 Found 10982 hits Enz. Inhib. hit(s) with Target = 'Cholinesterase'
Found 10982 hits Enz. Inhib. hit(s) with Target = 'Cholinesterase' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cholinesterase
(Homo sapiens (Human)) | BDBM50247011

(CHEMBL4080419)Show SMILES Cl.CN(C)CCN(CC1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2ccccc2c1 Show InChI InChI=1S/C30H37N3O.ClH/c1-31(2)16-17-33(30(34)28-14-13-24-9-3-4-10-25(24)18-28)22-23-8-7-15-32(21-23)29-19-26-11-5-6-12-27(26)20-29;/h3-6,9-14,18,23,29H,7-8,15-17,19-22H2,1-2H3;1H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Competitive inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate at pH 8 by stopped flow assay |
J Med Chem 61: 119-139 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01086
BindingDB Entry DOI: 10.7270/Q2S75JRJ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50247011

(CHEMBL4080419)Show SMILES Cl.CN(C)CCN(CC1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2ccccc2c1 Show InChI InChI=1S/C30H37N3O.ClH/c1-31(2)16-17-33(30(34)28-14-13-24-9-3-4-10-25(24)18-28)22-23-8-7-15-32(21-23)29-19-26-11-5-6-12-27(26)20-29;/h3-6,9-14,18,23,29H,7-8,15-17,19-22H2,1-2H3;1H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Competitive inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate at pH 8 by stopped flow assay |
J Med Chem 61: 119-139 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01086
BindingDB Entry DOI: 10.7270/Q2S75JRJ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50005193
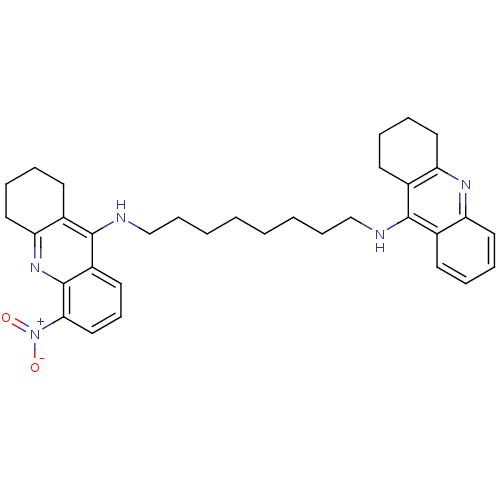
(CHEMBL3099496)Show SMILES [O-][N+](=O)c1cccc2c(NCCCCCCCCNc3c4CCCCc4nc4ccccc34)c3CCCCc3nc12 Show InChI InChI=1S/C34H41N5O2/c40-39(41)31-21-13-17-27-33(26-16-7-10-20-30(26)38-34(27)31)36-23-12-4-2-1-3-11-22-35-32-24-14-5-8-18-28(24)37-29-19-9-6-15-25(29)32/h5,8,13-14,17-18,21H,1-4,6-7,9-12,15-16,19-20,22-23H2,(H,35,37)(H,36,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human butyrylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50599186
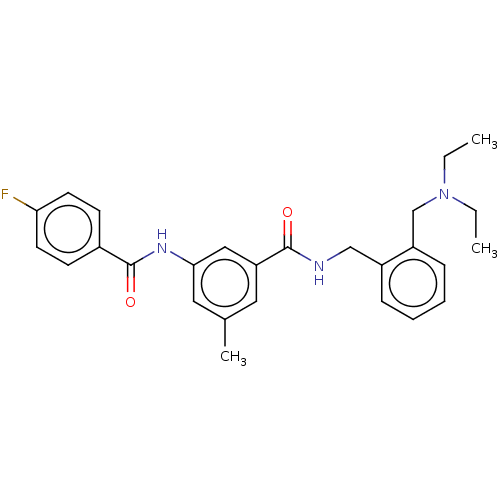
(CHEMBL5201089)Show SMILES CCN(CC)Cc1ccccc1CNC(=O)c1cc(C)cc(NC(=O)c2ccc(F)cc2)c1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00944
BindingDB Entry DOI: 10.7270/Q2HX1HP4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50234795
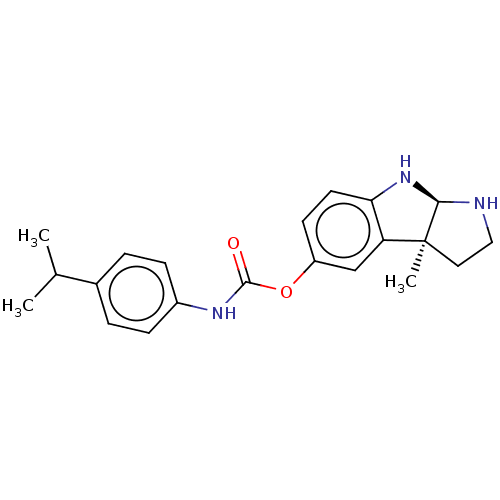
(CHEMBL4089082)Show SMILES [H][C@]12NCC[C@@]1(C)c1cc(OC(=O)Nc3ccc(cc3)C(C)C)ccc1N2 |r| Show InChI InChI=1S/C21H25N3O2/c1-13(2)14-4-6-15(7-5-14)23-20(25)26-16-8-9-18-17(12-16)21(3)10-11-22-19(21)24-18/h4-9,12-13,19,22,24H,10-11H2,1-3H3,(H,23,25)/t19-,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.131 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Martin-Luther University Halle-Wittenberg
Curated by ChEMBL
| Assay Description
Inhibition of human serum BChE using butyrylthiocholine iodide as substrate by Ellman's method |
Eur J Med Chem 126: 652-668 (2017)
Article DOI: 10.1016/j.ejmech.2016.11.056
BindingDB Entry DOI: 10.7270/Q2X0698K |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM8963
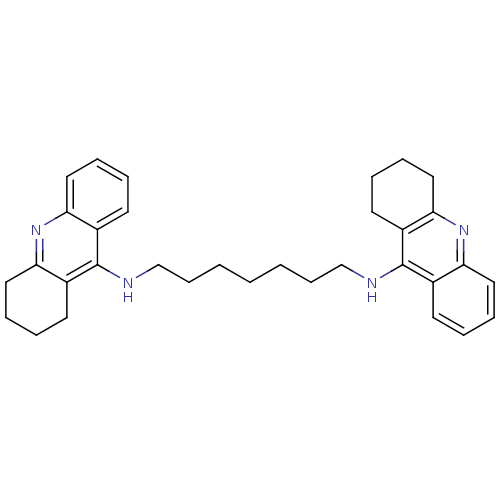
(CHEMBL32823 | Homodimeric Tacrine Analog 3b | N-[7...)Show SMILES C(CCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C33H40N4/c1(2-12-22-34-32-24-14-4-8-18-28(24)36-29-19-9-5-15-25(29)32)3-13-23-35-33-26-16-6-10-20-30(26)37-31-21-11-7-17-27(31)33/h4,6,8,10,14,16,18,20H,1-3,5,7,9,11-13,15,17,19,21-23H2,(H,34,36)(H,35,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.163 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Competitive inhibition of recombinant human BChE using butyrylthiocholine iodide as substrate at pH 8 by stopped flow assay |
J Med Chem 61: 119-139 (2018)
Article DOI: 10.1021/acs.jmedchem.7b01086
