 Found 37 hits in this display
Found 37 hits in this display Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
P2X purinoceptor 4
(Homo sapiens (Human)) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 27.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Max-Planck Institute for Experimental Medicine
Curated by PDSP Ki Database
| |
Mol Pharmacol 51: 109-18 (1997)
Article DOI: 10.1124/mol.51.1.109
BindingDB Entry DOI: 10.7270/Q218351B |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 4
(RAT) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Max-Planck Institute for Experimental Medicine
Curated by PDSP Ki Database
| |
Mol Pharmacol 51: 109-18 (1997)
Article DOI: 10.1124/mol.51.1.109
BindingDB Entry DOI: 10.7270/Q218351B |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 3
(Homo sapiens (Human)) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 3.47E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 407-16 (2004)
Article DOI: 10.1124/jpet.103.064907
BindingDB Entry DOI: 10.7270/Q2SQ8XZR |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 2
(Homo sapiens (Human)) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 4.02E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 310: 407-16 (2004)
Article DOI: 10.1124/jpet.103.064907
BindingDB Entry DOI: 10.7270/Q2SQ8XZR |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 1
(Mus musculus) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 99 | n/a | n/a | n/a | n/a | n/a | n/a |
Gwangju Institute of Science and Technology (GIST)
Curated by ChEMBL
| Assay Description
Antagonist activity at mouse P2X1 receptor |
Eur J Med Chem 70: 811-30 (2013)
Article DOI: 10.1016/j.ejmech.2013.10.026
BindingDB Entry DOI: 10.7270/Q2M61MQH |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 1
(RAT) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 99 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Inhibition of inward ion current elicited by ATP at P2X1 receptor expressed in Xenopus oocytes |
J Med Chem 44: 340-9 (2001)
BindingDB Entry DOI: 10.7270/Q2FN15HC |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 3
(Homo sapiens (Human)) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 240 | n/a | n/a | n/a | n/a | n/a | n/a |
Gwangju Institute of Science and Technology (GIST)
Curated by ChEMBL
| Assay Description
Antagonist activity at human P2X3 receptor |
Eur J Med Chem 70: 811-30 (2013)
Article DOI: 10.1016/j.ejmech.2013.10.026
BindingDB Entry DOI: 10.7270/Q2M61MQH |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 3
(RAT) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 240 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Inhibition of inward ion current elicited by ATP was determined at recombinant P2X3 receptor expressed in Xenopus oocytes |
J Med Chem 44: 340-9 (2001)
BindingDB Entry DOI: 10.7270/Q2FN15HC |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 1
(RAT) | BDBM50540409
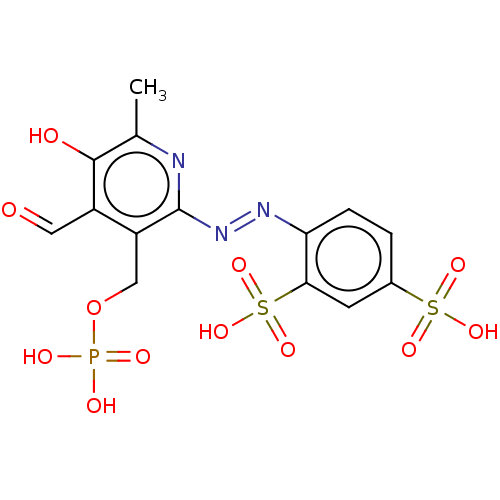
(CHEBI:34941 | CHEMBL69234)Show SMILES Cc1nc(\N=N\c2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28)/b17-16+ | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bonn
Curated by ChEMBL
| Assay Description
Antagonist activity at rat P2X1 receptor |
J Med Chem 63: 6164-6178 (2020)
Article DOI: 10.1021/acs.jmedchem.0c00435
BindingDB Entry DOI: 10.7270/Q24F1V93 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 7
(Homo sapiens (Human)) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 1.18E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Gwangju Institute of Science and Technology (GIST)
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human P2X7 receptor assessed as inhibition of BzATP-mediated Yo-Pro uptake measured for 1 hr by FLIPR assay |
Eur J Med Chem 70: 811-30 (2013)
Article DOI: 10.1016/j.ejmech.2013.10.026
BindingDB Entry DOI: 10.7270/Q2M61MQH |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
P2X purinoceptor 2
(RAT) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Inhibition of inward ion current elicited by ATP at P2X2 receptor expressed in Xenopus oocytes |
J Med Chem 44: 340-9 (2001)
BindingDB Entry DOI: 10.7270/Q2FN15HC |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 6
(Rattus norvegicus) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Inhibition of inward ion current elicited by ATP at rat P2X6 receptor (mutant type) |
J Med Chem 44: 340-9 (2001)
BindingDB Entry DOI: 10.7270/Q2FN15HC |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 2
(Homo sapiens (Human)) | BDBM50540409

(CHEBI:34941 | CHEMBL69234)Show SMILES Cc1nc(\N=N\c2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28)/b17-16+ | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 2.02E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114491
BindingDB Entry DOI: 10.7270/Q2BG2T1P |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 4
(RAT) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Inhibition of inward ion current elicited by ATP at rat P2X4 receptor (mutant type) |
J Med Chem 44: 340-9 (2001)
BindingDB Entry DOI: 10.7270/Q2FN15HC |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 4
(Homo sapiens (Human)) | BDBM50540409

(CHEBI:34941 | CHEMBL69234)Show SMILES Cc1nc(\N=N\c2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28)/b17-16+ | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 6.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114491
BindingDB Entry DOI: 10.7270/Q2BG2T1P |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 5
(Homo sapiens (Human)) | BDBM50540409

