 Found 14 hits of Enzyme Inhibition Constant Data
Found 14 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50200841
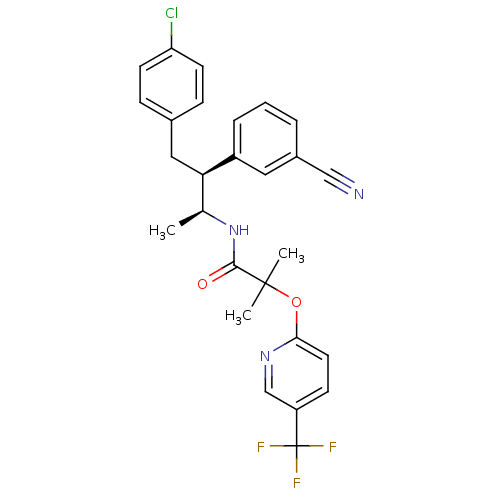
(CHEMBL220360 | MK-0364 | MK-0634 | N-((2S,3S)-4-(4...)Show SMILES C[C@H](NC(=O)C(C)(C)Oc1ccc(cn1)C(F)(F)F)[C@@H](Cc1ccc(Cl)cc1)c1cccc(c1)C#N |r| Show InChI InChI=1S/C27H25ClF3N3O2/c1-17(34-25(35)26(2,3)36-24-12-9-21(16-33-24)27(29,30)31)23(14-18-7-10-22(28)11-8-18)20-6-4-5-19(13-20)15-32/h4-13,16-17,23H,14H2,1-3H3,(H,34,35)/t17-,23+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]CP55940 from human recombinant CB1R expressed in CHO cells |
J Med Chem 50: 3427-30 (2007)
Article DOI: 10.1021/jm070131b
BindingDB Entry DOI: 10.7270/Q2Z037VJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50217220

(CHEMBL226590 | N-{(1S,2S)-2-(3-cyanophenyl)-3-[4-(...)Show SMILES C[C@H](NC(=O)C(C)(C)Oc1cc(C)ccn1)[C@@H](Cc1ccc(OCCF)cc1)c1cccc(c1)C#N Show InChI InChI=1S/C29H32FN3O3/c1-20-12-14-32-27(16-20)36-29(3,4)28(34)33-21(2)26(24-7-5-6-23(17-24)19-31)18-22-8-10-25(11-9-22)35-15-13-30/h5-12,14,16-17,21,26H,13,15,18H2,1-4H3,(H,33,34)/t21-,26+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.470 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]CP55940 from human recombinant CB1R expressed in CHO cells |
J Med Chem 50: 3427-30 (2007)
Article DOI: 10.1021/jm070131b
BindingDB Entry DOI: 10.7270/Q2Z037VJ |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50217216
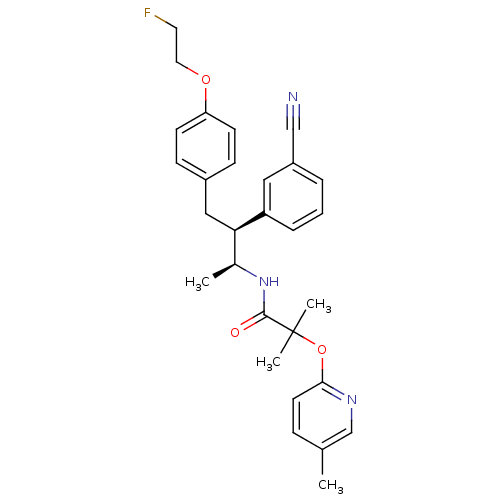
(CHEMBL226591 | CHEMBL226642 | N-{(1S,2S)-2-(3-cyan...)Show SMILES C[C@H](NC(=O)C(C)(C)Oc1ccc(C)cn1)[C@@H](Cc1ccc(OCCF)cc1)c1cccc(c1)C#N Show InChI InChI=1S/C29H32FN3O3/c1-20-8-13-27(32-19-20)36-29(3,4)28(34)33-21(2)26(24-7-5-6-23(16-24)18-31)17-22-9-11-25(12-10-22)35-15-14-30/h5-13,16,19,21,26H,14-15,17H2,1-4H3,(H,33,34)/t21-,26+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]CP55940 from human recombinant CB1R expressed in CHO cells |