BindingDB Entry DOI: 10.7270/Q2S75JRJ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50604405
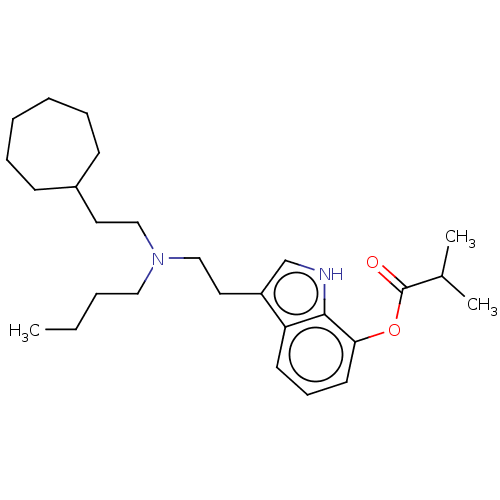
(CHEMBL5186889)Show SMILES CCCCN(CCC1CCCCCC1)CCc1c[nH]c2c(OC(=O)C(C)C)cccc12 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114248
BindingDB Entry DOI: 10.7270/Q2125XRJ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50604405

(CHEMBL5186889)Show SMILES CCCCN(CCC1CCCCCC1)CCc1c[nH]c2c(OC(=O)C(C)C)cccc12 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114248
BindingDB Entry DOI: 10.7270/Q2125XRJ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50271323
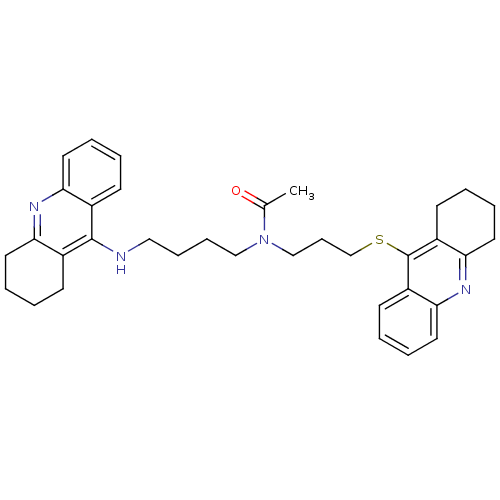
(CHEMBL501587 | N-{4-[(1,2,3, 4-Tetrahydroacridin-9...)Show SMILES CC(=O)N(CCCCNc1c2CCCCc2nc2ccccc12)CCCSc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C35H42N4OS/c1-25(40)39(23-12-24-41-35-28-15-4-8-19-32(28)38-33-20-9-5-16-29(33)35)22-11-10-21-36-34-26-13-2-6-17-30(26)37-31-18-7-3-14-27(31)34/h2,4,6,8,13,15,17,19H,3,5,7,9-12,14,16,18,20-24H2,1H3,(H,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM8971
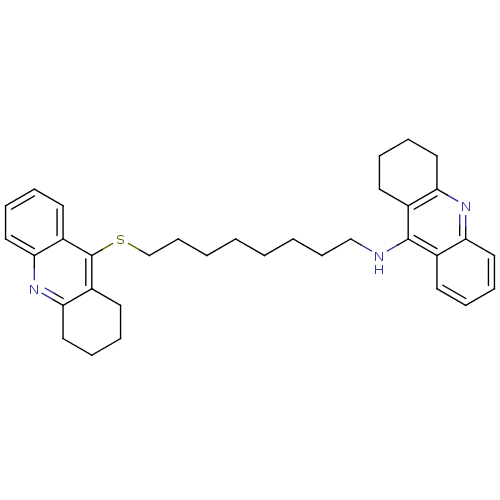
(CHEMBL129108 | N-[8-(1,2,3,4-tetrahydroacridin-9-y...)Show SMILES C(CCCCSc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H41N3S/c1(3-13-23-35-33-25-15-5-9-19-29(25)36-30-20-10-6-16-26(30)33)2-4-14-24-38-34-27-17-7-11-21-31(27)37-32-22-12-8-18-28(32)34/h5,7,9,11,15,17,19,21H,1-4,6,8,10,12-14,16,18,20,22-24H2,(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of Equine Butyrylcholinesterase |
J Med Chem 46: 1-4 (2002)
Article DOI: 10.1021/jm0255668
BindingDB Entry DOI: 10.7270/Q2GF0V7X |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50271468
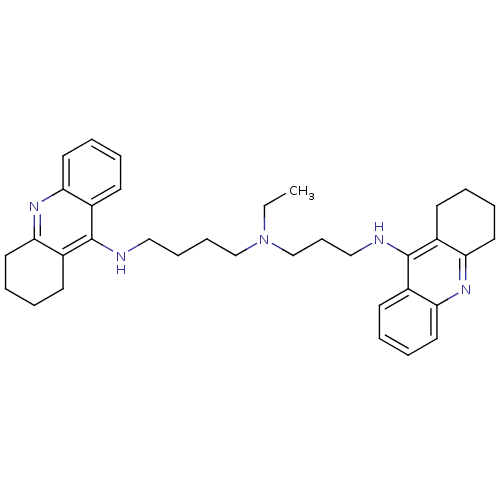
(CHEMBL490060 | N-Ethyl-N-(1,2,3,4-tetrahydroacridi...)Show SMILES CCN(CCCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C35H45N5/c1-2-40(25-13-23-37-35-28-16-5-9-20-32(28)39-33-21-10-6-17-29(33)35)24-12-11-22-36-34-26-14-3-7-18-30(26)38-31-19-8-4-15-27(31)34/h3,5,7,9,14,16,18,20H,2,4,6,8,10-13,15,17,19,21-25H2,1H3,(H,36,38)(H,37,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.435 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Mus musculus (Mouse)) | BDBM50377920
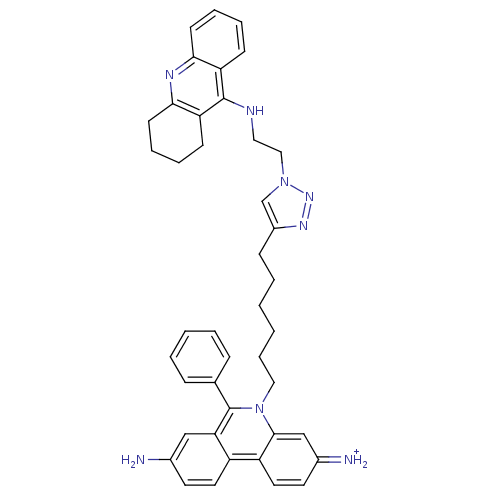
(CHEMBL540657)Show SMILES Nc1ccc2c(c1)c(-c1ccccc1)n(CCCCCCc1cn(CCNc3c4CCCCc4nc4ccccc34)nn1)c1cc(=[NH2+])ccc21 Show InChI InChI=1S/C42H44N8/c43-30-19-21-33-34-22-20-31(44)27-40(34)50(42(37(33)26-30)29-12-4-3-5-13-29)24-11-2-1-6-14-32-28-49(48-47-32)25-23-45-41-35-15-7-9-17-38(35)46-39-18-10-8-16-36(39)41/h3-5,7,9,12-13,15,17,19-22,26-28,44H,1-2,6,8,10-11,14,16,18,23-25,43H2,(H,45,46)/p+1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of mouse BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50599167
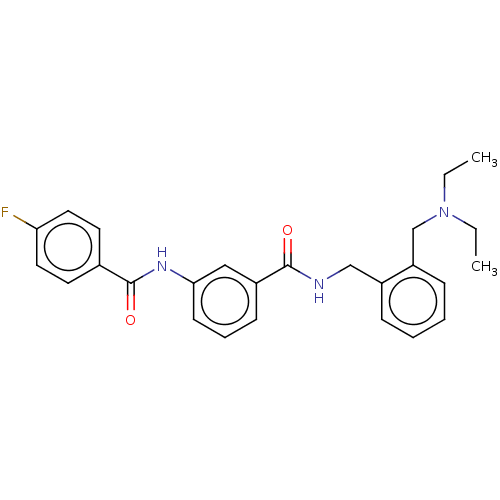
(CHEMBL5195228)Show SMILES CCN(CC)Cc1ccccc1CNC(=O)c1cccc(NC(=O)c2ccc(F)cc2)c1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.610 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00944
BindingDB Entry DOI: 10.7270/Q2HX1HP4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50210854
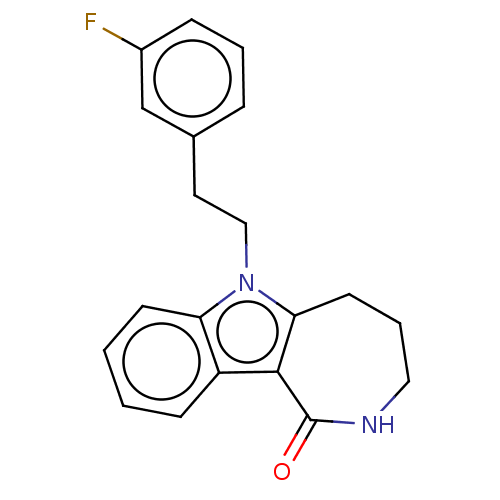
(CHEMBL3960040)Show InChI InChI=1S/C20H19FN2O/c21-15-6-3-5-14(13-15)10-12-23-17-8-2-1-7-16(17)19-18(23)9-4-11-22-20(19)24/h1-3,5-8,13H,4,9-12H2,(H,22,24) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bari"A. Moro"
Curated by ChEMBL
| Assay Description
Competitive inhibition of horse serum BChE in presence of varying levels of butyrylthiocholine iodide substrate by Lineweaver-burk plot method |
Eur J Med Chem 125: 288-298 (2017)
Article DOI: 10.1016/j.ejmech.2016.09.037
BindingDB Entry DOI: 10.7270/Q2G44S9D |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Mus musculus (Mouse)) | BDBM50149201
