

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9022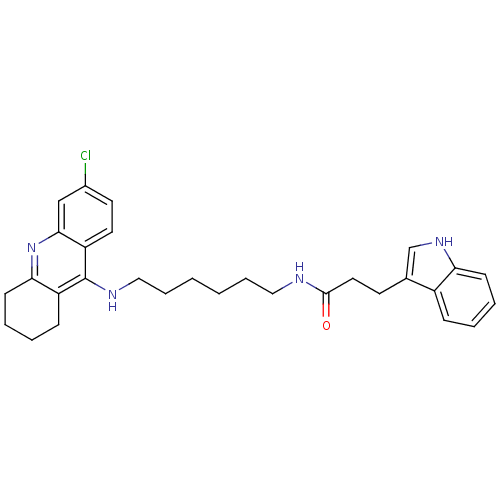 (CHEMBL225567 | Indole-Tacrine Heterodimer 5 | N-[5...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0200 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9023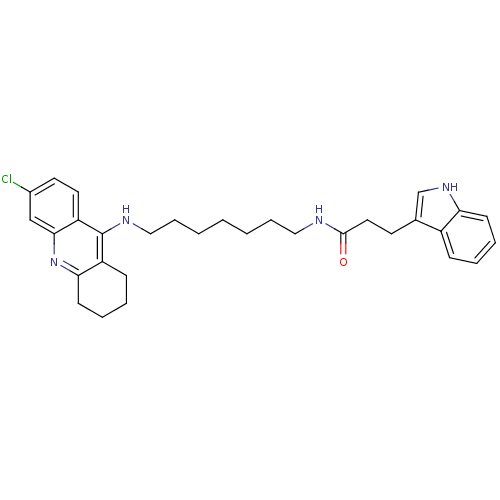 (CHEMBL225198 | Indole-Tacrine Heterodimer 6 | N-[7...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0600 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271142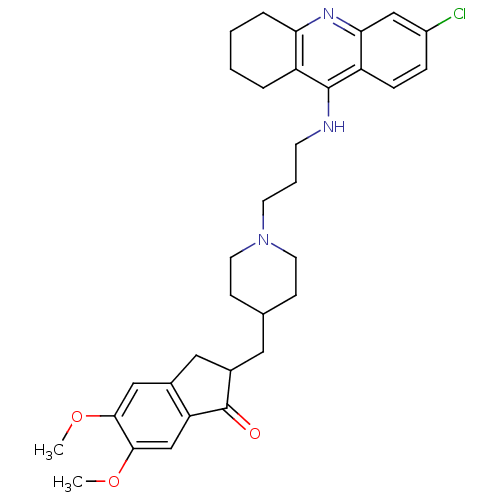 (6-Chloro-9-[(3-{4-[(5,6-Dimethoxy-1-oxoindan-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50262988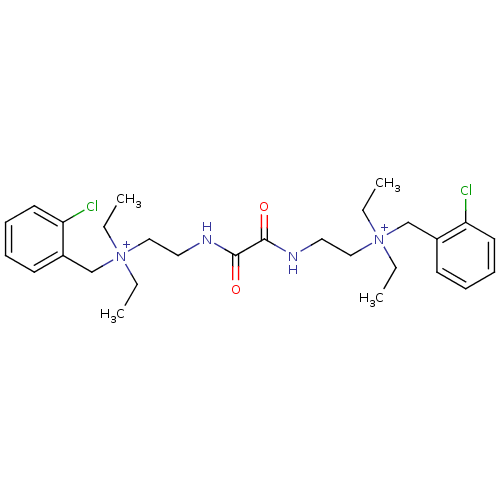 (CHEMBL1200541 | N-(2-chlorobenzyl)-2-(2-(2-((2-chl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
Università di Bari Curated by ChEMBL | Assay Description Inhibition of bovine AChE by Ellman's method | Bioorg Med Chem 16: 7450-6 (2008) Article DOI: 10.1016/j.bmc.2008.06.022 BindingDB Entry DOI: 10.7270/Q2NC611F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50262879 (3-hydroxy-5-(4-(3-hydroxy-5-(trimethylammonio)phen...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a |
Università di Bari Curated by ChEMBL | Assay Description Inhibition of bovine AChE by Ellman's method | Bioorg Med Chem 16: 7450-6 (2008) Article DOI: 10.1016/j.bmc.2008.06.022 BindingDB Entry DOI: 10.7270/Q2NC611F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9009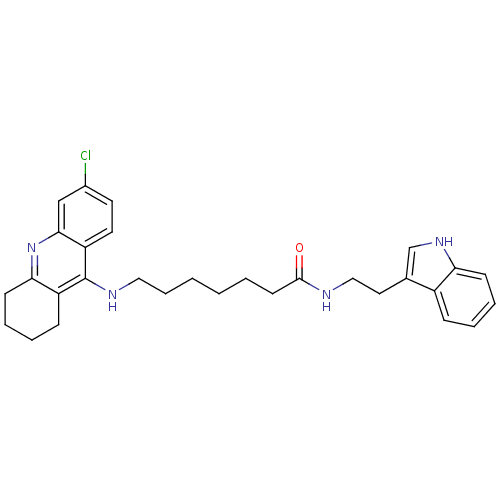 (7-(6-Chloro-1,2,3,4-tetrahydro-acridin-9-ylamino)-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Tehran University of Medical Sciences Curated by ChEMBL | Assay Description Inhibitory activity was determined against bovine liver beta galactosidase | Eur J Med Chem 128: 332-345 (2017) Article DOI: 10.1016/j.ejmech.2016.10.060 BindingDB Entry DOI: 10.7270/Q2HM5BQX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9035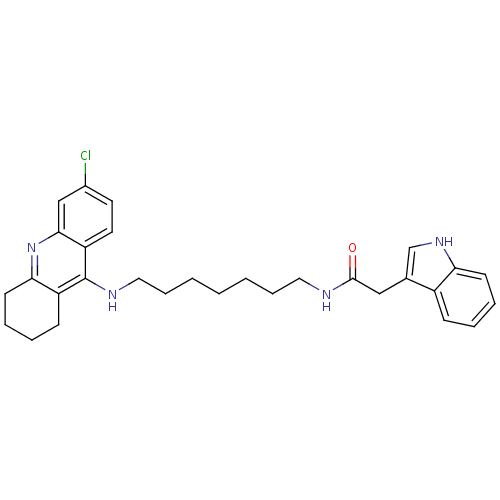 (Indole-Tacrine Heterodimer 18 | N-[7-(6-Chloro-1,2...