 Found 40 hits Enz. Inhib. hit(s) with all data for entry = 50043561
Found 40 hits Enz. Inhib. hit(s) with all data for entry = 50043561 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442991

(CHEMBL432537 | GNF-Pf-3777 | US10669273, Compound ...)Show SMILES [O-][N+](=O)c1ccc-2c(c1)C(=O)c1nc3ccccc3c(=O)n-21 Show InChI InChI=1S/C15H7N3O4/c19-13-10-7-8(18(21)22)5-6-12(10)17-14(13)16-11-4-2-1-3-9(11)15(17)20/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 54 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM48009

(8-fluoranylindolo[2,1-b]quinazoline-6,12-dione | 8...)Show InChI InChI=1S/C15H7FN2O2/c16-8-5-6-12-10(7-8)13(19)14-17-11-4-2-1-3-9(11)15(20)18(12)14/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 161 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442987

(8-Bromotryptanthrin | CHEMBL72165)Show InChI InChI=1S/C15H7BrN2O2/c16-8-5-6-12-10(7-8)13(19)14-17-11-4-2-1-3-9(11)15(20)18(12)14/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 389 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442993

(CHEMBL3087012)Show SMILES O[C@H]1[C@@H]2Nc3ccccc3C(=O)N2c2ccc(F)cc12 |r| Show InChI InChI=1S/C15H11FN2O2/c16-8-5-6-12-10(7-8)13(19)14-17-11-4-2-1-3-9(11)15(20)18(12)14/h1-7,13-14,17,19H/t13-,14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.54E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50240612
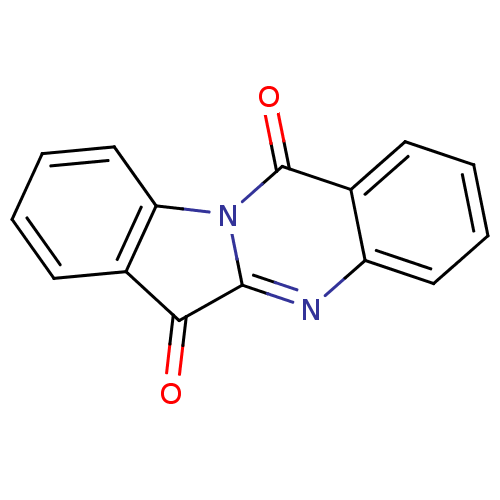
(CHEMBL306946 | GNF-PF-2691 | Indolo[2,1-b]quinazol...)Show InChI InChI=1S/C15H8N2O2/c18-13-10-6-2-4-8-12(10)17-14(13)16-11-7-3-1-5-9(11)15(17)19/h1-8H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.81E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Mixed competitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442988
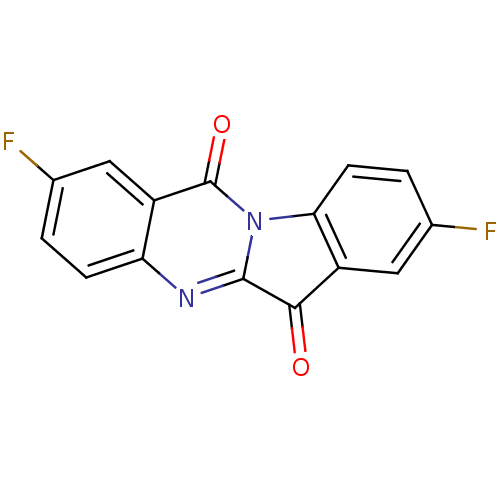
(CHEMBL3087009)Show InChI InChI=1S/C15H6F2N2O2/c16-7-1-3-11-9(5-7)15(21)19-12-4-2-8(17)6-10(12)13(20)14(19)18-11/h1-6H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.48E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442992

