Show InChI InChI=1S/C19H20F3NO3/c1-23(13-18(24)25)12-11-17(14-5-3-2-4-6-14)26-16-9-7-15(8-10-16)19(20,21)22/h2-10,17H,11-13H2,1H3,(H,24,25)/p-1/t17-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organon Research and Development Group
Curated by ChEMBL
| Assay Description
Binding affinity of compound towards serotonin 5-hydroxytryptamine 2A receptor was determined |
Bioorg Med Chem Lett 11: 2007-9 (2001)
BindingDB Entry DOI: 10.7270/Q2X0687Q |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(RAT) | BDBM50421506

(CHEMBL61861 | ORG-24598)Show SMILES CN(CC[C@@H](Oc1ccc(cc1)C(F)(F)F)c1ccccc1)CC([O-])=O Show InChI InChI=1S/C19H20F3NO3/c1-23(13-18(24)25)12-11-17(14-5-3-2-4-6-14)26-16-9-7-15(8-10-16)19(20,21)22/h2-10,17H,11-13H2,1H3,(H,24,25)/p-1/t17-/m1/s1 | Reactome pathway
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organon Research and Development Group
Curated by ChEMBL
| Assay Description
Binding affinity of compound towards Dopamine receptor D4 was determined |
Bioorg Med Chem Lett 11: 2007-9 (2001)
BindingDB Entry DOI: 10.7270/Q2X0687Q |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(RAT) | BDBM50421506

(CHEMBL61861 | ORG-24598)Show SMILES CN(CC[C@@H](Oc1ccc(cc1)C(F)(F)F)c1ccccc1)CC([O-])=O Show InChI InChI=1S/C19H20F3NO3/c1-23(13-18(24)25)12-11-17(14-5-3-2-4-6-14)26-16-9-7-15(8-10-16)19(20,21)22/h2-10,17H,11-13H2,1H3,(H,24,25)/p-1/t17-/m1/s1 | PDB
Reactome pathway
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organon Research and Development Group
Curated by ChEMBL
| Assay Description
Binding affinity of compound towards serotonin 5-hydroxytryptamine 6 receptor was determined |
Bioorg Med Chem Lett 11: 2007-9 (2001)
BindingDB Entry DOI: 10.7270/Q2X0687Q |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Rattus norvegicus (rat)) | BDBM50421506

(CHEMBL61861 | ORG-24598)Show SMILES CN(CC[C@@H](Oc1ccc(cc1)C(F)(F)F)c1ccccc1)CC([O-])=O Show InChI InChI=1S/C19H20F3NO3/c1-23(13-18(24)25)12-11-17(14-5-3-2-4-6-14)26-16-9-7-15(8-10-16)19(20,21)22/h2-10,17H,11-13H2,1H3,(H,24,25)/p-1/t17-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organon Research and Development Group
Curated by ChEMBL
| Assay Description
Binding affinity of compound towards 5-hydroxytryptamine 1A receptor was determined |
Bioorg Med Chem Lett 11: 2007-9 (2001)
BindingDB Entry DOI: 10.7270/Q2X0687Q |
More data for this
Ligand-Target Pair | |
Alpha-1A adrenergic receptor
(Homo sapiens (Human)) | BDBM50421506

(CHEMBL61861 | ORG-24598)Show SMILES CN(CC[C@@H](Oc1ccc(cc1)C(F)(F)F)c1ccccc1)CC([O-])=O Show InChI InChI=1S/C19H20F3NO3/c1-23(13-18(24)25)12-11-17(14-5-3-2-4-6-14)26-16-9-7-15(8-10-16)19(20,21)22/h2-10,17H,11-13H2,1H3,(H,24,25)/p-1/t17-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organon Research and Development Group
Curated by ChEMBL
| Assay Description
Binding affinity of compound towards Binding affinity towards Alpha-1A adrenergic receptor was determined |
Bioorg Med Chem Lett 11: 2007-9 (2001)
BindingDB Entry DOI: 10.7270/Q2X0687Q |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2C
(Rattus norvegicus (Rat)) | BDBM50421506