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9009 (7-(6-Chloro-1,2,3,4-tetrahydro-acridin-9-ylamino)-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Instituto de Quimica Medica (CSIC) | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 412 nm were recorded for 5 min with ... | J Med Chem 49: 459-62 (2006) Article DOI: 10.1021/jm050746d BindingDB Entry DOI: 10.7270/Q2VD6WN2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50369748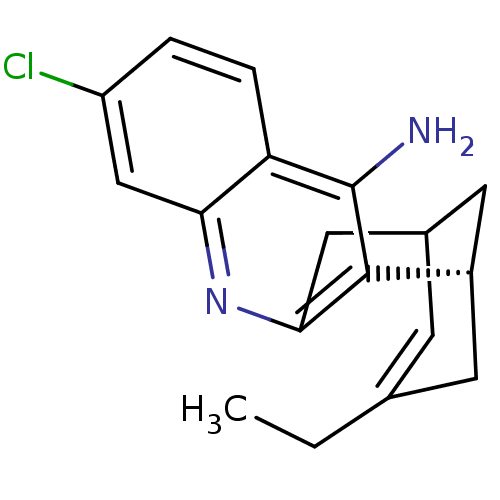 (CHEMBL208599) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase activity from bovine erythrocytes after 30 minutes of incubation | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50369748 (CHEMBL208599) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase activity from bovine erythrocytes after 30 minutes of incubation | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271140 (9-[(3-{4-[(5,6-Dimethoxy-1-oxoindan-2-yl)methyl]pi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9037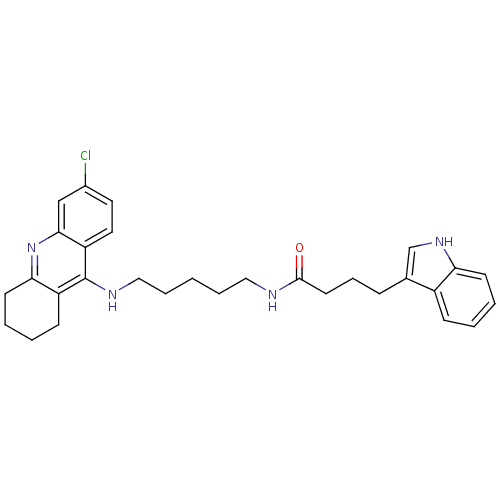 (Indole-Tacrine Heterodimer 20 | N-[5-(6-Chloro-1,2...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50262878 (3-hydroxy-5-(3-(3-hydroxy-5-(trimethylammonio)phen...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
Università di Bari Curated by ChEMBL | Assay Description Inhibition of bovine AChE by Ellman's method | Bioorg Med Chem 16: 7450-6 (2008) Article DOI: 10.1016/j.bmc.2008.06.022 BindingDB Entry DOI: 10.7270/Q2NC611F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9038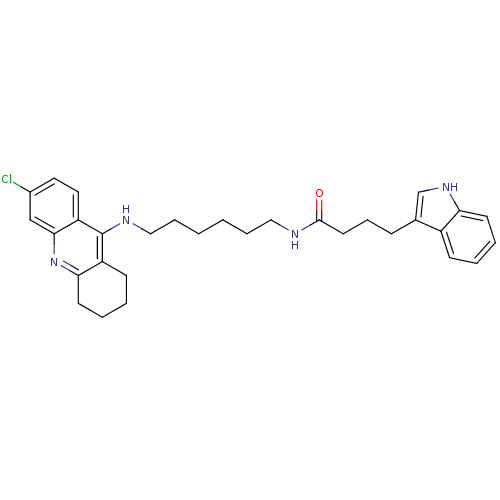 (Indole-Tacrine Heterodimer 21 | N-[6-(6-Chloro-1,2...