(CHEMBL2314972)Show SMILES O[C@H]1[C@@H]2Nc3ccccc3C(=O)N2c2ccccc12 |r| Show InChI InChI=1S/C15H12N2O2/c18-13-10-6-2-4-8-12(10)17-14(13)16-11-7-3-1-5-9(11)15(17)19/h1-8,13-14,16,18H/t13-,14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8.58E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442989
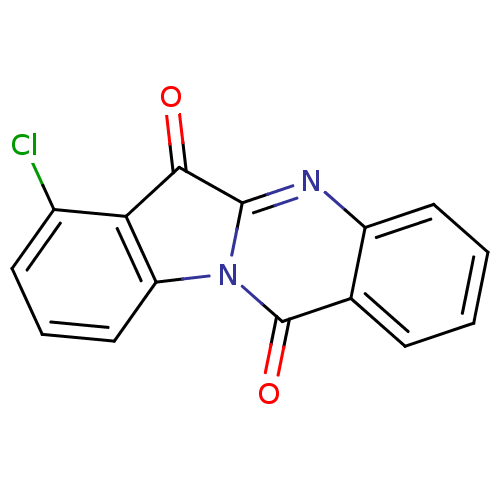
(CHEMBL3087010)Show InChI InChI=1S/C15H7ClN2O2/c16-9-5-3-7-11-12(9)13(19)14-17-10-6-2-1-4-8(10)15(20)18(11)14/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9.65E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50241727
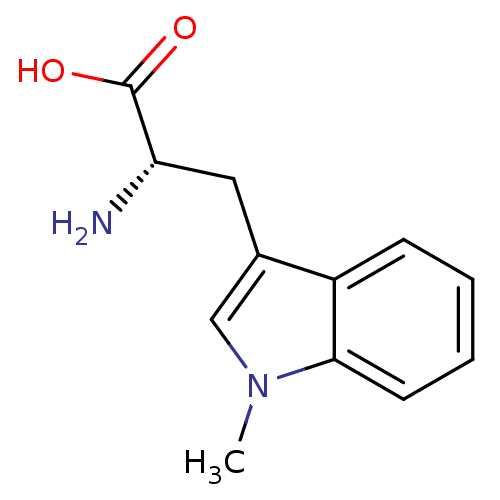
((S)-2-amino-3-(1-methyl-1H-indol-3-yl)propanoic ac...)Show InChI InChI=1S/C12H14N2O2/c1-14-7-8(6-10(13)12(15)16)9-4-2-3-5-11(9)14/h2-5,7,10H,6,13H2,1H3,(H,15,16)/t10-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 3.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Competitive inhibition of human IDO1 in presence of tryptophan |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50241727

((S)-2-amino-3-(1-methyl-1H-indol-3-yl)propanoic ac...)Show InChI InChI=1S/C12H14N2O2/c1-14-7-8(6-10(13)12(15)16)9-4-2-3-5-11(9)14/h2-5,7,10H,6,13H2,1H3,(H,15,16)/t10-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 4.23E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442990
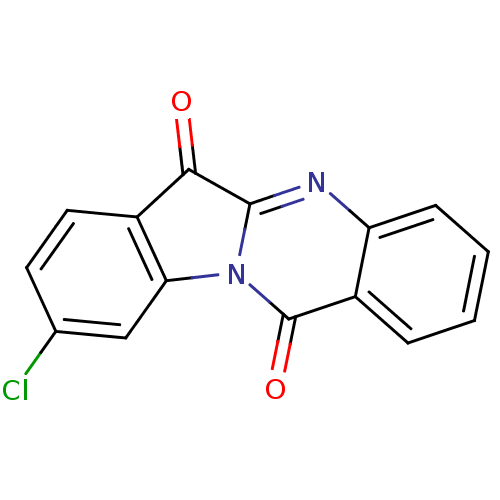
(CHEMBL1276265)Show InChI InChI=1S/C15H7ClN2O2/c16-8-5-6-10-12(7-8)18-14(13(10)19)17-11-4-2-1-3-9(11)15(18)20/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.94E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442986
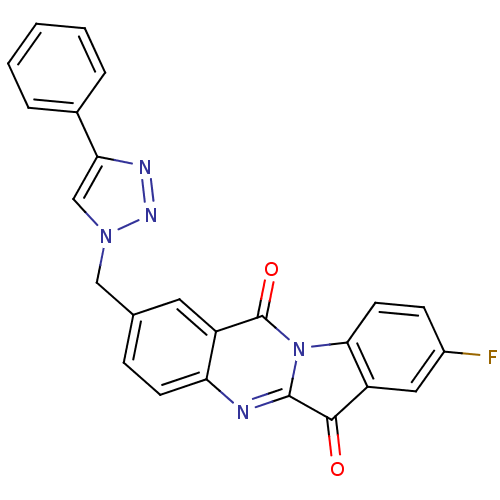
(CHEMBL3087014)Show SMILES Fc1ccc-2c(c1)C(=O)c1nc3ccc(Cn4cc(nn4)-c4ccccc4)cc3c(=O)n-21 Show InChI InChI=1S/C24H14FN5O2/c25-16-7-9-21-18(11-16)22(31)23-26-19-8-6-14(10-17(19)24(32)30(21)23)12-29-13-20(27-28-29)15-4-2-1-3-5-15/h1-11,13H,12H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 6.33E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442994
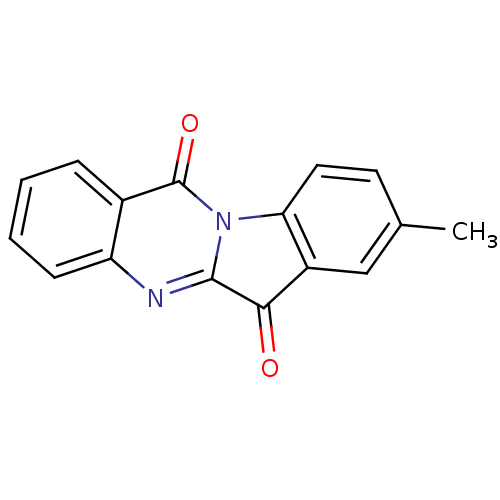
(CHEMBL312537 | US10669273, Compound 5b)Show InChI InChI=1S/C16H10N2O2/c1-9-6-7-13-11(8-9)14(19)15-17-12-5-3-2-4-10(12)16(20)18(13)15/h2-8H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.19E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Mixed competitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442991