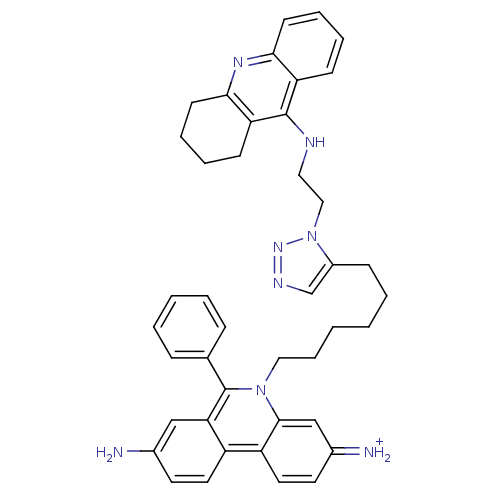
(3,8-DIAMINO-6-PHENYL-5-[6-[1-[2-[(1,2,3,4-TETRAHYD...)Show SMILES Nc1ccc2c(c1)c(-c1ccccc1)n(CCCCCCc1cnnn1CCNc1c3CCCCc3nc3ccccc13)c1cc(=[NH2+])ccc21 Show InChI InChI=1S/C42H44N8/c43-30-19-21-33-34-22-20-31(44)27-40(34)49(42(37(33)26-30)29-12-4-3-5-13-29)24-11-2-1-6-14-32-28-46-48-50(32)25-23-45-41-35-15-7-9-17-38(35)47-39-18-10-8-16-36(39)41/h3-5,7,9,12-13,15,17,19-22,26-28,44H,1-2,6,8,10-11,14,16,18,23-25,43H2,(H,45,47)/p+1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.720 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of mouse BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50271325
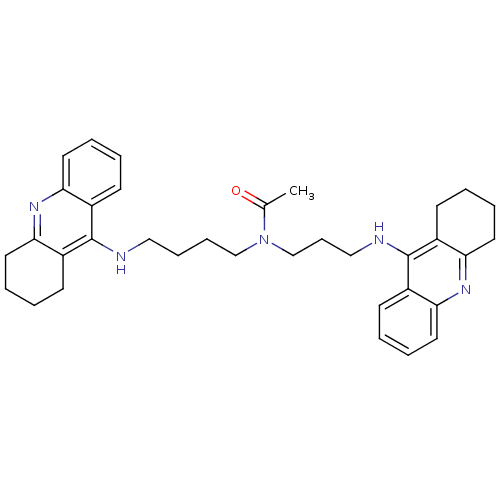
(CHEMBL451277 | N-{4-[(1,2,3,4-Tetrahydroacridin-9-...)Show SMILES CC(=O)N(CCCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C35H43N5O/c1-25(41)40(24-12-22-37-35-28-15-4-8-19-32(28)39-33-20-9-5-16-29(33)35)23-11-10-21-36-34-26-13-2-6-17-30(26)38-31-18-7-3-14-27(31)34/h2,4,6,8,13,15,17,19H,3,5,7,9-12,14,16,18,20-24H2,1H3,(H,36,38)(H,37,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.725 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM8977
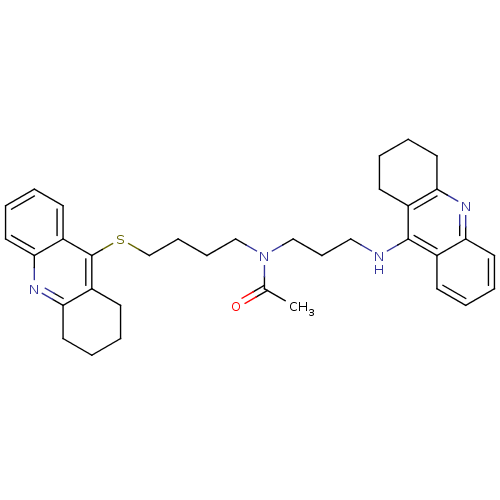
(CHEMBL175949 | N-[3-(1,2,3,4-Tetrahydroacridin-9-y...)Show SMILES CC(=O)N(CCCCSc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C35H42N4OS/c1-25(40)39(23-12-21-36-34-26-13-2-6-17-30(26)37-31-18-7-3-14-27(31)34)22-10-11-24-41-35-28-15-4-8-19-32(28)38-33-20-9-5-16-29(33)35/h2,4,6,8,13,15,17,19H,3,5,7,9-12,14,16,18,20-24H2,1H3,(H,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50271324
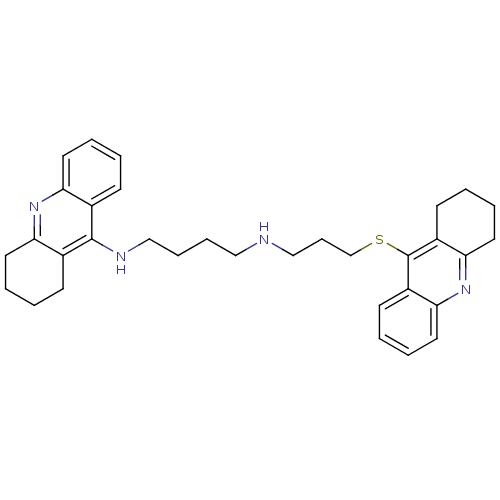
(CHEMBL482512 | N-(1,2,3,4-Tetrahydroacridin-9-yl)-...)Show SMILES C(CCNc1c2CCCCc2nc2ccccc12)CNCCCSc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C33H40N4S/c1-5-16-28-24(12-1)32(25-13-2-6-17-29(25)36-28)35-22-10-9-20-34-21-11-23-38-33-26-14-3-7-18-30(26)37-31-19-8-4-15-27(31)33/h1,3,5,7,12,14,16,18,34H,2,4,6,8-11,13,15,17,19-23H2,(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.779 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50271367
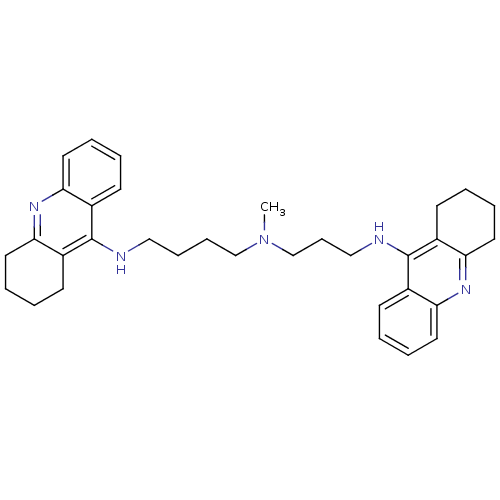
(CHEMBL489454 | N-Methyl-N-(1,2,3,4-tetrahydroacrid...)Show SMILES CN(CCCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H43N5/c1-39(24-12-22-36-34-27-15-4-8-19-31(27)38-32-20-9-5-16-28(32)34)23-11-10-21-35-33-25-13-2-6-17-29(25)37-30-18-7-3-14-26(30)33/h2,4,6,8,13,15,17,19H,3,5,7,9-12,14,16,18,20-24H2,1H3,(H,35,37)(H,36,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.819 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50271367

(CHEMBL489454 | N-Methyl-N-(1,2,3,4-tetrahydroacrid...)Show SMILES CN(CCCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H43N5/c1-39(24-12-22-36-34-27-15-4-8-19-31(27)38-32-20-9-5-16-28(32)34)23-11-10-21-35-33-25-13-2-6-17-29(25)37-30-18-7-3-14-26(30)33/h2,4,6,8,13,15,17,19H,3,5,7,9-12,14,16,18,20-24H2,1H3,(H,35,37)(H,36,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.820 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human butyrylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50271472
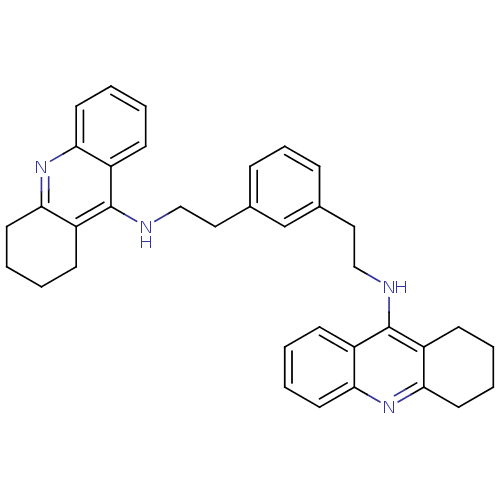
(1,3-bis{[(1,2,3,4-Tetrahydroacridin-9-yl)amino]eth...)Show SMILES C(Cc1cccc(CCNc2c3CCCCc3nc3ccccc23)c1)Nc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C36H38N4/c1-5-16-31-27(12-1)35(28-13-2-6-17-32(28)39-31)37-22-20-25-10-9-11-26(24-25)21-23-38-36-29-14-3-7-18-33(29)40-34-19-8-4-15-30(34)36/h1,3,5,7,9-12,14,16,18,24H,2,4,6,8,13,15,17,19-23H2,(H,37,39)(H,38,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.924 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50515472
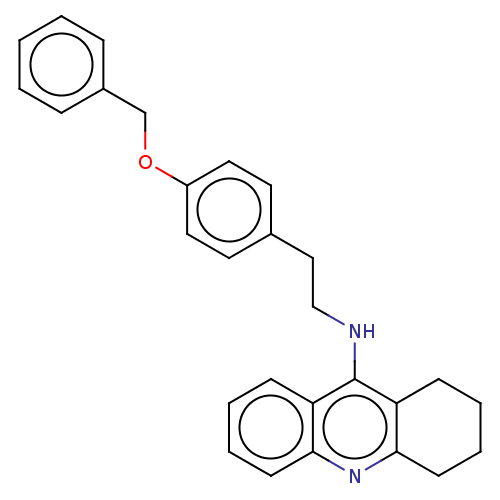