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9024 (Indole-Tacrine Heterodimer 7 | N-[8-(6-Chloro-1,2,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50094626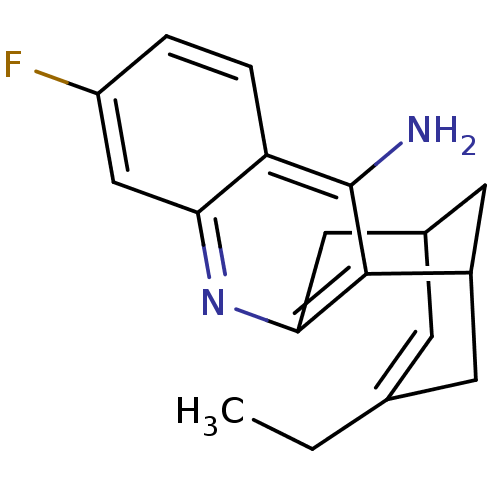 ((+)-15-ethyl-7-fluoro-10-azatetracyclo[11.3.1.02,1...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase activity from bovine erythrocytes after 30 minutes of incubation | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271141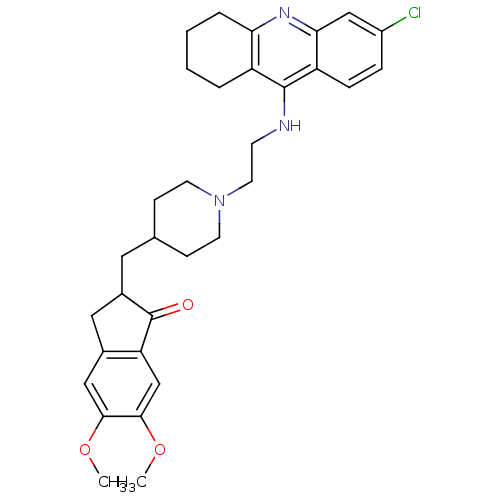 (6-Chloro-9-[(2-{4-[(5,6-dimethoxy-1-oxoindan-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.570 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9036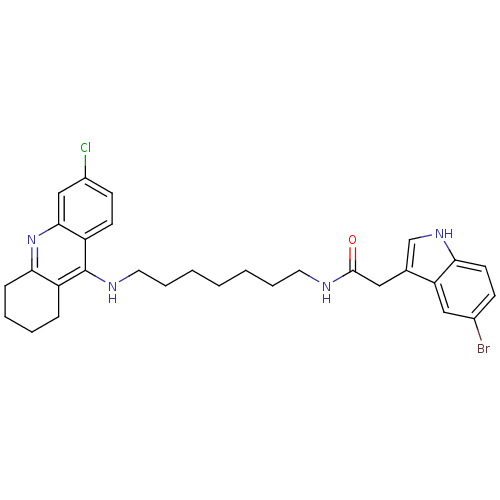 (2-(5-Bromo-1H-indol-3-yl)-N-[7-(6-chloro-1,2,3,4-t...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM10592 (7-chloro-15-methyl-10-azatetracyclo[11.3.1.0^{2,11...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.640 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase activity from bovine erythrocytes after 30 minutes of incubation | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM10592 (7-chloro-15-methyl-10-azatetracyclo[11.3.1.0^{2,11...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.640 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase activity from bovine erythrocytes after 30 minutes of incubation | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9029 (Indole-Tacrine Heterodimer 12 | N-[6-(6-Chloro-1,2...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9042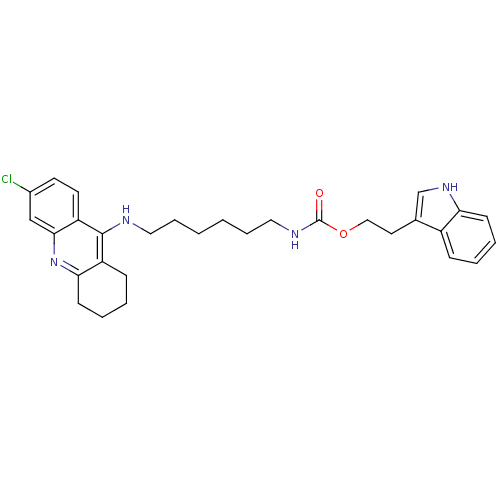 (2-(1H-indol-3-yl)ethyl N-{6-[(6-chloro-1,2,3,4-tet...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271192 (6-Chloro-9-[(3-{4-[(5,6-dimethoxyindan-2-yl)methyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.820 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271190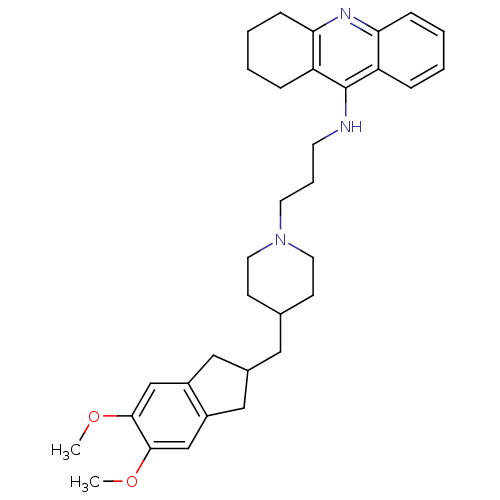 (9-[(3-{4-[(5,6-dimethoxyindan-2-yl)methyl]piperidi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.820 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9007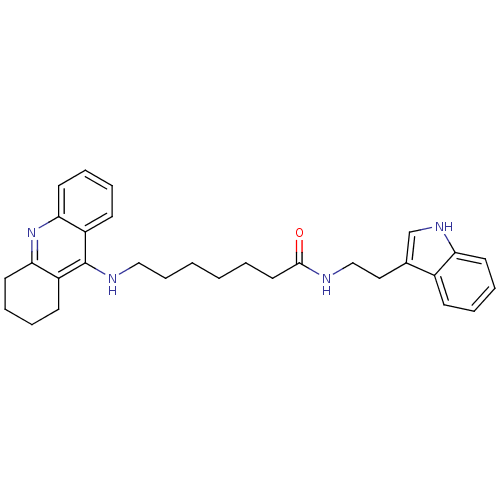 (7-(1,2,3,4-Tetrahydro-acridin-9-ylamino)-heptanoic...