(CHEMBL432537 | GNF-Pf-3777 | US10669273, Compound ...)Show SMILES [O-][N+](=O)c1ccc-2c(c1)C(=O)c1nc3ccccc3c(=O)n-21 Show InChI InChI=1S/C15H7N3O4/c19-13-10-7-8(18(21)22)5-6-12(10)17-14(13)16-11-4-2-1-3-9(11)15(17)20/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0180 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Inhibition of human IDO1 expressed in HEK293 cells assessed as kynurenine release after 5 hrs by spectrophotometry |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM48009

(8-fluoranylindolo[2,1-b]quinazoline-6,12-dione | 8...)Show InChI InChI=1S/C15H7FN2O2/c16-8-5-6-12-10(7-8)13(19)14-17-11-4-2-1-3-9(11)15(20)18(12)14/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Inhibition of human IDO1 expressed in HEK293 cells assessed as kynurenine release after 5 hrs by spectrophotometry |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442993

(CHEMBL3087012)Show SMILES O[C@H]1[C@@H]2Nc3ccccc3C(=O)N2c2ccc(F)cc12 |r| Show InChI InChI=1S/C15H11FN2O2/c16-8-5-6-12-10(7-8)13(19)14-17-11-4-2-1-3-9(11)15(20)18(12)14/h1-7,13-14,17,19H/t13-,14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Inhibition of human IDO1 expressed in HEK293 cells assessed as kynurenine release after 5 hrs by spectrophotometry |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442989

(CHEMBL3087010)Show InChI InChI=1S/C15H7ClN2O2/c16-9-5-3-7-11-12(9)13(19)14-17-10-6-2-1-4-8(10)15(20)18(11)14/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Inhibition of human IDO1 expressed in HEK293 cells assessed as kynurenine release after 5 hrs by spectrophotometry |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442987

(8-Bromotryptanthrin | CHEMBL72165)Show InChI InChI=1S/C15H7BrN2O2/c16-8-5-6-12-10(7-8)13(19)14-17-11-4-2-1-3-9(11)15(20)18(12)14/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 54 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Inhibition of human IDO1 expressed in HEK293 cells assessed as kynurenine release after 5 hrs by spectrophotometry |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50240612

(CHEMBL306946 | GNF-PF-2691 | Indolo[2,1-b]quinazol...)Show InChI InChI=1S/C15H8N2O2/c18-13-10-6-2-4-8-12(10)17-14(13)16-11-7-3-1-5-9(11)15(17)19/h1-8H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 54 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Inhibition of human IDO1 expressed in HEK293 cells assessed as kynurenine release after 5 hrs by spectrophotometry |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442991