(CHEMBL61861 | ORG-24598)Show SMILES CN(CC[C@@H](Oc1ccc(cc1)C(F)(F)F)c1ccccc1)CC([O-])=O Show InChI InChI=1S/C19H20F3NO3/c1-23(13-18(24)25)12-11-17(14-5-3-2-4-6-14)26-16-9-7-15(8-10-16)19(20,21)22/h2-10,17H,11-13H2,1H3,(H,24,25)/p-1/t17-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organon Research and Development Group
Curated by ChEMBL
| Assay Description
Binding affinity of compound towards serotonin 5-hydroxytryptamine 2C receptor was determined |
Bioorg Med Chem Lett 11: 2007-9 (2001)
BindingDB Entry DOI: 10.7270/Q2X0687Q |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Rattus norvegicus (rat)) | BDBM50421506

(CHEMBL61861 | ORG-24598)Show SMILES CN(CC[C@@H](Oc1ccc(cc1)C(F)(F)F)c1ccccc1)CC([O-])=O Show InChI InChI=1S/C19H20F3NO3/c1-23(13-18(24)25)12-11-17(14-5-3-2-4-6-14)26-16-9-7-15(8-10-16)19(20,21)22/h2-10,17H,11-13H2,1H3,(H,24,25)/p-1/t17-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organon Research and Development Group
Curated by ChEMBL
| Assay Description
Binding affinity of compound towards Dopamine receptor D2S was determined |
Bioorg Med Chem Lett 11: 2007-9 (2001)
BindingDB Entry DOI: 10.7270/Q2X0687Q |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 7
(Rattus norvegicus (rat)) | BDBM50421506

(CHEMBL61861 | ORG-24598)Show SMILES CN(CC[C@@H](Oc1ccc(cc1)C(F)(F)F)c1ccccc1)CC([O-])=O Show InChI InChI=1S/C19H20F3NO3/c1-23(13-18(24)25)12-11-17(14-5-3-2-4-6-14)26-16-9-7-15(8-10-16)19(20,21)22/h2-10,17H,11-13H2,1H3,(H,24,25)/p-1/t17-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organon Research and Development Group
Curated by ChEMBL
| Assay Description
Binding affinity of compound towards serotonin 5-hydroxytryptamine 7 receptor was determined |
Bioorg Med Chem Lett 11: 2007-9 (2001)
BindingDB Entry DOI: 10.7270/Q2X0687Q |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50502628

(CHEMBL4469630)Show SMILES CC(C)(C)CN1C[C@@]2(CCC[C@](C)(Cn3cnc4ccc(cc34)C#N)C2)OC1=O |r| Show InChI InChI=1S/C23H30N4O2/c1-21(2,3)13-26-15-23(29-20(26)28)9-5-8-22(4,12-23)14-27-16-25-18-7-6-17(11-24)10-19(18)27/h6-7,10,16H,5,8-9,12-15H2,1-4H3/t22-,23-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human TRPV4 expressed in baculovirus infected HEK/MSR 2 cells pre-incubated for 10 mins followed by GSK634775A addition by Fura-2 dye b... |
ACS Med Chem Lett 10: 1228-1233 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00274
BindingDB Entry DOI: 10.7270/Q2RX9GBF |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50502635
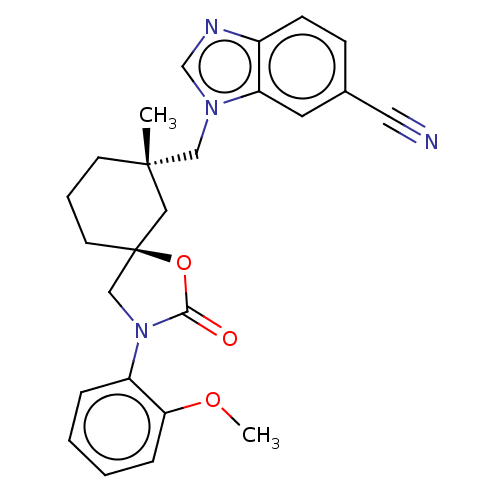
(CHEMBL4521512)Show SMILES COc1ccccc1N1C[C@@]2(CCC[C@](C)(Cn3cnc4ccc(cc34)C#N)C2)OC1=O |r| Show InChI InChI=1S/C25H26N4O3/c1-24(15-28-17-27-19-9-8-18(13-26)12-21(19)28)10-5-11-25(14-24)16-29(23(30)32-25)20-6-3-4-7-22(20)31-2/h3-4,6-9,12,17H,5,10-11,14-16H2,1-2H3/t24-,25-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human TRPV4 expressed in baculovirus infected HEK/MSR 2 cells pre-incubated for 10 mins followed by GSK634775A addition by Fura-2 dye b... |
ACS Med Chem Lett 10: 1228-1233 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00274
BindingDB Entry DOI: 10.7270/Q2RX9GBF |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50502648
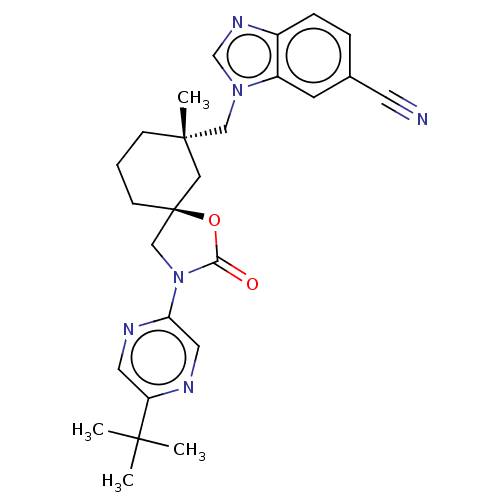