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Instituto de Quimica Medica (CSIC) | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 412 nm were recorded for 5 min with ... | J Med Chem 49: 459-62 (2006) Article DOI: 10.1021/jm050746d BindingDB Entry DOI: 10.7270/Q2VD6WN2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50262926 (3-(4-(3,4-dimethyl-2-oxo-2H-chromen-7-yloxy)butoxy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Università di Bari Curated by ChEMBL | Assay Description Inhibition of bovine AChE by Ellman's method | Bioorg Med Chem 16: 7450-6 (2008) Article DOI: 10.1016/j.bmc.2008.06.022 BindingDB Entry DOI: 10.7270/Q2NC611F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9006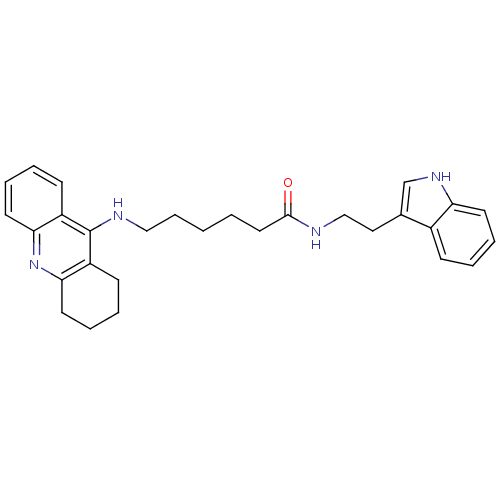 (6-(1,2,3,4-Tetrahydro-acridin-9-ylamino)-hexanoic ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Tehran University of Medical Sciences Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AchE using acetylthiocholine as substrate by Ellman's method | Eur J Med Chem 128: 332-345 (2017) Article DOI: 10.1016/j.ejmech.2016.10.060 BindingDB Entry DOI: 10.7270/Q2HM5BQX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM10592 (7-chloro-15-methyl-10-azatetracyclo[11.3.1.0^{2,11...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description In vitro inhibitory activity against bovine acetylcholinesterase | J Med Chem 47: 4471-82 (2004) Article DOI: 10.1021/jm049877p BindingDB Entry DOI: 10.7270/Q26974WJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50200334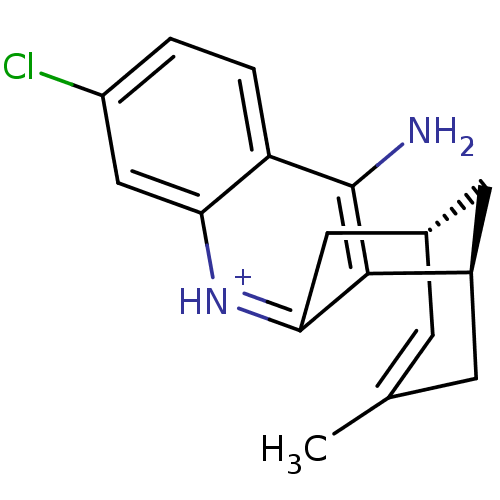 ((-)-(7S,11S)-huprine H3) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.15 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE | J Med Chem 49: 6833-40 (2006) Article DOI: 10.1021/jm060945c BindingDB Entry DOI: 10.7270/Q29K4C1S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM10592 (7-chloro-15-methyl-10-azatetracyclo[11.3.1.0^{2,11...