(CHEMBL432537 | GNF-Pf-3777 | US10669273, Compound ...)Show SMILES [O-][N+](=O)c1ccc-2c(c1)C(=O)c1nc3ccccc3c(=O)n-21 Show InChI InChI=1S/C15H7N3O4/c19-13-10-7-8(18(21)22)5-6-12(10)17-14(13)16-11-4-2-1-3-9(11)15(17)20/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 103 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442990

(CHEMBL1276265)Show InChI InChI=1S/C15H7ClN2O2/c16-8-5-6-10-12(7-8)18-14(13(10)19)17-11-4-2-1-3-9(11)15(18)20/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 183 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Inhibition of human IDO1 expressed in HEK293 cells assessed as kynurenine release after 5 hrs by spectrophotometry |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442986

(CHEMBL3087014)Show SMILES Fc1ccc-2c(c1)C(=O)c1nc3ccc(Cn4cc(nn4)-c4ccccc4)cc3c(=O)n-21 Show InChI InChI=1S/C24H14FN5O2/c25-16-7-9-21-18(11-16)22(31)23-26-19-8-6-14(10-17(19)24(32)30(21)23)12-29-13-20(27-28-29)15-4-2-1-3-5-15/h1-11,13H,12H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 185 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Inhibition of human IDO1 expressed in HEK293 cells assessed as kynurenine release after 5 hrs by spectrophotometry |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442988

(CHEMBL3087009)Show InChI InChI=1S/C15H6F2N2O2/c16-7-1-3-11-9(5-7)15(21)19-12-4-2-8(17)6-10(12)13(20)14(19)18-11/h1-6H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 466 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Inhibition of human IDO1 expressed in HEK293 cells assessed as kynurenine release after 5 hrs by spectrophotometry |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM48009

(8-fluoranylindolo[2,1-b]quinazoline-6,12-dione | 8...)Show InChI InChI=1S/C15H7FN2O2/c16-8-5-6-12-10(7-8)13(19)14-17-11-4-2-1-3-9(11)15(20)18(12)14/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 534 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442987

(8-Bromotryptanthrin | CHEMBL72165)Show InChI InChI=1S/C15H7BrN2O2/c16-8-5-6-12-10(7-8)13(19)14-17-11-4-2-1-3-9(11)15(20)18(12)14/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 574 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442992

(CHEMBL2314972)Show SMILES O[C@H]1[C@@H]2Nc3ccccc3C(=O)N2c2ccccc12 |r| Show InChI InChI=1S/C15H12N2O2/c18-13-10-6-2-4-8-12(10)17-14(13)16-11-7-3-1-5-9(11)15(17)19/h1-8,13-14,16,18H/t13-,14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 779 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Inhibition of human IDO1 expressed in HEK293 cells assessed as kynurenine release after 5 hrs by spectrophotometry |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442988

(CHEMBL3087009)Show InChI InChI=1S/C15H6F2N2O2/c16-7-1-3-11-9(5-7)15(21)19-12-4-2-8(17)6-10(12)13(20)14(19)18-11/h1-6H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.72E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442993

(CHEMBL3087012)Show SMILES O[C@H]1[C@@H]2Nc3ccccc3C(=O)N2c2ccc(F)cc12 |r| Show InChI InChI=1S/C15H11FN2O2/c16-8-5-6-12-10(7-8)13(19)14-17-11-4-2-1-3-9(11)15(20)18(12)14/h1-7,13-14,17,19H/t13-,14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.21E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50240612

(CHEMBL306946 | GNF-PF-2691 | Indolo[2,1-b]quinazol...)Show InChI InChI=1S/C15H8N2O2/c18-13-10-6-2-4-8-12(10)17-14(13)16-11-7-3-1-5-9(11)15(17)19/h1-8H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7.15E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Mixed competitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442989

(CHEMBL3087010)Show InChI InChI=1S/C15H7ClN2O2/c16-9-5-3-7-11-12(9)13(19)14-17-10-6-2-1-4-8(10)15(20)18(11)14/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.19E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442992

(CHEMBL2314972)Show SMILES O[C@H]1[C@@H]2Nc3ccccc3C(=O)N2c2ccccc12 |r| Show InChI InChI=1S/C15H12N2O2/c18-13-10-6-2-4-8-12(10)17-14(13)16-11-7-3-1-5-9(11)15(17)19/h1-8,13-14,16,18H/t13-,14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50241727