J Med Chem 50: 3427-30 (2007)
Article DOI: 10.1021/jm070131b
BindingDB Entry DOI: 10.7270/Q2Z037VJ |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50217216

(CHEMBL226591 | CHEMBL226642 | N-{(1S,2S)-2-(3-cyan...)Show SMILES C[C@H](NC(=O)C(C)(C)Oc1ccc(C)cn1)[C@@H](Cc1ccc(OCCF)cc1)c1cccc(c1)C#N Show InChI InChI=1S/C29H32FN3O3/c1-20-8-13-27(32-19-20)36-29(3,4)28(34)33-21(2)26(24-7-5-6-23(16-24)18-31)17-22-9-11-25(12-10-22)35-15-14-30/h5-13,16,19,21,26H,14-15,17H2,1-4H3,(H,33,34)/t21-,26+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.730 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]CP55940 from human recombinant CB1R expressed in CHO cells |
J Med Chem 50: 3427-30 (2007)
Article DOI: 10.1021/jm070131b
BindingDB Entry DOI: 10.7270/Q2Z037VJ |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50217217
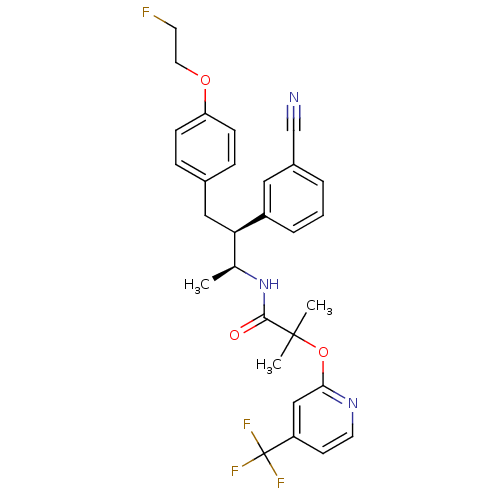
(CHEMBL376794 | N-{(1S,2S)-2-(3-cyanophenyl)-3-[4-(...)Show SMILES C[C@H](NC(=O)C(C)(C)Oc1cc(ccn1)C(F)(F)F)[C@@H](Cc1ccc(OCCF)cc1)c1cccc(c1)C#N Show InChI InChI=1S/C29H29F4N3O3/c1-19(36-27(37)28(2,3)39-26-17-23(11-13-35-26)29(31,32)33)25(22-6-4-5-21(15-22)18-34)16-20-7-9-24(10-8-20)38-14-12-30/h4-11,13,15,17,19,25H,12,14,16H2,1-3H3,(H,36,37)/t19-,25+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.770 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]CP55940 from human recombinant CB1R expressed in CHO cells |
J Med Chem 50: 3427-30 (2007)
Article DOI: 10.1021/jm070131b
BindingDB Entry DOI: 10.7270/Q2Z037VJ |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50217213
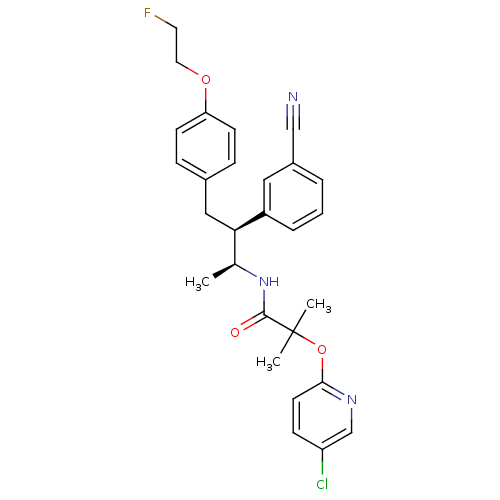