(CHEMBL4469822)Show SMILES C(Cc1ccc(OCc2ccccc2)cc1)Nc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C28H28N2O/c1-2-8-22(9-3-1)20-31-23-16-14-21(15-17-23)18-19-29-28-24-10-4-6-12-26(24)30-27-13-7-5-11-25(27)28/h1-4,6,8-10,12,14-17H,5,7,11,13,18-20H2,(H,29,30) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Sevilla
Curated by ChEMBL
| Assay Description
Inhibition of equine serum BuChE assessed as dissociation constant for enzyme-substrate-inhibitor complex using butyrylthiocholine iodide as substrat... |
Eur J Med Chem 181: (2019)
Article DOI: 10.1016/j.ejmech.2019.07.053
BindingDB Entry DOI: 10.7270/Q23R0X60 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50468733
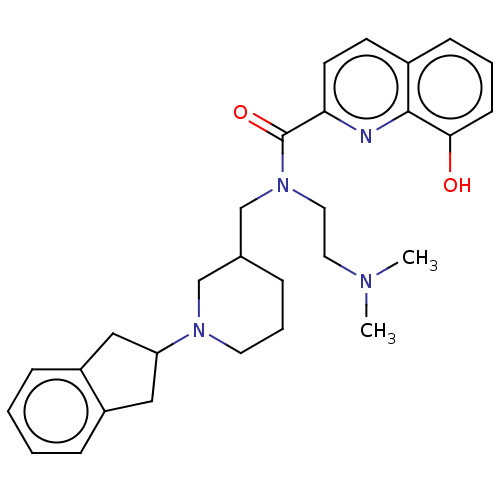
(CHEMBL4294570)Show SMILES CN(C)CCN(CC1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2cccc(O)c2n1 Show InChI InChI=1S/C29H36N4O2/c1-31(2)15-16-33(29(35)26-13-12-22-10-5-11-27(34)28(22)30-26)20-21-7-6-14-32(19-21)25-17-23-8-3-4-9-24(23)18-25/h3-5,8-13,21,25,34H,6-7,14-20H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Competitive inhibition of human BChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition and measured... |
Eur J Med Chem 156: 598-617 (2018)
Article DOI: 10.1016/j.ejmech.2018.07.033
BindingDB Entry DOI: 10.7270/Q27W6FW2 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50515454
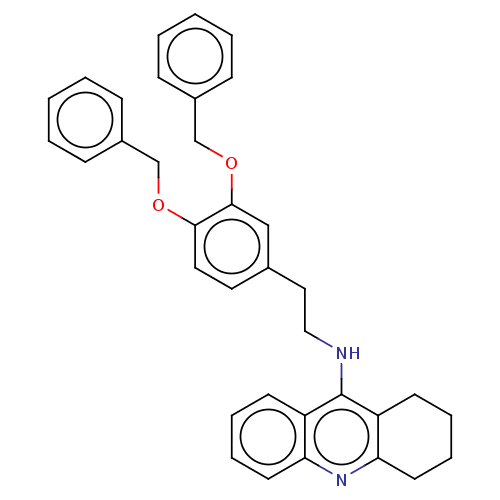
(CHEMBL4550977)Show SMILES C(Cc1ccc(OCc2ccccc2)c(OCc2ccccc2)c1)Nc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C35H34N2O2/c1-3-11-27(12-4-1)24-38-33-20-19-26(23-34(33)39-25-28-13-5-2-6-14-28)21-22-36-35-29-15-7-9-17-31(29)37-32-18-10-8-16-30(32)35/h1-7,9,11-15,17,19-20,23H,8,10,16,18,21-22,24-25H2,(H,36,37) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Sevilla
Curated by ChEMBL
| Assay Description
Inhibition of equine serum BuChE assessed as dissociation constant for enzyme-substrate-inhibitor complex using butyrylthiocholine iodide as substrat... |
Eur J Med Chem 181: (2019)
Article DOI: 10.1016/j.ejmech.2019.07.053
BindingDB Entry DOI: 10.7270/Q23R0X60 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50271470
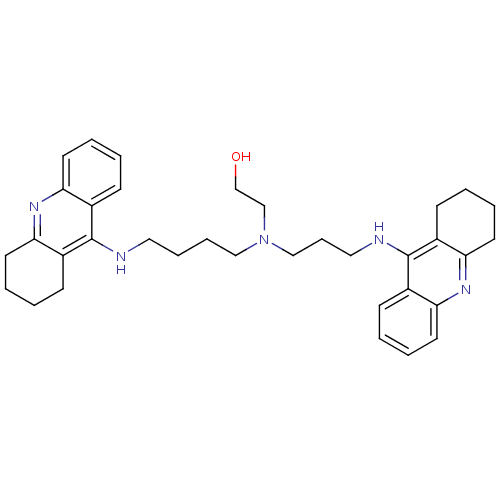
(CHEMBL499224 | N-(2-Hydroxyethyl)-N-(1,2,3,4-tetra...)Show SMILES OCCN(CCCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C35H45N5O/c41-25-24-40(23-11-21-37-35-28-14-3-7-18-32(28)39-33-19-8-4-15-29(33)35)22-10-9-20-36-34-26-12-1-5-16-30(26)38-31-17-6-2-13-27(31)34/h1,3,5,7,12,14,16,18,41H,2,4,6,8-11,13,15,17,19-25H2,(H,36,38)(H,37,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50265253
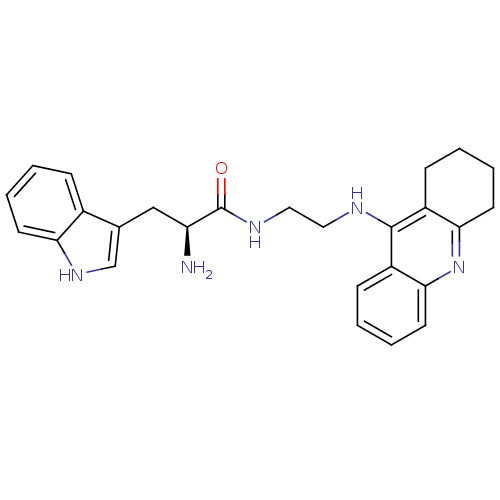
((S)-2-amino-3-(1H-indol-3-yl)-N-(2-(1,2,3,4-tetrah...)Show SMILES N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCCNc1c2CCCCc2nc2ccccc12 |r| Show InChI InChI=1S/C26H29N5O/c27-21(15-17-16-30-22-10-4-1-7-18(17)22)26(32)29-14-13-28-25-19-8-2-5-11-23(19)31-24-12-6-3-9-20(24)25/h1-2,4-5,7-8,10-11,16,21,30H,3,6,9,12-15,27H2,(H,28,31)(H,29,32)/t21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
European Research Centre for Drug Discovery and Development (NatSynDrugs)
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BChE |
Bioorg Med Chem Lett 18: 5213-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.08.076
BindingDB Entry DOI: 10.7270/Q29W0F9S |
More data for this
Ligand-Target Pair | |
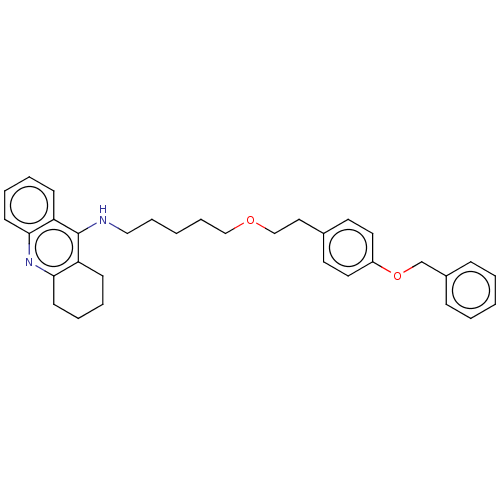
Cholinesterase
(Equus caballus (Horse)) | BDBM50515453
(CHEMBL4588525)Show SMILES C(CCNc1c2CCCCc2nc2ccccc12)CCOCCc1ccc(OCc2ccccc2)cc1 Show InChI InChI=1S/C33H38N2O2/c1-3-11-27(12-4-1)25-37-28-19-17-26(18-20-28)21-24-36-23-10-2-9-22-34-33-29-13-5-7-15-31(29)35-32-16-8-6-14-30(32)33/h1,3-5,7,11-13,15,17-20H,2,6,8-10,14,16,21-25H2,(H,34,35) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Sevilla