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.15 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase of bovine erythrocytes | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50462398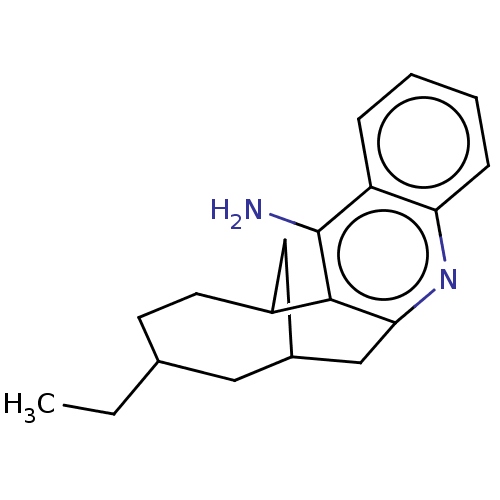 (CHEMBL4249848) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Punjabi University Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AchE using acetylthiocholine iodide as substrate after 15 mins by spectrophotometric analysis | Eur J Med Chem 151: 62-97 (2018) Article DOI: 10.1016/j.ejmech.2018.03.057 BindingDB Entry DOI: 10.7270/Q2NC63V1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50462402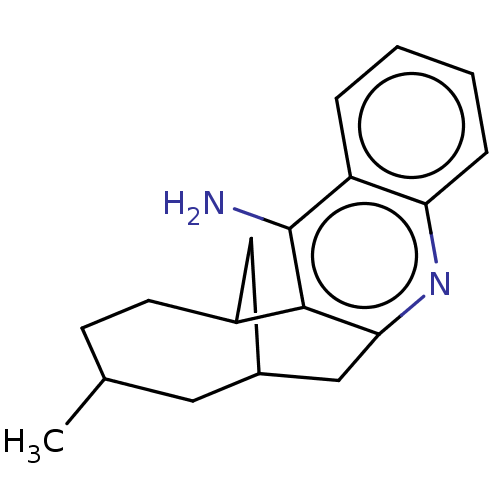 (CHEMBL4249379) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Punjabi University Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AchE using acetylthiocholine iodide as substrate after 15 mins by spectrophotometric analysis | Eur J Med Chem 151: 62-97 (2018) Article DOI: 10.1016/j.ejmech.2018.03.057 BindingDB Entry DOI: 10.7270/Q2NC63V1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM10597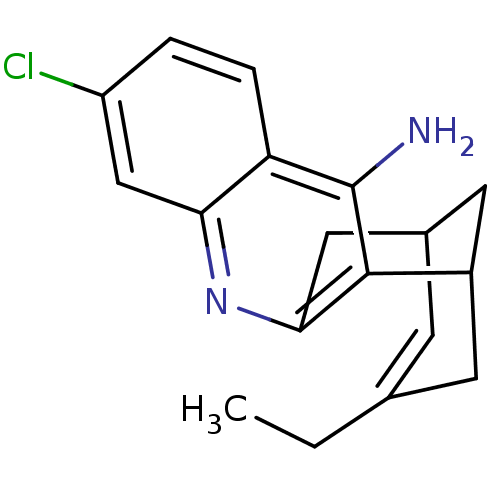 ((1S)-7-chloro-15-ethyl-10-azatetracyclo[11.3.1.0^{...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description In vitro inhibitory activity against bovine acetylcholinesterase | J Med Chem 47: 4471-82 (2004) Article DOI: 10.1021/jm049877p BindingDB Entry DOI: 10.7270/Q26974WJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50200343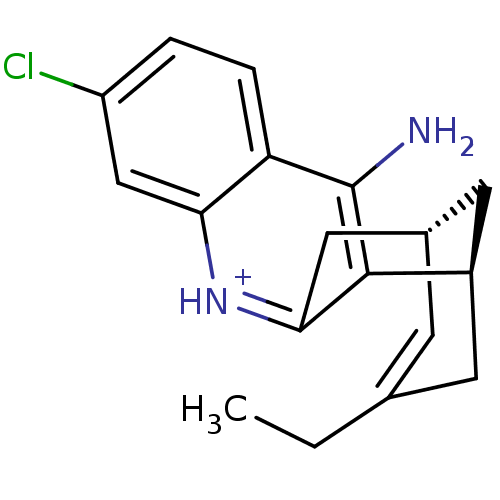 ((-)-(7S,11S)-huprine H7) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE | J Med Chem 49: 6833-40 (2006) Article DOI: 10.1021/jm060945c BindingDB Entry DOI: 10.7270/Q29K4C1S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9041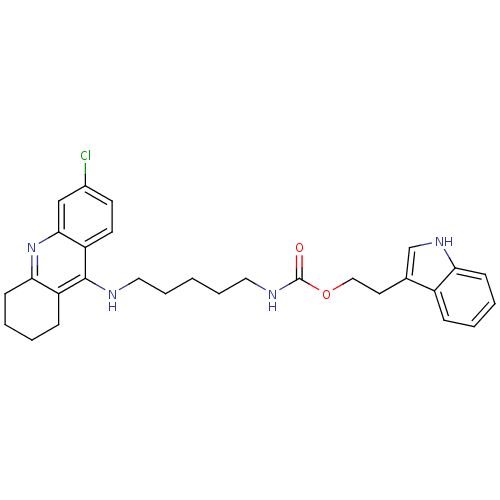 (2-(1H-indol-3-yl)ethyl N-{5-[(6-chloro-1,2,3,4-tet...