(CHEMBL226589 | N-{(1S,2S)-2-(3-cyanophenyl)-3-[4-(...)Show SMILES C[C@H](NC(=O)C(C)(C)Oc1ccc(Cl)cn1)[C@@H](Cc1ccc(OCCF)cc1)c1cccc(c1)C#N Show InChI InChI=1S/C28H29ClFN3O3/c1-19(33-27(34)28(2,3)36-26-12-9-23(29)18-32-26)25(22-6-4-5-21(15-22)17-31)16-20-7-10-24(11-8-20)35-14-13-30/h4-12,15,18-19,25H,13-14,16H2,1-3H3,(H,33,34)/t19-,25+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]CP55940 from human recombinant CB1R expressed in CHO cells |
J Med Chem 50: 3427-30 (2007)
Article DOI: 10.1021/jm070131b
BindingDB Entry DOI: 10.7270/Q2Z037VJ |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50217222

(CHEMBL226534 | N-{(1S,2S)-2-(3-cyanophenyl)-3-[4-(...)Show SMILES C[C@H](NC(=O)C(C)(C)Oc1ccc(cn1)C(F)(F)F)[C@@H](Cc1ccc(OCCF)cc1)c1cccc(c1)C#N Show InChI InChI=1S/C29H29F4N3O3/c1-19(36-27(37)28(2,3)39-26-12-9-23(18-35-26)29(31,32)33)25(22-6-4-5-21(15-22)17-34)16-20-7-10-24(11-8-20)38-14-13-30/h4-12,15,18-19,25H,13-14,16H2,1-3H3,(H,36,37)/t19-,25+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]CP55940 from human recombinant CB1R expressed in CHO cells |
J Med Chem 50: 3427-30 (2007)
Article DOI: 10.1021/jm070131b
BindingDB Entry DOI: 10.7270/Q2Z037VJ |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50217221
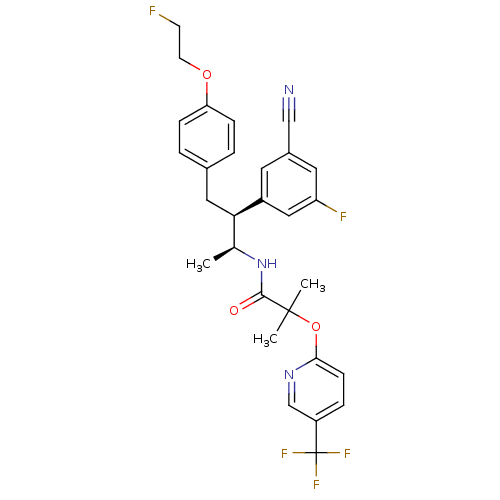
(CHEMBL376767 | N-{(1S,2S)-2-(3-cyano-5-fluoropheny...)Show SMILES C[C@H](NC(=O)C(C)(C)Oc1ccc(cn1)C(F)(F)F)[C@@H](Cc1ccc(OCCF)cc1)c1cc(F)cc(c1)C#N Show InChI InChI=1S/C29H28F5N3O3/c1-18(37-27(38)28(2,3)40-26-9-6-22(17-36-26)29(32,33)34)25(21-12-20(16-35)13-23(31)15-21)14-19-4-7-24(8-5-19)39-11-10-30/h4-9,12-13,15,17-18,25H,10-11,14H2,1-3H3,(H,37,38)/t18-,25+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]CP55940 from human recombinant CB1R expressed in CHO cells |
J Med Chem 50: 3427-30 (2007)
Article DOI: 10.1021/jm070131b
BindingDB Entry DOI: 10.7270/Q2Z037VJ |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50217223
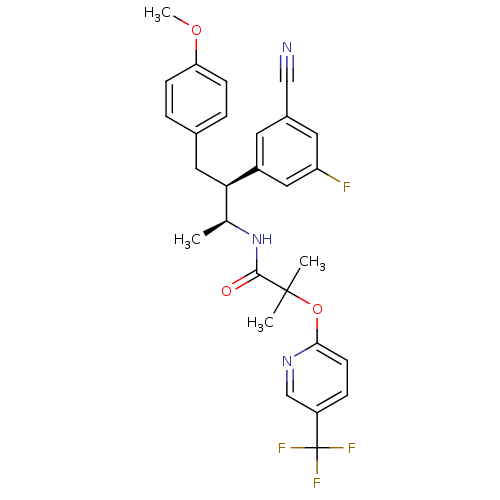