(CHEMBL4588831)Show SMILES CC(C)(C)c1cnc(cn1)N1C[C@@]2(CCC[C@](C)(Cn3cnc4ccc(cc34)C#N)C2)OC1=O |r| Show InChI InChI=1S/C26H30N6O2/c1-24(2,3)21-12-29-22(13-28-21)32-16-26(34-23(32)33)9-5-8-25(4,14-26)15-31-17-30-19-7-6-18(11-27)10-20(19)31/h6-7,10,12-13,17H,5,8-9,14-16H2,1-4H3/t25-,26-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human TRPV4 expressed in baculovirus infected HEK/MSR 2 cells pre-incubated for 10 mins followed by GSK634775A addition by Fura-2 dye b... |
ACS Med Chem Lett 10: 1228-1233 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00274
BindingDB Entry DOI: 10.7270/Q2RX9GBF |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50502626
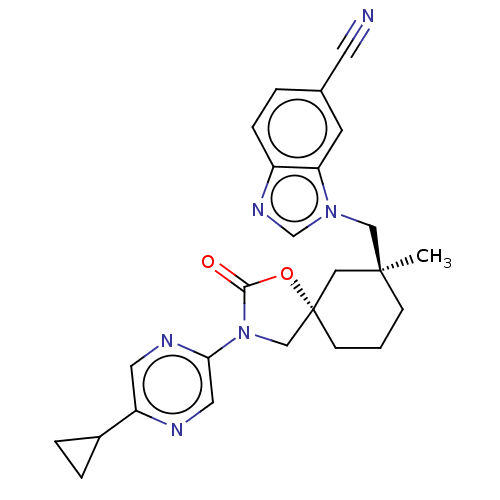
(CHEMBL4536058)Show SMILES C[C@]1(Cn2cnc3ccc(cc23)C#N)CCC[C@@]2(CN(C(=O)O2)c2cnc(cn2)C2CC2)C1 |r| Show InChI InChI=1S/C25H26N6O2/c1-24(14-30-16-29-19-6-3-17(10-26)9-21(19)30)7-2-8-25(13-24)15-31(23(32)33-25)22-12-27-20(11-28-22)18-4-5-18/h3,6,9,11-12,16,18H,2,4-5,7-8,13-15H2,1H3/t24-,25-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human TRPV4 expressed in baculovirus infected HEK/MSR 2 cells pre-incubated for 10 mins followed by GSK634775A addition by Fura-2 dye b... |
ACS Med Chem Lett 10: 1228-1233 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00274
BindingDB Entry DOI: 10.7270/Q2RX9GBF |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50232815

(CHEMBL4104743)Show SMILES CCOc1ccc2c(C(=O)NC3(CC3)c3ccccc3)c(CN3CCC(CC3)N3CCCCC3)c(nc2c1)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C39H43F3N4O2/c1-2-48-31-14-15-32-34(25-31)43-36(27-10-9-13-29(24-27)39(40,41)42)33(26-45-22-16-30(17-23-45)46-20-7-4-8-21-46)35(32)37(47)44-38(18-19-38)28-11-5-3-6-12-28/h3,5-6,9-15,24-25,30H,2,4,7-8,16-23,26H2,1H3,(H,44,47) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Antagonist activity at rat TRPV4 expressed in HEK293 cells assessed as inhibition of GSK634775A-induced calcium influx preincubated for 10 mins follo... |
ACS Med Chem Lett 8: 549-554 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00094
BindingDB Entry DOI: 10.7270/Q2HD7XWW |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50502640

(CHEMBL4470585)Show SMILES CC(C)(O)c1cnc(cn1)N1C[C@@]2(CCC[C@](C)(Cn3cnc4ccc(cc34)C#N)C2)OC1=O |r| Show InChI InChI=1S/C25H28N6O3/c1-23(2,33)20-11-28-21(12-27-20)31-15-25(34-22(31)32)8-4-7-24(3,13-25)14-30-16-29-18-6-5-17(10-26)9-19(18)30/h5-6,9,11-12,16,33H,4,7-8,13-15H2,1-3H3/t24-,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of rat TRPV4 expressed in baculovirus infected HEK/MSR 2 cells pre-incubated for 10 mins followed by GSK634775A addition by Fura-2 dye bas... |
ACS Med Chem Lett 10: 1228-1233 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00274
BindingDB Entry DOI: 10.7270/Q2RX9GBF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50502649
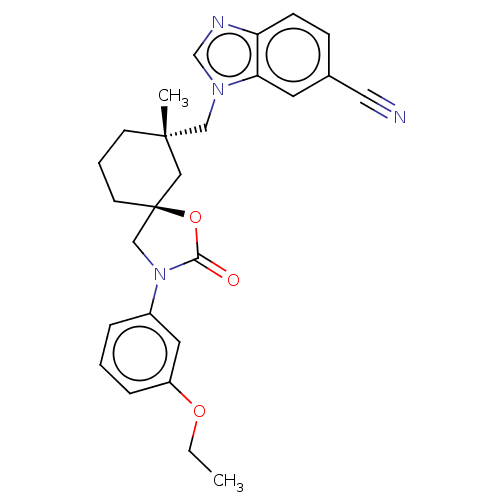
(CHEMBL4532369)Show SMILES CCOc1cccc(c1)N1C[C@@]2(CCC[C@](C)(Cn3cnc4ccc(cc34)C#N)C2)OC1=O |r| Show InChI InChI=1S/C26H28N4O3/c1-3-32-21-7-4-6-20(13-21)30-17-26(33-24(30)31)11-5-10-25(2,15-26)16-29-18-28-22-9-8-19(14-27)12-23(22)29/h4,6-9,12-13,18H,3,5,10-11,15-17H2,1-2H3/t25-,26-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human TRPV4 expressed in baculovirus infected HEK/MSR 2 cells pre-incubated for 10 mins followed by GSK634775A addition by Fura-2 dye b... |