(CHEBI:34941 | CHEMBL69234)Show SMILES Cc1nc(\N=N\c2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28)/b17-16+ | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 8.45E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114491
BindingDB Entry DOI: 10.7270/Q2BG2T1P |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 1
(Meleagris gallopavo) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 1.66E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Inhibition of 10 nM 2-MeSADP-stimulated phospholipase C in turkey erythrocyte membranes using [3H]-inositol |
J Med Chem 44: 340-9 (2001)
BindingDB Entry DOI: 10.7270/Q2FN15HC |
More data for this
Ligand-Target Pair | |
P2X purinoceptor
(Mus musculus) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 2.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Inhibition of inward ion current elicited by ATP at mouse P2X4 receptor (mutant type) |
J Med Chem 44: 340-9 (2001)
BindingDB Entry DOI: 10.7270/Q2FN15HC |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 4
(Homo sapiens (Human)) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 2.75E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Inhibition of inward ion current elicited by ATP at human P2X4 receptor (mutant type) |
J Med Chem 44: 340-9 (2001)
BindingDB Entry DOI: 10.7270/Q2FN15HC |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 1
(RAT) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.85E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Compound was tested in a functional ion channel assay of ATP-induced current at recombinant rat P2X1 receptor expressed in Xenopus oocytes. |
J Med Chem 41: 2201-6 (1998)
Article DOI: 10.1021/jm980183o
BindingDB Entry DOI: 10.7270/Q2DB80ZC |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 3
(RAT) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Compound was tested in a functional ion channel assay of ATP-induced current at recombinant rat P2X3 receptor expressed in Xenopus oocytes. |
J Med Chem 41: 2201-6 (1998)
Article DOI: 10.1021/jm980183o
BindingDB Entry DOI: 10.7270/Q2DB80ZC |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 4
(RAT) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | >5.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Inhibition of inward ion current elicited by ATP at rat P2X4 receptor (wild type) |
J Med Chem 44: 340-9 (2001)
BindingDB Entry DOI: 10.7270/Q2FN15HC |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 6
(Rattus norvegicus) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | >5.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes and Digestive and Kidney Diseases
Curated by ChEMBL
| Assay Description
Inhibition of inward ion current elicited by ATP at rat P2X6 receptor (wild type) |
J Med Chem 44: 340-9 (2001)
BindingDB Entry DOI: 10.7270/Q2FN15HC |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 2
(RAT) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against recombinant rat receptor P2X purinoceptor 2 (P2X2) at 30 uM,expressed in Xenopus oocytes |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 4
(RAT) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Antagonist activity against recombinant rat P2X purinoceptor 4 (P2X4) at 3 uM,expressed in Xenopus oocytes |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 4
(Homo sapiens (Human)) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Antagonist activity against recombinant human P2X purinoceptor 4 (P2X4) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 6
(Rattus norvegicus) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Antagonist activity against recombinant rat P2X purinoceptor 6 (P2X6 ) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 1
(Homo sapiens (Human)) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Evaluated for agonist activity against phospholipase C coupled P2Y purinoceptor 1 (P2Y1) of human erythrocytes |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 12
(Rattus norvegicus) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Antagonist activity against phospholipase C coupled rat P2Y purinoceptor 12 (P2Y12) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 3
(Homo sapiens (Human)) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against recombinant rat P2X purinoceptor 3 (P2X3) at 10 uM, expressed in Xenopus oocytes |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 1
(RAT) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 99 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against recombinant rat P2X purinoceptor 1 (P2X1) at 1 uM,expressed in Xenopus oocytes |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 7
(Rattus norvegicus (Rat)) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 6.20E+4 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against recombinant rat P2X purinoceptor 7 (P2X7) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 2
(Homo sapiens (Human)) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Evaluated for agonist activity against phospholipase C coupled recombinant human P2Y purinoceptor 2 (P2Y2) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 4
(Homo sapiens (Human)) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against phospholipase C coupled recombinant human P2Y purinoceptor 4 (P2Y4) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 5
(Rattus norvegicus) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Antagonist activity against recombinant rat P2X purinoceptor 5 (P2X5) |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2X purinoceptor 3
(Homo sapiens (Human)) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 240 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
The compound was evaluated for antagonist activity against recombinant rat P2X purinoceptor 3 (P2X3) at 10 uM,expressed in Xenopus oocytes |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |
P2Y purinoceptor 1
(Homo sapiens (Human)) | BDBM85043

(CAS_149017-66-3 | CHEMBL69234 | NSC_6093163 | PPAD...)Show SMILES Cc1nc(N=Nc2ccc(cc2S(O)(=O)=O)S(O)(=O)=O)c(COP(O)(O)=O)c(C=O)c1O |w:4.3| Show InChI InChI=1S/C14H14N3O12PS2/c1-7-13(19)9(5-18)10(6-29-30(20,21)22)14(15-7)17-16-11-3-2-8(31(23,24)25)4-12(11)32(26,27)28/h2-5,19H,6H2,1H3,(H2,20,21,22)(H,23,24,25)(H,26,27,28) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | n/a | n/a | 1.82E+4 | n/a | n/a | n/a | n/a |
National Institute of Diabetes
Curated by ChEMBL
| Assay Description
Evaluated for agonist activity against phospholipase C coupled P2Y purinoceptor 1 (P2Y1) of rat erythrocytes |
J Med Chem 45: 4057-93 (2002)
BindingDB Entry DOI: 10.7270/Q2VX0H71 |
More data for this
Ligand-Target Pair | |