Curated by ChEMBL
| Assay Description
Inhibition of equine serum BuChE assessed as dissociation constant for enzyme-substrate-inhibitor complex using butyrylthiocholine iodide as substrat... |
Eur J Med Chem 181: (2019)
Article DOI: 10.1016/j.ejmech.2019.07.053
BindingDB Entry DOI: 10.7270/Q23R0X60 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM8971

(CHEMBL129108 | N-[8-(1,2,3,4-tetrahydroacridin-9-y...)Show SMILES C(CCCCSc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H41N3S/c1(3-13-23-35-33-25-15-5-9-19-29(25)36-30-20-10-6-16-26(30)33)2-4-14-24-38-34-27-17-7-11-21-31(27)37-32-22-12-8-18-28(32)34/h5,7,9,11,15,17,19,21H,1-4,6,8,10,12-14,16,18,20,22-24H2,(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1.65 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM8971

(CHEMBL129108 | N-[8-(1,2,3,4-tetrahydroacridin-9-y...)Show SMILES C(CCCCSc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H41N3S/c1(3-13-23-35-33-25-15-5-9-19-29(25)36-30-20-10-6-16-26(30)33)2-4-14-24-38-34-27-17-7-11-21-31(27)37-32-22-12-8-18-28(32)34/h5,7,9,11,15,17,19,21H,1-4,6,8,10,12-14,16,18,20,22-24H2,(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1.65 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
European Research Centre for Drug Discovery and Development (NatSynDrugs)
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BChE |
Bioorg Med Chem Lett 18: 5213-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.08.076
BindingDB Entry DOI: 10.7270/Q29W0F9S |
More data for this
Ligand-Target Pair | |
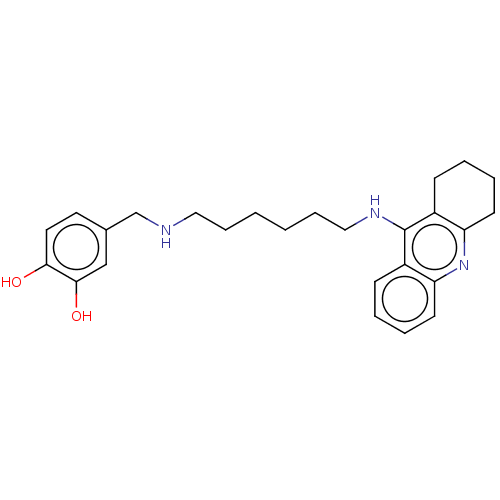
Cholinesterase
(Equus caballus (Horse)) | BDBM50515473
(CHEMBL4572757)Show InChI InChI=1S/C26H33N3O2/c30-24-14-13-19(17-25(24)31)18-27-15-7-1-2-8-16-28-26-20-9-3-5-11-22(20)29-23-12-6-4-10-21(23)26/h3,5,9,11,13-14,17,27,30-31H,1-2,4,6-8,10,12,15-16,18H2,(H,28,29) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Sevilla
Curated by ChEMBL
| Assay Description
Inhibition of equine serum BuChE assessed as dissociation constant for enzyme-substrate-inhibitor complex using butyrylthiocholine iodide as substrat... |
Eur J Med Chem 181: (2019)
Article DOI: 10.1016/j.ejmech.2019.07.053
BindingDB Entry DOI: 10.7270/Q23R0X60 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50468736
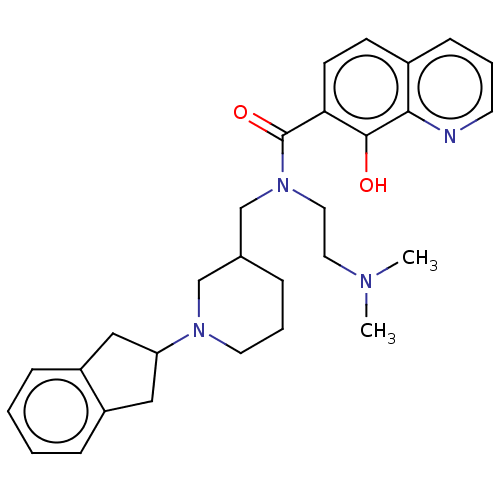
(CHEMBL4283585)Show SMILES CN(C)CCN(CC1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2cccnc2c1O Show InChI InChI=1S/C29H36N4O2/c1-31(2)15-16-33(29(35)26-12-11-22-10-5-13-30-27(22)28(26)34)20-21-7-6-14-32(19-21)25-17-23-8-3-4-9-24(23)18-25/h3-5,8-13,21,25,34H,6-7,14-20H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Competitive inhibition of human BChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition and measured... |
Eur J Med Chem 156: 598-617 (2018)
Article DOI: 10.1016/j.ejmech.2018.07.033
BindingDB Entry DOI: 10.7270/Q27W6FW2 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50433313
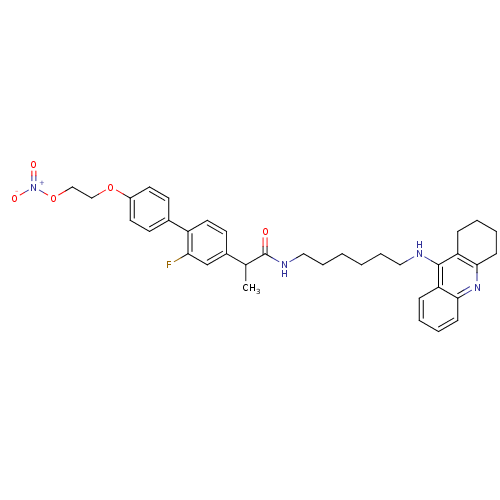
(CHEMBL2376474)Show SMILES CC(C(=O)NCCCCCCNc1c2CCCCc2nc2ccccc12)c1ccc(c(F)c1)-c1ccc(OCCO[N+]([O-])=O)cc1 Show InChI InChI=1S/C36H41FN4O5/c1-25(27-16-19-29(32(37)24-27)26-14-17-28(18-15-26)45-22-23-46-41(43)44)36(42)39-21-9-3-2-8-20-38-35-30-10-4-6-12-33(30)40-34-13-7-5-11-31(34)35/h4,6,10,12,14-19,24-25H,2-3,5,7-9,11,13,20-23H2,1H3,(H,38,40)(H,39,42) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Competitive inhibition of equine serum BChE by Lineweaver Burk reciprocal plot analysis in presence of acetylcholine |
Bioorg Med Chem 21: 2462-70 (2013)
Article DOI: 10.1016/j.bmc.2013.03.005
BindingDB Entry DOI: 10.7270/Q2319X82 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM8971