ACS Med Chem Lett 10: 1228-1233 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00274
BindingDB Entry DOI: 10.7270/Q2RX9GBF |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50502643
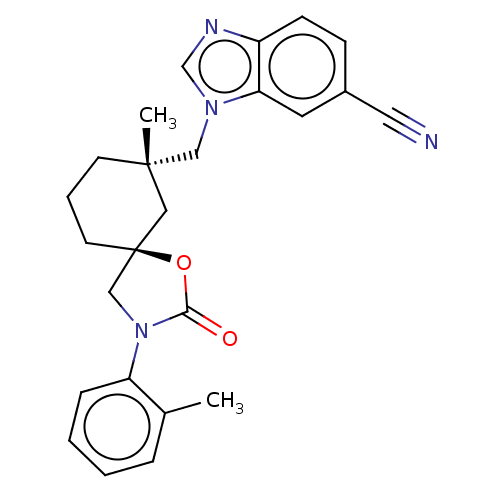
(CHEMBL4541827)Show SMILES Cc1ccccc1N1C[C@@]2(CCC[C@](C)(Cn3cnc4ccc(cc34)C#N)C2)OC1=O |r| Show InChI InChI=1S/C25H26N4O2/c1-18-6-3-4-7-21(18)29-16-25(31-23(29)30)11-5-10-24(2,14-25)15-28-17-27-20-9-8-19(13-26)12-22(20)28/h3-4,6-9,12,17H,5,10-11,14-16H2,1-2H3/t24-,25-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human TRPV4 expressed in baculovirus infected HEK/MSR 2 cells pre-incubated for 10 mins followed by GSK634775A addition by Fura-2 dye b... |
ACS Med Chem Lett 10: 1228-1233 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00274
BindingDB Entry DOI: 10.7270/Q2RX9GBF |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50502640

(CHEMBL4470585)Show SMILES CC(C)(O)c1cnc(cn1)N1C[C@@]2(CCC[C@](C)(Cn3cnc4ccc(cc34)C#N)C2)OC1=O |r| Show InChI InChI=1S/C25H28N6O3/c1-23(2,33)20-11-28-21(12-27-20)31-15-25(34-22(31)32)8-4-7-24(3,13-25)14-30-16-29-18-6-5-17(10-26)9-19(18)30/h5-6,9,11-12,16,33H,4,7-8,13-15H2,1-3H3/t24-,25-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human TRPV4 expressed in baculovirus infected HEK/MSR 2 cells pre-incubated for 10 mins followed by GSK634775A addition by Fura-2 dye b... |
ACS Med Chem Lett 10: 1228-1233 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00274
BindingDB Entry DOI: 10.7270/Q2RX9GBF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50232797
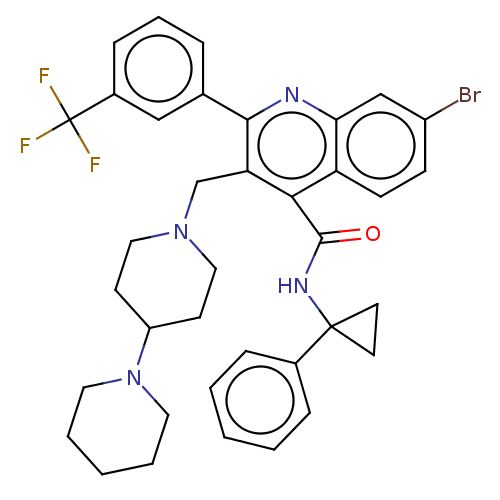
(CHEMBL4073922)Show SMILES FC(F)(F)c1cccc(c1)-c1nc2cc(Br)ccc2c(C(=O)NC2(CC2)c2ccccc2)c1CN1CCC(CC1)N1CCCCC1 Show InChI InChI=1S/C37H38BrF3N4O/c38-28-12-13-30-32(23-28)42-34(25-8-7-11-27(22-25)37(39,40)41)31(24-44-20-14-29(15-21-44)45-18-5-2-6-19-45)33(30)35(46)43-36(16-17-36)26-9-3-1-4-10-26/h1,3-4,7-13,22-23,29H,2,5-6,14-21,24H2,(H,43,46) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Antagonist activity at rat TRPV4 expressed in HEK293 cells assessed as inhibition of GSK634775A-induced calcium influx preincubated for 10 mins follo... |
ACS Med Chem Lett 8: 549-554 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00094
BindingDB Entry DOI: 10.7270/Q2HD7XWW |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50232804
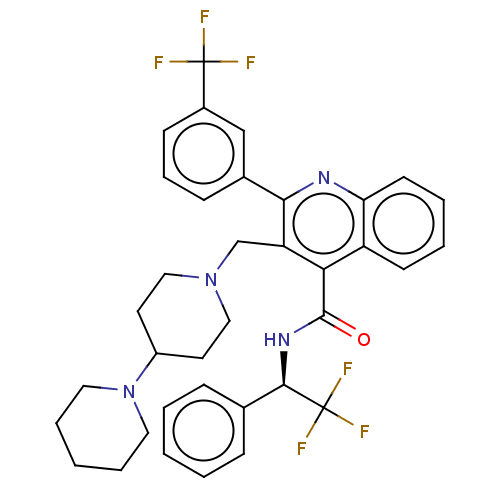
(CHEMBL4087716)Show SMILES FC(F)(F)[C@H](NC(=O)c1c(CN2CCC(CC2)N2CCCCC2)c(nc2ccccc12)-c1cccc(c1)C(F)(F)F)c1ccccc1 |r| Show InChI InChI=1S/C36H36F6N4O/c37-35(38,39)26-13-9-12-25(22-26)32-29(23-45-20-16-27(17-21-45)46-18-7-2-8-19-46)31(28-14-5-6-15-30(28)43-32)34(47)44-33(36(40,41)42)24-10-3-1-4-11-24/h1,3-6,9-15,22,27,33H,2,7-8,16-21,23H2,(H,44,47)/t33-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Antagonist activity at rat TRPV4 expressed in HEK293 cells assessed as inhibition of GSK634775A-induced calcium influx preincubated for 10 mins follo... |
ACS Med Chem Lett 8: 549-554 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00094
BindingDB Entry DOI: 10.7270/Q2HD7XWW |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50232813