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 5 min with ... | J Med Chem 48: 7223-33 (2005) Article DOI: 10.1021/jm0503289 BindingDB Entry DOI: 10.7270/Q2QN64Z5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50377921 (SODIUM NITROPRUSSIDE) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank MCE KEGG PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.74 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271191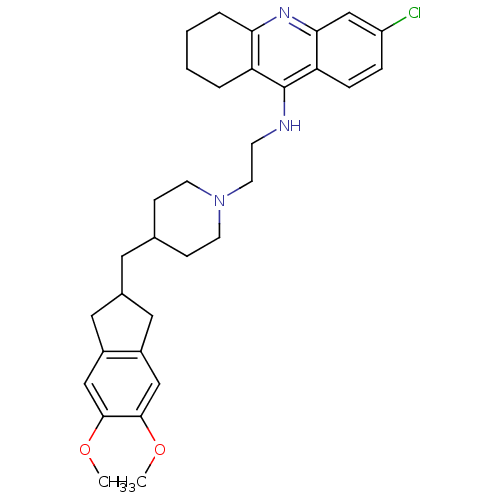 (6-Chloro-9-[(2-{4-[(5,6-dimethoxyindan-2-yl)methyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.86 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50094626 ((+)-15-ethyl-7-fluoro-10-azatetracyclo[11.3.1.02,1...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 1.91 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase activity from bovine erythrocytes after 0 minutes of incubation | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9008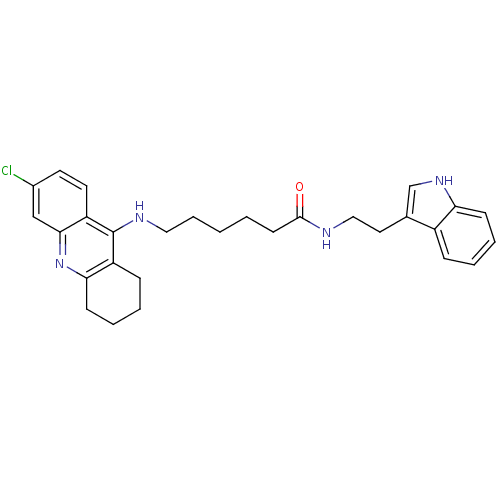 (6-(6-Chloro-1,2,3,4-tetrahydro-acridin-9-ylamino)-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Instituto de Quimica Medica (CSIC) | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 412 nm were recorded for 5 min with ... | J Med Chem 49: 459-62 (2006) Article DOI: 10.1021/jm050746d BindingDB Entry DOI: 10.7270/Q2VD6WN2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9012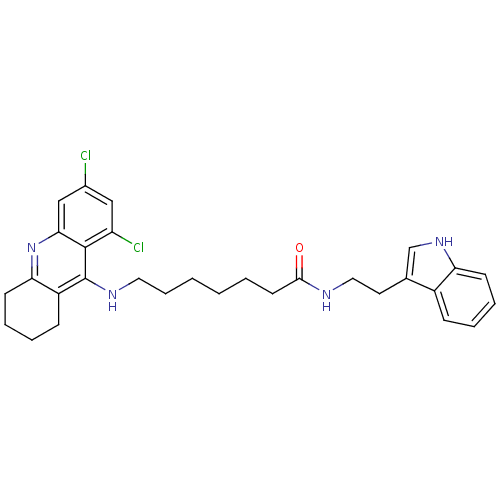 (7-(6,8-Dichloro-1,2,3,4-tetrahydro-acridin-9-ylami...