(CHEMBL129108 | N-[8-(1,2,3,4-tetrahydroacridin-9-y...)Show SMILES C(CCCCSc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H41N3S/c1(3-13-23-35-33-25-15-5-9-19-29(25)36-30-20-10-6-16-26(30)33)2-4-14-24-38-34-27-17-7-11-21-31(27)37-32-22-12-8-18-28(32)34/h5,7,9,11,15,17,19,21H,1-4,6,8,10,12-14,16,18,20,22-24H2,(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human butyrylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50265256
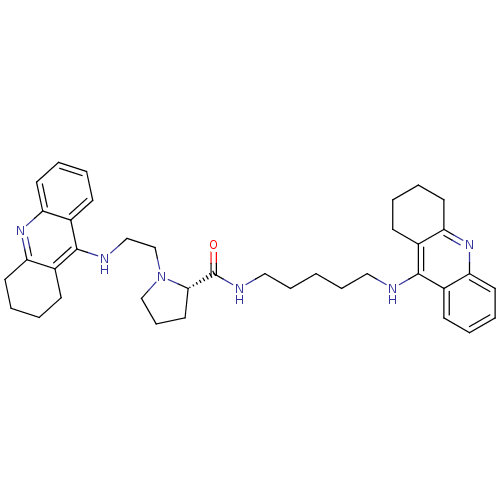
((S)-1-(2-(1,2,3,4-tetrahydroacridin-9-ylamino)ethy...)Show SMILES O=C(NCCCCCNc1c2CCCCc2nc2ccccc12)[C@@H]1CCCN1CCNc1c2CCCCc2nc2ccccc12 |r| Show InChI InChI=1S/C38H48N6O/c45-38(41-23-11-1-10-22-39-36-27-13-2-6-17-31(27)42-32-18-7-3-14-28(32)36)35-21-12-25-44(35)26-24-40-37-29-15-4-8-19-33(29)43-34-20-9-5-16-30(34)37/h2,4,6,8,13,15,17,19,35H,1,3,5,7,9-12,14,16,18,20-26H2,(H,39,42)(H,40,43)(H,41,45)/t35-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.77 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
European Research Centre for Drug Discovery and Development (NatSynDrugs)
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BChE |
Bioorg Med Chem Lett 18: 5213-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.08.076
BindingDB Entry DOI: 10.7270/Q29W0F9S |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50265252
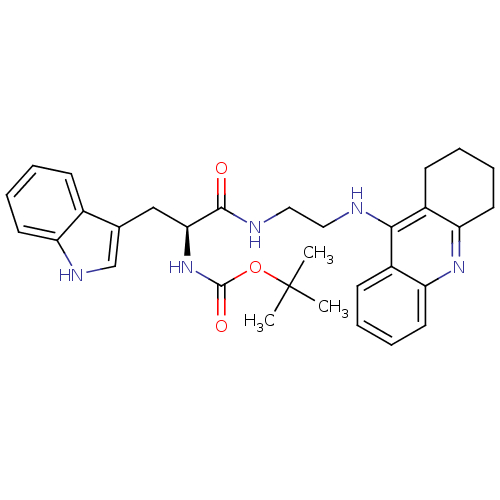
((S)-tert-butyl 3-(1H-indol-3-yl)-1-oxo-1-(2-(1,2,3...)Show SMILES CC(C)(C)OC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)NCCNc1c2CCCCc2nc2ccccc12 |r| Show InChI InChI=1S/C31H37N5O3/c1-31(2,3)39-30(38)36-27(18-20-19-34-24-13-7-4-10-21(20)24)29(37)33-17-16-32-28-22-11-5-8-14-25(22)35-26-15-9-6-12-23(26)28/h4-5,7-8,10-11,13-14,19,27,34H,6,9,12,15-18H2,1-3H3,(H,32,35)(H,33,37)(H,36,38)/t27-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.87 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
European Research Centre for Drug Discovery and Development (NatSynDrugs)
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BChE |
Bioorg Med Chem Lett 18: 5213-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.08.076
BindingDB Entry DOI: 10.7270/Q29W0F9S |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50271473
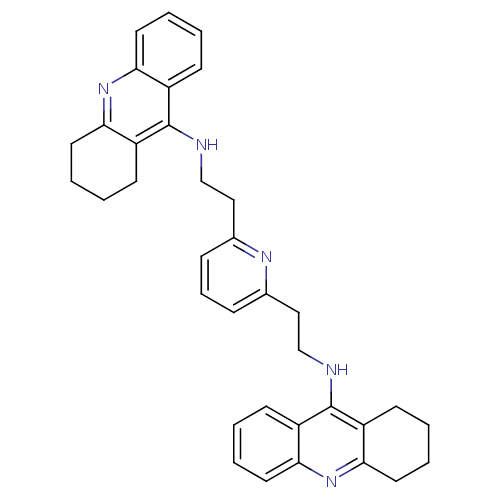
(2,6-bis{[(1,2,3,4-Tetrahydroacridin-9-yl)amino]eth...)Show SMILES C(Cc1cccc(CCNc2c3CCCCc3nc3ccccc23)n1)Nc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C35H37N5/c1-5-16-30-26(12-1)34(27-13-2-6-17-31(27)39-30)36-22-20-24-10-9-11-25(38-24)21-23-37-35-28-14-3-7-18-32(28)40-33-19-8-4-15-29(33)35/h1,3,5,7,9-12,14,16,18H,2,4,6,8,13,15,17,19-23H2,(H,36,39)(H,37,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.87 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50515465
(CHEMBL4536715)Show InChI InChI=1S/C20H18N2O2/c23-18-10-9-13(11-19(18)24)12-21-20-14-5-1-3-7-16(14)22-17-8-4-2-6-15(17)20/h1,3,5,7,9-12,23-24H,2,4,6,8H2/b21-12+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Sevilla
Curated by ChEMBL
| Assay Description
Inhibition of equine serum BuChE assessed as dissociation constant for enzyme-substrate-inhibitor complex using butyrylthiocholine iodide as substrat... |
Eur J Med Chem 181: (2019)
Article DOI: 10.1016/j.ejmech.2019.07.053
BindingDB Entry DOI: 10.7270/Q23R0X60 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50515453

(CHEMBL4588525)Show SMILES C(CCNc1c2CCCCc2nc2ccccc12)CCOCCc1ccc(OCc2ccccc2)cc1 Show InChI InChI=1S/C33H38N2O2/c1-3-11-27(12-4-1)25-37-28-19-17-26(18-20-28)21-24-36-23-10-2-9-22-34-33-29-13-5-7-15-31(29)35-32-16-8-6-14-30(32)33/h1,3-5,7,11-13,15,17-20H,2,6,8-10,14,16,21-25H2,(H,34,35) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Sevilla
Curated by ChEMBL
| Assay Description
Inhibition of equine serum BuChE assessed as dissociation constant for enzyme-inhibitor complex using butyrylthiocholine iodide as substrate measured... |