(CHEMBL226684 | N-[(1S,2S)-2-(3-cyano-5-fluoropheny...)Show SMILES COc1ccc(C[C@H]([C@H](C)NC(=O)C(C)(C)Oc2ccc(cn2)C(F)(F)F)c2cc(F)cc(c2)C#N)cc1 Show InChI InChI=1S/C28H27F4N3O3/c1-17(35-26(36)27(2,3)38-25-10-7-21(16-34-25)28(30,31)32)24(13-18-5-8-23(37-4)9-6-18)20-11-19(15-33)12-22(29)14-20/h5-12,14,16-17,24H,13H2,1-4H3,(H,35,36)/t17-,24+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]CP55940 from human recombinant CB1R expressed in CHO cells |
J Med Chem 50: 3427-30 (2007)
Article DOI: 10.1021/jm070131b
BindingDB Entry DOI: 10.7270/Q2Z037VJ |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50217218

(CHEMBL227329 | N-{(1S,2S)-2-(3-cyanophenyl)-3-[4-(...)Show SMILES C[C@H](NC(=O)C(C)(C)Oc1ccccn1)[C@@H](Cc1ccc(OCCF)cc1)c1cccc(c1)C#N Show InChI InChI=1S/C28H30FN3O3/c1-20(32-27(33)28(2,3)35-26-9-4-5-15-31-26)25(23-8-6-7-22(17-23)19-30)18-21-10-12-24(13-11-21)34-16-14-29/h4-13,15,17,20,25H,14,16,18H2,1-3H3,(H,32,33)/t20-,25+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]CP55940 from human recombinant CB1R expressed in CHO cells |
J Med Chem 50: 3427-30 (2007)
Article DOI: 10.1021/jm070131b
BindingDB Entry DOI: 10.7270/Q2Z037VJ |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50217224
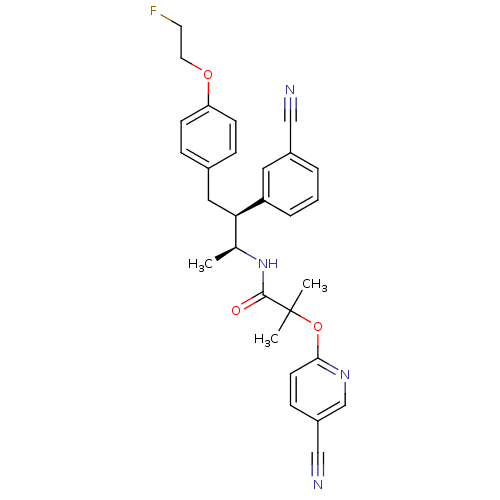
(CHEMBL387526 | N-{(1S,2S)-2-(3-cyanophenyl)-3-[4-(...)Show SMILES C[C@H](NC(=O)C(C)(C)Oc1ccc(cn1)C#N)[C@@H](Cc1ccc(OCCF)cc1)c1cccc(c1)C#N Show InChI InChI=1S/C29H29FN4O3/c1-20(34-28(35)29(2,3)37-27-12-9-23(18-32)19-33-27)26(24-6-4-5-22(15-24)17-31)16-21-7-10-25(11-8-21)36-14-13-30/h4-12,15,19-20,26H,13-14,16H2,1-3H3,(H,34,35)/t20-,26+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]CP55940 from human recombinant CB1R expressed in CHO cells |
J Med Chem 50: 3427-30 (2007)
Article DOI: 10.1021/jm070131b
BindingDB Entry DOI: 10.7270/Q2Z037VJ |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50217214