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Instituto de Quimica Medica (CSIC) | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 412 nm were recorded for 5 min with ... | J Med Chem 49: 459-62 (2006) Article DOI: 10.1021/jm050746d BindingDB Entry DOI: 10.7270/Q2VD6WN2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50262880 (3-(3-(3,4-dimethyl-2-oxo-2H-chromen-7-yloxy)propox...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Università di Bari Curated by ChEMBL | Assay Description Inhibition of bovine AChE by Ellman's method | Bioorg Med Chem 16: 7450-6 (2008) Article DOI: 10.1016/j.bmc.2008.06.022 BindingDB Entry DOI: 10.7270/Q2NC611F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50271189 (9-[(2-{4-[(5,6-Dimethoxyindan-2-yl)methyl]piperidi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.28 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratori de Qu�mica Farmac�utica (Unitat Associada al CSIC) Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE by Ellman's assay | J Med Chem 51: 3588-98 (2008) Article DOI: 10.1021/jm8001313 BindingDB Entry DOI: 10.7270/Q2F76DGK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9014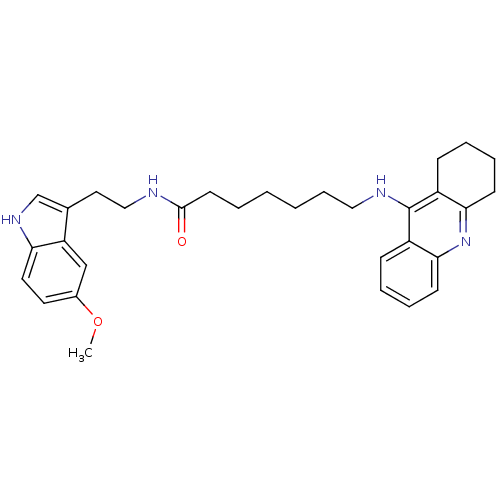 (7-(1,2,3,4-Tetrahydro-acridin-9-ylamino)-heptanoic...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Instituto de Quimica Medica (CSIC) | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 412 nm were recorded for 5 min with ... | J Med Chem 49: 459-62 (2006) Article DOI: 10.1021/jm050746d BindingDB Entry DOI: 10.7270/Q2VD6WN2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9073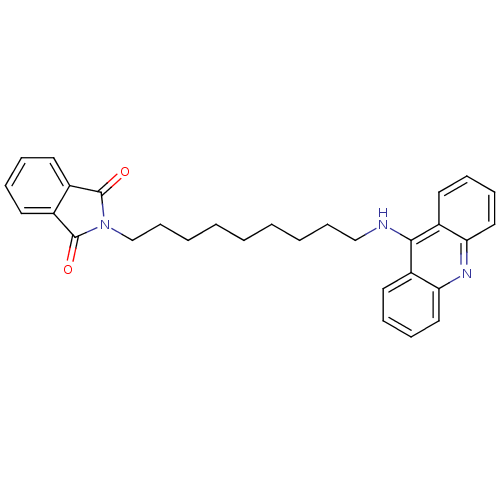 (2-[9-(Acridin-9-ylamino)-nonyl]-isoindole-1,3-dion...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 10 min with... | Bioorg Med Chem 13: 6588-97 (2005) Article DOI: 10.1016/j.bmc.2005.09.029 BindingDB Entry DOI: 10.7270/Q2G73BW9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50094633 (5,7-difluoro-15-methyl-10-azatetracyclo[11.3.1.02,...