Eur J Med Chem 181: (2019)
Article DOI: 10.1016/j.ejmech.2019.07.053
BindingDB Entry DOI: 10.7270/Q23R0X60 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50515470
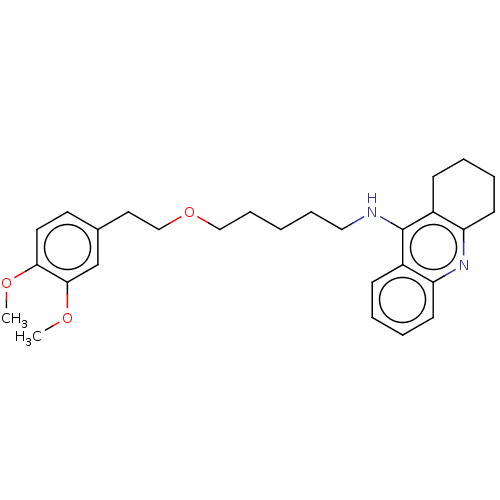
(CHEMBL4555120)Show SMILES COc1ccc(CCOCCCCCNc2c3CCCCc3nc3ccccc23)cc1OC Show InChI InChI=1S/C28H36N2O3/c1-31-26-15-14-21(20-27(26)32-2)16-19-33-18-9-3-8-17-29-28-22-10-4-6-12-24(22)30-25-13-7-5-11-23(25)28/h4,6,10,12,14-15,20H,3,5,7-9,11,13,16-19H2,1-2H3,(H,29,30) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Sevilla
Curated by ChEMBL
| Assay Description
Inhibition of equine serum BuChE assessed as dissociation constant for enzyme-substrate-inhibitor complex using butyrylthiocholine iodide as substrat... |
Eur J Med Chem 181: (2019)
Article DOI: 10.1016/j.ejmech.2019.07.053
BindingDB Entry DOI: 10.7270/Q23R0X60 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM8963

(CHEMBL32823 | Homodimeric Tacrine Analog 3b | N-[7...)Show SMILES C(CCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C33H40N4/c1(2-12-22-34-32-24-14-4-8-18-28(24)36-29-19-9-5-15-25(29)32)3-13-23-35-33-26-16-6-10-20-30(26)37-31-21-11-7-17-27(31)33/h4,6,8,10,14,16,18,20H,1-3,5,7,9,11-13,15,17,19,21-23H2,(H,34,36)(H,35,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of Equine Butyrylcholinesterase |
J Med Chem 46: 1-4 (2002)
Article DOI: 10.1021/jm0255668
BindingDB Entry DOI: 10.7270/Q2GF0V7X |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM8963

(CHEMBL32823 | Homodimeric Tacrine Analog 3b | N-[7...)Show SMILES C(CCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C33H40N4/c1(2-12-22-34-32-24-14-4-8-18-28(24)36-29-19-9-5-15-25(29)32)3-13-23-35-33-26-16-6-10-20-30(26)37-31-21-11-7-17-27(31)33/h4,6,8,10,14,16,18,20H,1-3,5,7,9,11-13,15,17,19,21-23H2,(H,34,36)(H,35,37) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of horse BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM8969
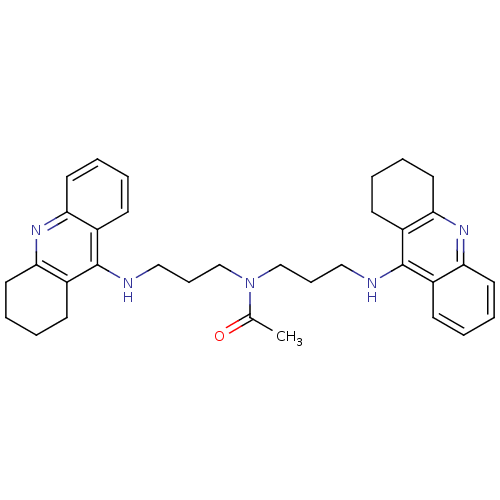
(CHEMBL131410 | N,N-bis[3-(1,2,3,4-tetrahydroacridi...)Show SMILES CC(=O)N(CCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H41N5O/c1-24(40)39(22-10-20-35-33-25-12-2-6-16-29(25)37-30-17-7-3-13-26(30)33)23-11-21-36-34-27-14-4-8-18-31(27)38-32-19-9-5-15-28(32)34/h2,4,6,8,12,14,16,18H,3,5,7,9-11,13,15,17,19-23H2,1H3,(H,35,37)(H,36,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of Equine Butyrylcholinesterase |
J Med Chem 46: 1-4 (2002)
Article DOI: 10.1021/jm0255668
BindingDB Entry DOI: 10.7270/Q2GF0V7X |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50027375

(CHEMBL3338394)Show SMILES COCCN(C[C@@H]1CCCN(C1)C1Cc2ccccc2C1)C(=O)c1ccc2ccccc2c1 |r| Show InChI InChI=1S/C29H34N2O2/c1-33-16-15-31(29(32)27-13-12-23-8-2-3-9-24(23)17-27)21-22-7-6-14-30(20-22)28-18-25-10-4-5-11-26(25)19-28/h2-5,8-13,17,22,28H,6-7,14-16,18-21H2,1H3/t22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Ljubljana
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human BChE at 50 nM by stopped flow apparatus method |
J Med Chem 57: 8167-79 (2014)
Article DOI: 10.1021/jm501195e
BindingDB Entry DOI: 10.7270/Q22V2HQ7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cholinesterase
(Equus caballus (Horse)) | BDBM50515459
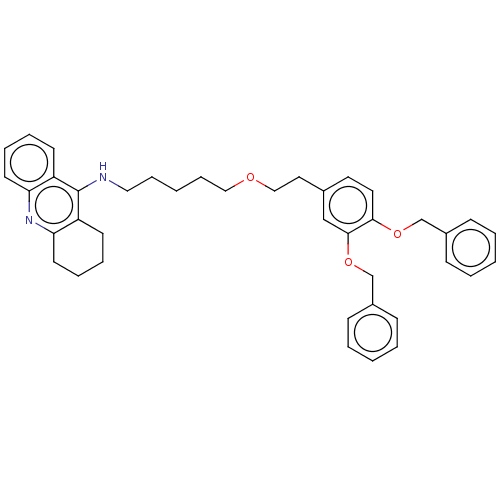
(CHEMBL4545701)Show SMILES C(CCNc1c2CCCCc2nc2ccccc12)CCOCCc1ccc(OCc2ccccc2)c(OCc2ccccc2)c1 Show InChI InChI=1S/C40H44N2O3/c1-4-14-32(15-5-1)29-44-38-23-22-31(28-39(38)45-30-33-16-6-2-7-17-33)24-27-43-26-13-3-12-25-41-40-34-18-8-10-20-36(34)42-37-21-11-9-19-35(37)40/h1-2,4-8,10,14-18,20,22-23,28H,3,9,11-13,19,21,24-27,29-30H2,(H,41,42) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Sevilla
Curated by ChEMBL
| Assay Description