((S)-2-amino-3-(1-methyl-1H-indol-3-yl)propanoic ac...)Show InChI InChI=1S/C12H14N2O2/c1-14-7-8(6-10(13)12(15)16)9-4-2-3-5-11(9)14/h2-5,7,10H,6,13H2,1H3,(H,15,16)/t10-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.84E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Inhibition of human IDO1 expressed in HEK293 cells assessed as kynurenine release after 5 hrs by spectrophotometry |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442986

(CHEMBL3087014)Show SMILES Fc1ccc-2c(c1)C(=O)c1nc3ccc(Cn4cc(nn4)-c4ccccc4)cc3c(=O)n-21 Show InChI InChI=1S/C24H14FN5O2/c25-16-7-9-21-18(11-16)22(31)23-26-19-8-6-14(10-17(19)24(32)30(21)23)12-29-13-20(27-28-29)15-4-2-1-3-5-15/h1-11,13H,12H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.91E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442990

(CHEMBL1276265)Show InChI InChI=1S/C15H7ClN2O2/c16-8-5-6-10-12(7-8)18-14(13(10)19)17-11-4-2-1-3-9(11)15(18)20/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.44E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442994

(CHEMBL312537 | US10669273, Compound 5b)Show InChI InChI=1S/C16H10N2O2/c1-9-6-7-13-11(8-9)14(19)15-17-12-5-3-2-4-10(12)16(20)18(13)15/h2-8H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.21E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Mixed competitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50241727

((S)-2-amino-3-(1-methyl-1H-indol-3-yl)propanoic ac...)Show InChI InChI=1S/C12H14N2O2/c1-14-7-8(6-10(13)12(15)16)9-4-2-3-5-11(9)14/h2-5,7,10H,6,13H2,1H3,(H,15,16)/t10-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Inhibition of human IDO1-mediated tryptophan degradation expressed in human tumor cells |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442996
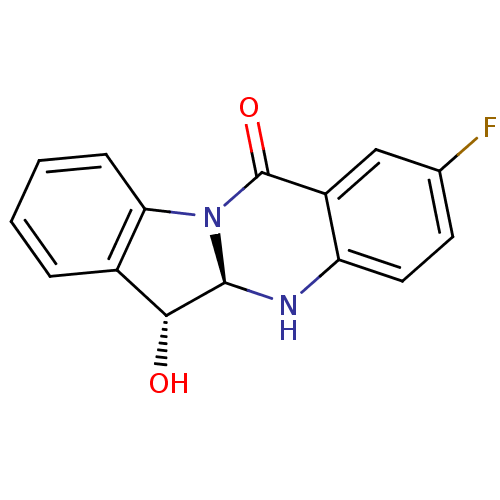
(CHEMBL3087011)Show SMILES O[C@H]1[C@@H]2Nc3ccc(F)cc3C(=O)N2c2ccccc12 |r| Show InChI InChI=1S/C15H11FN2O2/c16-8-5-6-11-10(7-8)15(20)18-12-4-2-1-3-9(12)13(19)14(18)17-11/h1-7,13-14,17,19H/t13-,14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.51E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50241727

((S)-2-amino-3-(1-methyl-1H-indol-3-yl)propanoic ac...)Show InChI InChI=1S/C12H14N2O2/c1-14-7-8(6-10(13)12(15)16)9-4-2-3-5-11(9)14/h2-5,7,10H,6,13H2,1H3,(H,15,16)/t10-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.99E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM50442995

(CHEMBL3086870)Show InChI InChI=1S/C15H7FN2O2/c16-8-5-6-11-10(7-8)15(20)18-12-4-2-1-3-9(12)13(19)14(18)17-11/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.64E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant IDO1 using L-tryptophan as substrate |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |
Indoleamine 2,3-dioxygenase 1
(Homo sapiens (Human)) | BDBM48009

(8-fluoranylindolo[2,1-b]quinazoline-6,12-dione | 8...)Show InChI InChI=1S/C15H7FN2O2/c16-8-5-6-12-10(7-8)13(19)14-17-11-4-2-1-3-9(11)15(20)18(12)14/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 4.68E+4 | n/a | n/a | n/a | n/a | n/a |
Fudan University
Curated by ChEMBL
| Assay Description
Binding affinity to human IDO1 by SPR assay |
J Med Chem 56: 8321-31 (2013)
Article DOI: 10.1021/jm401195n
BindingDB Entry DOI: 10.7270/Q2X34ZWS |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data