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.43 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase of bovine erythrocytes | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50094631 (15-ethyl-5,7-difluoro-10-azatetracyclo[11.3.1.02,1...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.62 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase of bovine erythrocytes | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50369748 (CHEMBL208599) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.77 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase of bovine erythrocytes | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM10597 ((1S)-7-chloro-15-ethyl-10-azatetracyclo[11.3.1.0^{...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 2.77 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Inhibition of bovine erythrocyte AChE | J Med Chem 49: 6833-40 (2006) Article DOI: 10.1021/jm060945c BindingDB Entry DOI: 10.7270/Q29K4C1S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM50369748 (CHEMBL208599) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2.77 | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of acetylcholinesterase of bovine erythrocytes | J Med Chem 43: 4657-66 (2001) BindingDB Entry DOI: 10.7270/Q2Q52Q9B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Bos taurus (bovine)) | BDBM9067 (Donepezil-tacrine hybrid 12 | N-[4-({2-[(6-chloro-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.80 | n/a | n/a | n/a | n/a | 8.0 | 30 |
Neuropharma | Assay Description The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 405 nm were recorded for 10 min with... | Bioorg Med Chem 13: 6588-97 (2005) Article DOI: 10.1016/j.bmc.2005.09.029 BindingDB Entry DOI: 10.7270/Q2G73BW9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 585 total ) | Next | Last >> |