Inhibition of equine serum BuChE assessed as dissociation constant for enzyme-substrate-inhibitor complex using butyrylthiocholine iodide as substrat... |
Eur J Med Chem 181: (2019)
Article DOI: 10.1016/j.ejmech.2019.07.053
BindingDB Entry DOI: 10.7270/Q23R0X60 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50515460
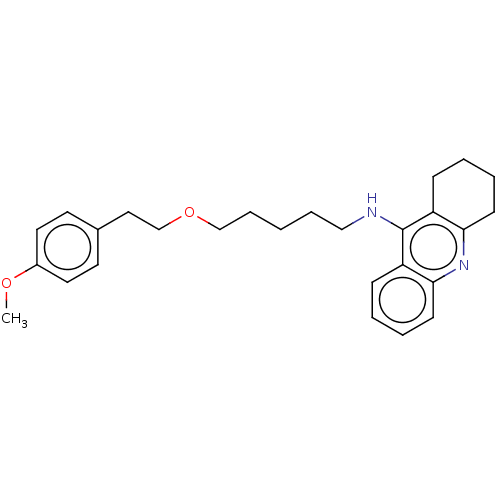
(CHEMBL4475228)Show InChI InChI=1S/C27H34N2O2/c1-30-22-15-13-21(14-16-22)17-20-31-19-8-2-7-18-28-27-23-9-3-5-11-25(23)29-26-12-6-4-10-24(26)27/h3,5,9,11,13-16H,2,4,6-8,10,12,17-20H2,1H3,(H,28,29) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Sevilla
Curated by ChEMBL
| Assay Description
Inhibition of equine serum BuChE assessed as dissociation constant for enzyme-substrate-inhibitor complex using butyrylthiocholine iodide as substrat... |
Eur J Med Chem 181: (2019)
Article DOI: 10.1016/j.ejmech.2019.07.053
BindingDB Entry DOI: 10.7270/Q23R0X60 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50604404
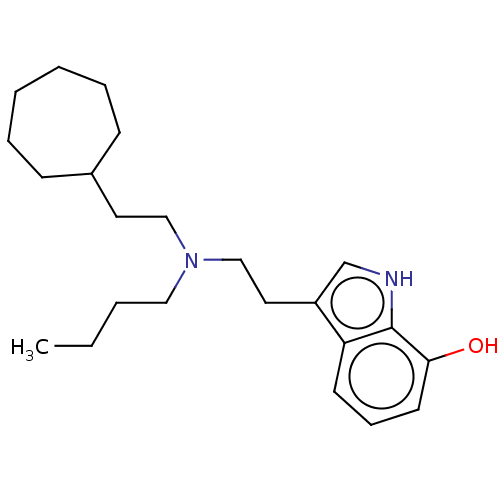
(CHEMBL5186857) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114248
BindingDB Entry DOI: 10.7270/Q2125XRJ |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM8975
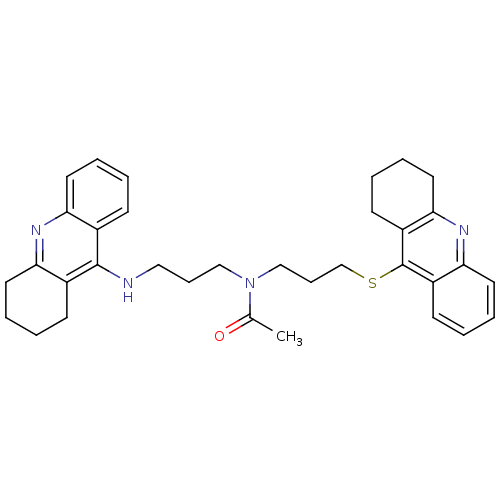
(CHEMBL179192 | N-[3-(1,2,3,4-Tetrahydroacridin-9-y...)Show SMILES CC(=O)N(CCCNc1c2CCCCc2nc2ccccc12)CCCSc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H40N4OS/c1-24(39)38(21-10-20-35-33-25-12-2-6-16-29(25)36-30-17-7-3-13-26(30)33)22-11-23-40-34-27-14-4-8-18-31(27)37-32-19-9-5-15-28(32)34/h2,4,6,8,12,14,16,18H,3,5,7,9-11,13,15,17,19-23H2,1H3,(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50515468
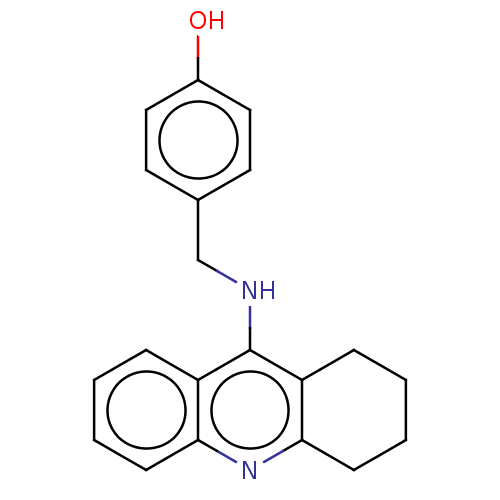
(CHEMBL4483710)Show InChI InChI=1S/C20H20N2O/c23-15-11-9-14(10-12-15)13-21-20-16-5-1-3-7-18(16)22-19-8-4-2-6-17(19)20/h1,3,5,7,9-12,23H,2,4,6,8,13H2,(H,21,22) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 3.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Sevilla
Curated by ChEMBL
| Assay Description
Inhibition of equine serum BuChE assessed as dissociation constant for enzyme-substrate-inhibitor complex using butyrylthiocholine iodide as substrat... |
Eur J Med Chem 181: (2019)
Article DOI: 10.1016/j.ejmech.2019.07.053
BindingDB Entry DOI: 10.7270/Q23R0X60 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Equus caballus (Horse)) | BDBM50515461
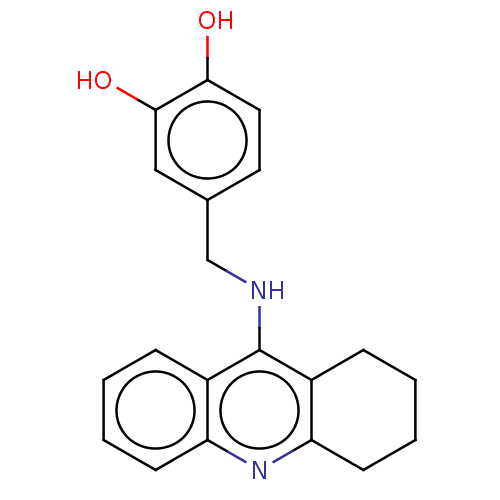
(CHEMBL4535585)Show InChI InChI=1S/C20H20N2O2/c23-18-10-9-13(11-19(18)24)12-21-20-14-5-1-3-7-16(14)22-17-8-4-2-6-15(17)20/h1,3,5,7,9-11,23-24H,2,4,6,8,12H2,(H,21,22) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 3.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad de Sevilla
Curated by ChEMBL
| Assay Description
Inhibition of equine serum BuChE assessed as dissociation constant for enzyme-substrate-inhibitor complex using butyrylthiocholine iodide as substrat... |
Eur J Med Chem 181: (2019)
Article DOI: 10.1016/j.ejmech.2019.07.053
BindingDB Entry DOI: 10.7270/Q23R0X60 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50271471
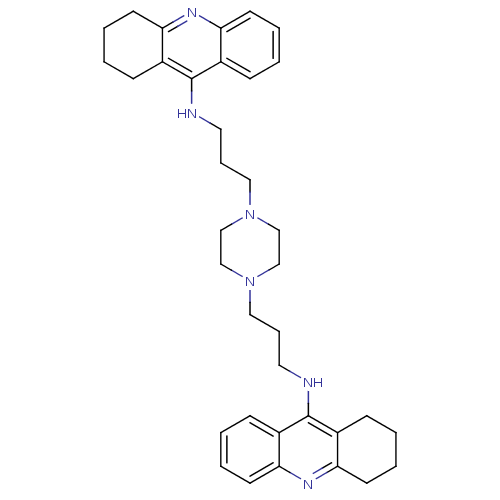
(1,4-bis[3-(1,2,3,4-Tetrahydroacridin-9-yl)aminopro...)Show SMILES C(CNc1c2CCCCc2nc2ccccc12)CN1CCN(CCCNc2c3CCCCc3nc3ccccc23)CC1 Show InChI InChI=1S/C36H46N6/c1-5-15-31-27(11-1)35(28-12-2-6-16-32(28)39-31)37-19-9-21-41-23-25-42(26-24-41)22-10-20-38-36-29-13-3-7-17-33(29)40-34-18-8-4-14-30(34)36/h1,3,5,7,11,13,15,17H,2,4,6,8-10,12,14,16,18-26H2,(H,37,39)(H,38,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Siena
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BuChE |
J Med Chem 51: 3154-70 (2008)
Article DOI: 10.1021/jm701253t
BindingDB Entry DOI: 10.7270/Q22Z16D4 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data