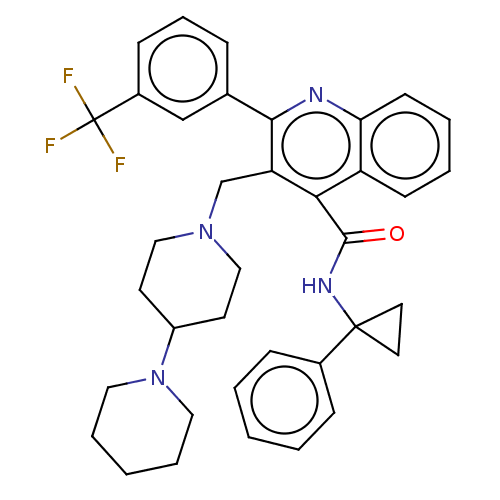
(CHEMBL4097859)Show SMILES FC(F)(F)c1cccc(c1)-c1nc2ccccc2c(C(=O)NC2(CC2)c2ccccc2)c1CN1CCC(CC1)N1CCCCC1 Show InChI InChI=1S/C37H39F3N4O/c38-37(39,40)28-13-9-10-26(24-28)34-31(25-43-22-16-29(17-23-43)44-20-7-2-8-21-44)33(30-14-5-6-15-32(30)41-34)35(45)42-36(18-19-36)27-11-3-1-4-12-27/h1,3-6,9-15,24,29H,2,7-8,16-23,25H2,(H,42,45) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Antagonist activity at rat TRPV4 expressed in HEK293 cells assessed as inhibition of GSK634775A-induced calcium influx preincubated for 10 mins follo... |
ACS Med Chem Lett 8: 549-554 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00094
BindingDB Entry DOI: 10.7270/Q2HD7XWW |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50232800
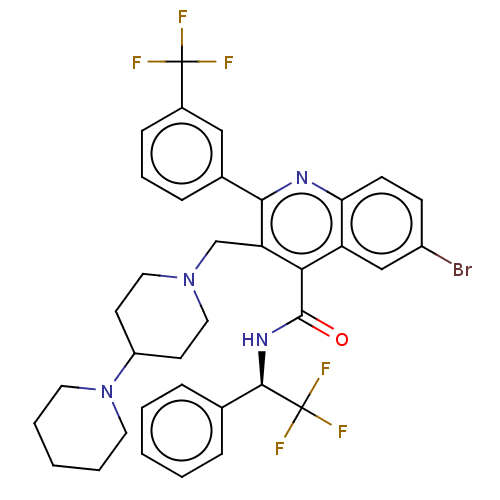
(CHEMBL4103166)Show SMILES FC(F)(F)[C@H](NC(=O)c1c(CN2CCC(CC2)N2CCCCC2)c(nc2ccc(Br)cc12)-c1cccc(c1)C(F)(F)F)c1ccccc1 |r| Show InChI InChI=1S/C36H35BrF6N4O/c37-26-12-13-30-28(21-26)31(34(48)45-33(36(41,42)43)23-8-3-1-4-9-23)29(32(44-30)24-10-7-11-25(20-24)35(38,39)40)22-46-18-14-27(15-19-46)47-16-5-2-6-17-47/h1,3-4,7-13,20-21,27,33H,2,5-6,14-19,22H2,(H,45,48)/t33-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Antagonist activity at rat TRPV4 expressed in HEK293 cells assessed as inhibition of GSK634775A-induced calcium influx preincubated for 10 mins follo... |
ACS Med Chem Lett 8: 549-554 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00094
BindingDB Entry DOI: 10.7270/Q2HD7XWW |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50232796

(CHEMBL4094483)Show SMILES COc1ccc2c(C(=O)NC3(CC3)c3ccccc3)c(CN3CCC(CC3)N3CCCCC3)c(nc2c1)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C38H41F3N4O2/c1-47-30-13-14-31-33(24-30)42-35(26-9-8-12-28(23-26)38(39,40)41)32(25-44-21-15-29(16-22-44)45-19-6-3-7-20-45)34(31)36(46)43-37(17-18-37)27-10-4-2-5-11-27/h2,4-5,8-14,23-24,29H,3,6-7,15-22,25H2,1H3,(H,43,46) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Antagonist activity at rat TRPV4 expressed in HEK293 cells assessed as inhibition of GSK634775A-induced calcium influx preincubated for 10 mins follo... |
ACS Med Chem Lett 8: 549-554 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00094
BindingDB Entry DOI: 10.7270/Q2HD7XWW |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50502653

(CHEMBL4514491)Show SMILES C[C@]1(Cn2cnc3ccc(cc23)C#N)CCC[C@@]2(CN(C(=O)O2)c2ccccc2Cl)C1 |r| Show InChI InChI=1S/C24H23ClN4O2/c1-23(14-28-16-27-19-8-7-17(12-26)11-21(19)28)9-4-10-24(13-23)15-29(22(30)31-24)20-6-3-2-5-18(20)25/h2-3,5-8,11,16H,4,9-10,13-15H2,1H3/t23-,24-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human TRPV4 expressed in baculovirus infected HEK/MSR 2 cells pre-incubated for 10 mins followed by GSK634775A addition by Fura-2 dye b... |
ACS Med Chem Lett 10: 1228-1233 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00274
BindingDB Entry DOI: 10.7270/Q2RX9GBF |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50232790
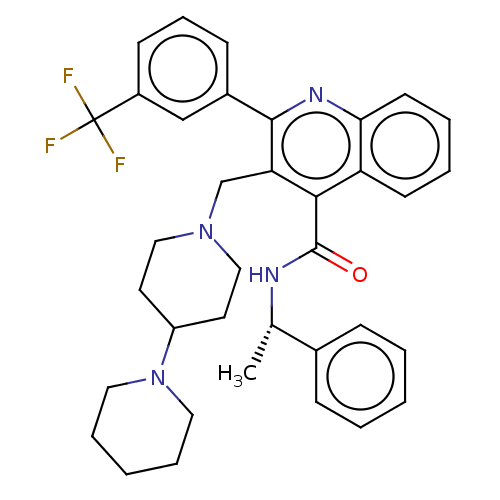