(CHEMBL226640 | N-{(1S,2S)-2-(3-cyanophenyl)-3-[4-(...)Show SMILES C[C@H](NC(=O)C(C)(C)Oc1cc(ncn1)C(F)(F)F)[C@@H](Cc1ccc(OCCF)cc1)c1cccc(c1)C#N Show InChI InChI=1S/C28H28F4N4O3/c1-18(36-26(37)27(2,3)39-25-15-24(28(30,31)32)34-17-35-25)23(21-6-4-5-20(13-21)16-33)14-19-7-9-22(10-8-19)38-12-11-29/h4-10,13,15,17-18,23H,11-12,14H2,1-3H3,(H,36,37)/t18-,23+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]CP55940 from human recombinant CB1R expressed in CHO cells |
J Med Chem 50: 3427-30 (2007)
Article DOI: 10.1021/jm070131b
BindingDB Entry DOI: 10.7270/Q2Z037VJ |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50217215
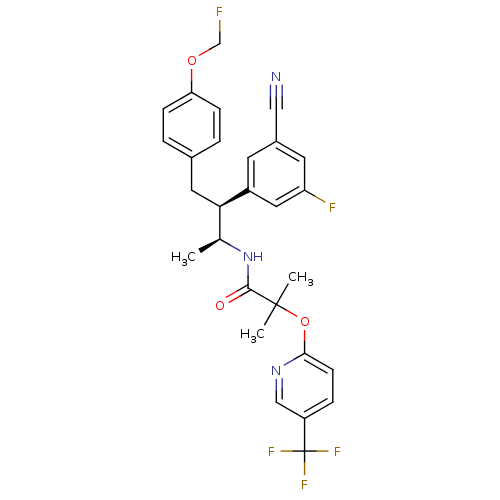
(CHEMBL226685 | N-[(1S,2S)-2-(3-cyano-5-fluoropheny...)Show SMILES C[C@H](NC(=O)C(C)(C)Oc1ccc(cn1)C(F)(F)F)[C@@H](Cc1ccc(OCF)cc1)c1cc(F)cc(c1)C#N Show InChI InChI=1S/C28H26F5N3O3/c1-17(36-26(37)27(2,3)39-25-9-6-21(15-35-25)28(31,32)33)24(20-10-19(14-34)11-22(30)13-20)12-18-4-7-23(8-5-18)38-16-29/h4-11,13,15,17,24H,12,16H2,1-3H3,(H,36,37)/t17-,24+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]CP55940 from human recombinant CB1R expressed in CHO cells |
J Med Chem 50: 3427-30 (2007)
Article DOI: 10.1021/jm070131b
BindingDB Entry DOI: 10.7270/Q2Z037VJ |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM50217216

(CHEMBL226591 | CHEMBL226642 | N-{(1S,2S)-2-(3-cyan...)Show SMILES C[C@H](NC(=O)C(C)(C)Oc1ccc(C)cn1)[C@@H](Cc1ccc(OCCF)cc1)c1cccc(c1)C#N Show InChI InChI=1S/C29H32FN3O3/c1-20-8-13-27(32-19-20)36-29(3,4)28(34)33-21(2)26(24-7-5-6-23(16-24)18-31)17-22-9-11-25(12-10-22)35-15-14-30/h5-13,16,19,21,26H,14-15,17H2,1-4H3,(H,33,34)/t21-,26+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 44 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Agonist activity at human CB2R |
J Med Chem 50: 3427-30 (2007)
Article DOI: 10.1021/jm070131b
BindingDB Entry DOI: 10.7270/Q2Z037VJ |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data