(CHEMBL4084542)Show SMILES C[C@H](NC(=O)c1c(CN2CCC(CC2)N2CCCCC2)c(nc2ccccc12)-c1cccc(c1)C(F)(F)F)c1ccccc1 |r| Show InChI InChI=1S/C36H39F3N4O/c1-25(26-11-4-2-5-12-26)40-35(44)33-30-15-6-7-16-32(30)41-34(27-13-10-14-28(23-27)36(37,38)39)31(33)24-42-21-17-29(18-22-42)43-19-8-3-9-20-43/h2,4-7,10-16,23,25,29H,3,8-9,17-22,24H2,1H3,(H,40,44)/t25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Antagonist activity at rat TRPV4 expressed in HEK293 cells assessed as inhibition of GSK634775A-induced calcium influx preincubated for 10 mins follo... |
ACS Med Chem Lett 8: 549-554 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00094
BindingDB Entry DOI: 10.7270/Q2HD7XWW |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50502636
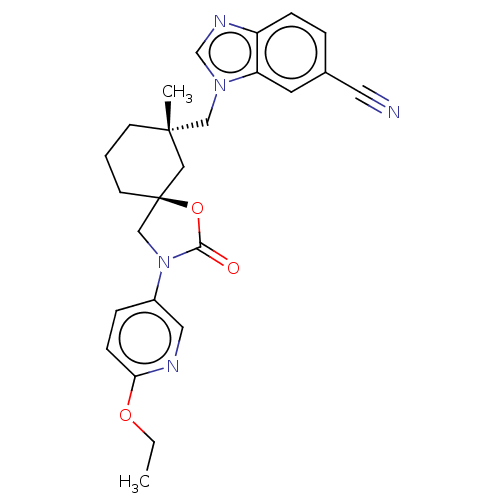
(CHEMBL4588158)Show SMILES CCOc1ccc(cn1)N1C[C@@]2(CCC[C@](C)(Cn3cnc4ccc(cc34)C#N)C2)OC1=O |r| Show InChI InChI=1S/C25H27N5O3/c1-3-32-22-8-6-19(13-27-22)30-16-25(33-23(30)31)10-4-9-24(2,14-25)15-29-17-28-20-7-5-18(12-26)11-21(20)29/h5-8,11,13,17H,3-4,9-10,14-16H2,1-2H3/t24-,25-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human TRPV4 expressed in baculovirus infected HEK/MSR 2 cells pre-incubated for 10 mins followed by GSK634775A addition by Fura-2 dye b... |
ACS Med Chem Lett 10: 1228-1233 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00274
BindingDB Entry DOI: 10.7270/Q2RX9GBF |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50426532
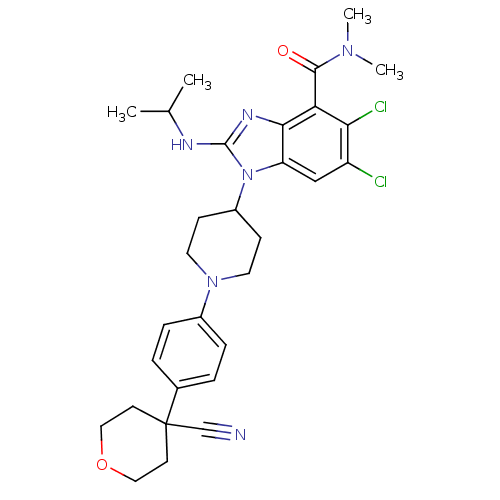
(CHEMBL2323936)Show SMILES CC(C)Nc1nc2c(C(=O)N(C)C)c(Cl)c(Cl)cc2n1C1CCN(CC1)c1ccc(cc1)C1(CCOCC1)C#N Show InChI InChI=1S/C30H36Cl2N6O2/c1-19(2)34-29-35-27-24(17-23(31)26(32)25(27)28(39)36(3)4)38(29)22-9-13-37(14-10-22)21-7-5-20(6-8-21)30(18-33)11-15-40-16-12-30/h5-8,17,19,22H,9-16H2,1-4H3,(H,34,35) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant TRPV4 expressed in BacMam virus infected HEK MSRII cells assessed as inhibition of GSK1016790A-induced calci... |
ACS Med Chem Lett 4: 293-6 (2013)
Article DOI: 10.1021/ml300449k
BindingDB Entry DOI: 10.7270/Q2VT1TDB |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50232799
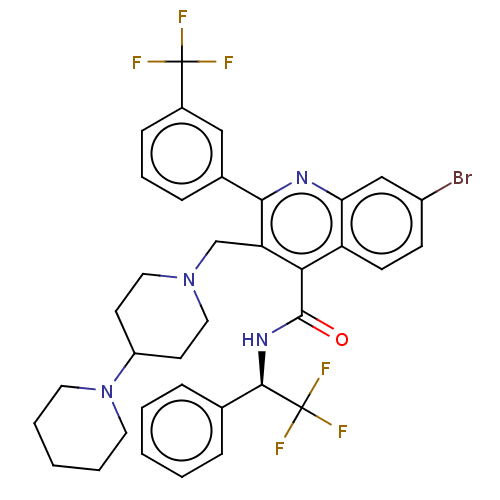
(CHEMBL4078886)Show SMILES FC(F)(F)[C@H](NC(=O)c1c(CN2CCC(CC2)N2CCCCC2)c(nc2cc(Br)ccc12)-c1cccc(c1)C(F)(F)F)c1ccccc1 |r| Show InChI InChI=1S/C36H35BrF6N4O/c37-26-12-13-28-30(21-26)44-32(24-10-7-11-25(20-24)35(38,39)40)29(22-46-18-14-27(15-19-46)47-16-5-2-6-17-47)31(28)34(48)45-33(36(41,42)43)23-8-3-1-4-9-23/h1,3-4,7-13,20-21,27,33H,2,5-6,14-19,22H2,(H,45,48)/t33-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Antagonist activity at rat TRPV4 expressed in HEK293 cells assessed as inhibition of GSK634775A-induced calcium influx preincubated for 10 mins follo... |
ACS Med Chem Lett 8: 549-554 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00094
BindingDB Entry DOI: 10.7270/Q2HD7XWW |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50232803
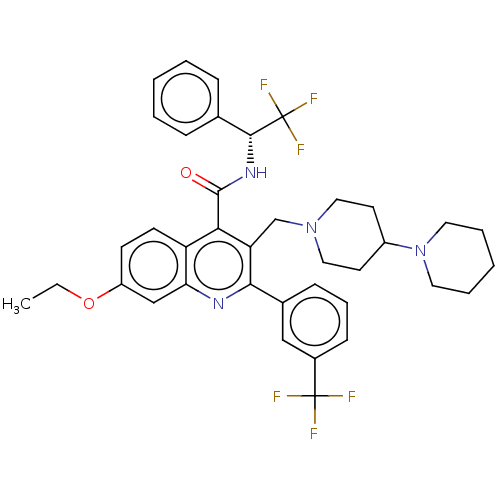
(CHEMBL4068023)Show SMILES CCOc1ccc2c(C(=O)N[C@H](c3ccccc3)C(F)(F)F)c(CN3CCC(CC3)N3CCCCC3)c(nc2c1)-c1cccc(c1)C(F)(F)F |r| Show InChI InChI=1S/C38H40F6N4O2/c1-2-50-29-14-15-30-32(23-29)45-34(26-12-9-13-27(22-26)37(39,40)41)31(24-47-20-16-28(17-21-47)48-18-7-4-8-19-48)33(30)36(49)46-35(38(42,43)44)25-10-5-3-6-11-25/h3,5-6,9-15,22-23,28,35H,2,4,7-8,16-21,24H2,1H3,(H,46,49)/t35-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Antagonist activity at rat TRPV4 expressed in HEK293 cells assessed as inhibition of GSK634775A-induced calcium influx preincubated for 10 mins follo... |
ACS Med Chem Lett 8: 549-554 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00094
BindingDB Entry DOI: 10.7270/Q2HD7XWW |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50232809
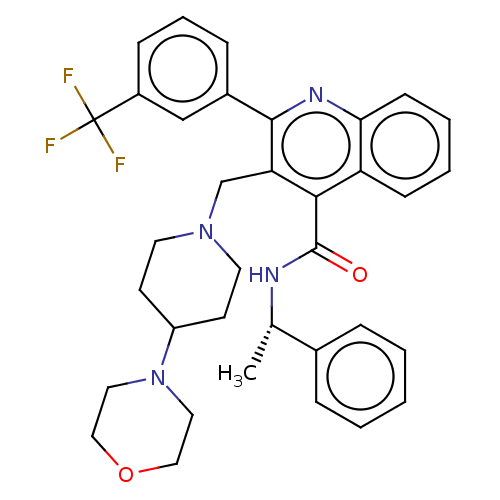
(CHEMBL4065154)Show SMILES C[C@H](NC(=O)c1c(CN2CCC(CC2)N2CCOCC2)c(nc2ccccc12)-c1cccc(c1)C(F)(F)F)c1ccccc1 |r| Show InChI InChI=1S/C35H37F3N4O2/c1-24(25-8-3-2-4-9-25)39-34(43)32-29-12-5-6-13-31(29)40-33(26-10-7-11-27(22-26)35(36,37)38)30(32)23-41-16-14-28(15-17-41)42-18-20-44-21-19-42/h2-13,22,24,28H,14-21,23H2,1H3,(H,39,43)/t24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Antagonist activity at rat TRPV4 expressed in HEK293 cells assessed as inhibition of GSK634775A-induced calcium influx preincubated for 10 mins follo... |
ACS Med Chem Lett 8: 549-554 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00094
BindingDB Entry DOI: 10.7270/Q2HD7XWW |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50232798
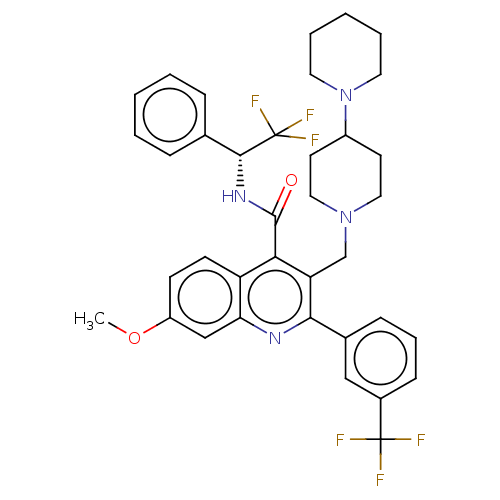
(CHEMBL4095413)Show SMILES COc1ccc2c(C(=O)N[C@H](c3ccccc3)C(F)(F)F)c(CN3CCC(CC3)N3CCCCC3)c(nc2c1)-c1cccc(c1)C(F)(F)F |r| Show InChI InChI=1S/C37H38F6N4O2/c1-49-28-13-14-29-31(22-28)44-33(25-11-8-12-26(21-25)36(38,39)40)30(23-46-19-15-27(16-20-46)47-17-6-3-7-18-47)32(29)35(48)45-34(37(41,42)43)24-9-4-2-5-10-24/h2,4-5,8-14,21-22,27,34H,3,6-7,15-20,23H2,1H3,(H,45,48)/t34-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Antagonist activity at rat TRPV4 expressed in HEK293 cells assessed as inhibition of GSK634775A-induced calcium influx preincubated for 10 mins follo... |
ACS Med Chem Lett 8: 549-554 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00094
BindingDB Entry DOI: 10.7270/Q2HD7XWW |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50502637

(CHEMBL4469982)Show SMILES CCOc1cnc(cn1)N1C[C@@]2(CCC[C@](C)(Cn3cnc4ccc(cc34)C#N)C2)OC1=O |r| Show InChI InChI=1S/C24H26N6O3/c1-3-32-21-12-26-20(11-27-21)30-15-24(33-22(30)31)8-4-7-23(2,13-24)14-29-16-28-18-6-5-17(10-25)9-19(18)29/h5-6,9,11-12,16H,3-4,7-8,13-15H2,1-2H3/t23-,24-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human TRPV4 expressed in baculovirus infected HEK/MSR 2 cells pre-incubated for 10 mins followed by GSK634775A addition by Fura-2 dye b... |
ACS Med Chem Lett 10: 1228-1233 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00274
BindingDB Entry DOI: 10.7270/Q2RX9GBF |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50502642
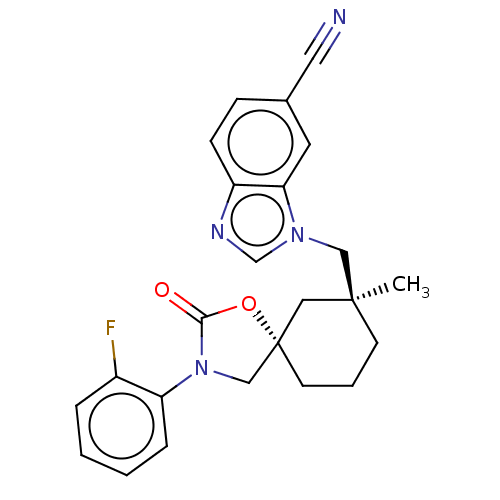
(CHEMBL4560266)Show SMILES C[C@]1(Cn2cnc3ccc(cc23)C#N)CCC[C@@]2(CN(C(=O)O2)c2ccccc2F)C1 |r| Show InChI InChI=1S/C24H23FN4O2/c1-23(14-28-16-27-19-8-7-17(12-26)11-21(19)28)9-4-10-24(13-23)15-29(22(30)31-24)20-6-3-2-5-18(20)25/h2-3,5-8,11,16H,4,9-10,13-15H2,1H3/t23-,24-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.20 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human TRPV4 expressed in baculovirus infected HEK/MSR 2 cells pre-incubated for 10 mins followed by GSK634775A addition by Fura-2 dye b... |
ACS Med Chem Lett 10: 1228-1233 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00274
BindingDB Entry DOI: 10.7270/Q2RX9GBF |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50502646
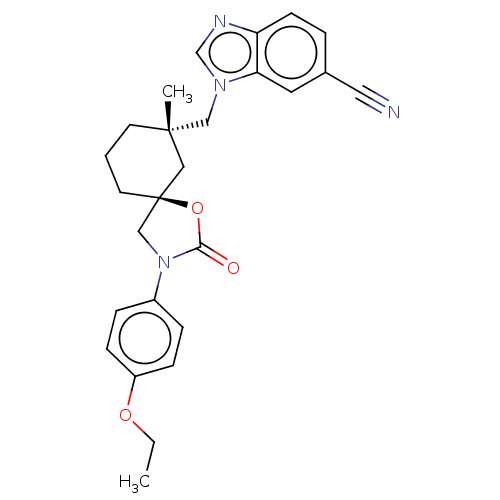
(CHEMBL4579838)Show SMILES CCOc1ccc(cc1)N1C[C@@]2(CCC[C@](C)(Cn3cnc4ccc(cc34)C#N)C2)OC1=O |r| Show InChI InChI=1S/C26H28N4O3/c1-3-32-21-8-6-20(7-9-21)30-17-26(33-24(30)31)12-4-11-25(2,15-26)16-29-18-28-22-10-5-19(14-27)13-23(22)29/h5-10,13,18H,3-4,11-12,15-17H2,1-2H3/t25-,26-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.20 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human TRPV4 expressed in baculovirus infected HEK/MSR 2 cells pre-incubated for 10 mins followed by GSK634775A addition by Fura-2 dye b... |
ACS Med Chem Lett 10: 1228-1233 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00274
BindingDB Entry DOI: 10.7270/Q2RX9GBF |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50502650
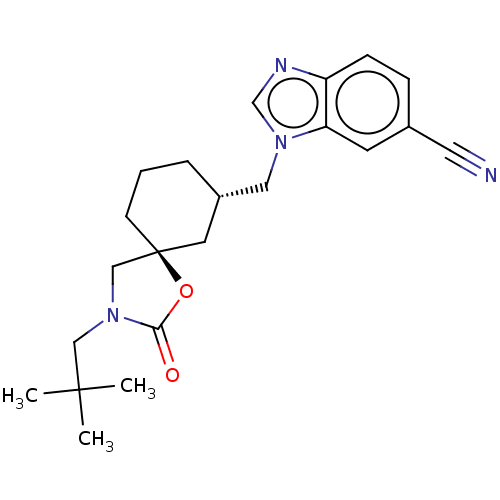
(CHEMBL4549613)Show SMILES CC(C)(C)CN1C[C@@]2(CCC[C@H](Cn3cnc4ccc(cc34)C#N)C2)OC1=O |r| Show InChI InChI=1S/C22H28N4O2/c1-21(2,3)13-26-14-22(28-20(26)27)8-4-5-17(10-22)12-25-15-24-18-7-6-16(11-23)9-19(18)25/h6-7,9,15,17H,4-5,8,10,12-14H2,1-3H3/t17-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of rat TRPV4 expressed in baculovirus infected HEK/MSR 2 cells pre-incubated for 10 mins followed by GSK634775A addition by Fura-2 dye bas... |
ACS Med Chem Lett 10: 1228-1233 (2019)
Article DOI: 10.1021/acsmedchemlett.9b00274
BindingDB Entry DOI: 10.7270/Q2RX9GBF |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50232805
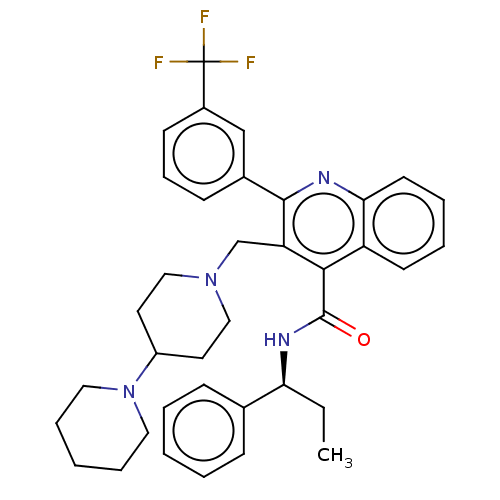
(CHEMBL4069979)Show SMILES CC[C@H](NC(=O)c1c(CN2CCC(CC2)N2CCCCC2)c(nc2ccccc12)-c1cccc(c1)C(F)(F)F)c1ccccc1 |r| Show InChI InChI=1S/C37H41F3N4O/c1-2-32(26-12-5-3-6-13-26)42-36(45)34-30-16-7-8-17-33(30)41-35(27-14-11-15-28(24-27)37(38,39)40)31(34)25-43-22-18-29(19-23-43)44-20-9-4-10-21-44/h3,5-8,11-17,24,29,32H,2,4,9-10,18-23,25H2,1H3,(H,42,45)/t32-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Antagonist activity at rat TRPV4 expressed in HEK293 cells assessed as inhibition of GSK634775A-induced calcium influx preincubated for 10 mins follo... |
ACS Med Chem Lett 8: 549-554 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00094
BindingDB Entry DOI: 10.7270/Q2HD7XWW |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Rattus norvegicus) | BDBM50232802
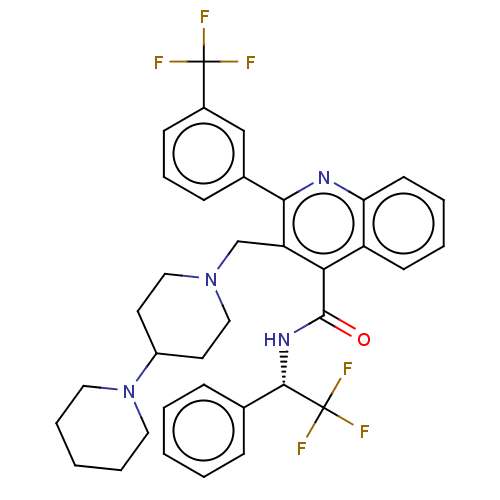
(CHEMBL4105593)Show SMILES FC(F)(F)[C@@H](NC(=O)c1c(CN2CCC(CC2)N2CCCCC2)c(nc2ccccc12)-c1cccc(c1)C(F)(F)F)c1ccccc1 |r| Show InChI InChI=1S/C36H36F6N4O/c37-35(38,39)26-13-9-12-25(22-26)32-29(23-45-20-16-27(17-21-45)46-18-7-2-8-19-46)31(28-14-5-6-15-30(28)43-32)34(47)44-33(36(40,41)42)24-10-3-1-4-11-24/h1,3-6,9-15,22,27,33H,2,7-8,16-21,23H2,(H,44,47)/t33-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Antagonist activity at rat TRPV4 expressed in HEK293 cells assessed as inhibition of GSK634775A-induced calcium influx preincubated for 10 mins follo... |
ACS Med Chem Lett 8: 549-554 (2017)
Article DOI: 10.1021/acsmedchemlett.7b00094
BindingDB Entry DOI: 10.7270/Q2HD7XWW |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data