

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM21278 (5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D M£lndal Curated by ChEMBL | Assay Description Agonist activity at human CB1 receptor assessed as stimulation of [35S]GTPgammaS binding | Bioorg Med Chem Lett 20: 479-82 (2010) Article DOI: 10.1016/j.bmcl.2009.11.125 BindingDB Entry DOI: 10.7270/Q2X34XKB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM50305984 (5,6-bis(4-chlorophenyl)-N-(4,4-difluorocyclohexyl)...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D M£lndal Curated by ChEMBL | Assay Description Agonist activity at human CB1 receptor assessed as stimulation of [35S]GTPgammaS binding | Bioorg Med Chem Lett 20: 479-82 (2010) Article DOI: 10.1016/j.bmcl.2009.11.125 BindingDB Entry DOI: 10.7270/Q2X34XKB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM50305985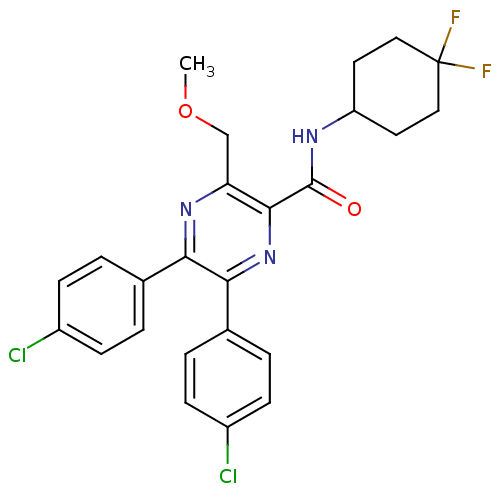 (5,6-bis(4-chlorophenyl)-N-(4,4-difluorocyclohexyl)...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D M£lndal Curated by ChEMBL | Assay Description Agonist activity at human CB1 receptor assessed as stimulation of [35S]GTPgammaS binding | Bioorg Med Chem Lett 20: 479-82 (2010) Article DOI: 10.1016/j.bmcl.2009.11.125 BindingDB Entry DOI: 10.7270/Q2X34XKB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM50305987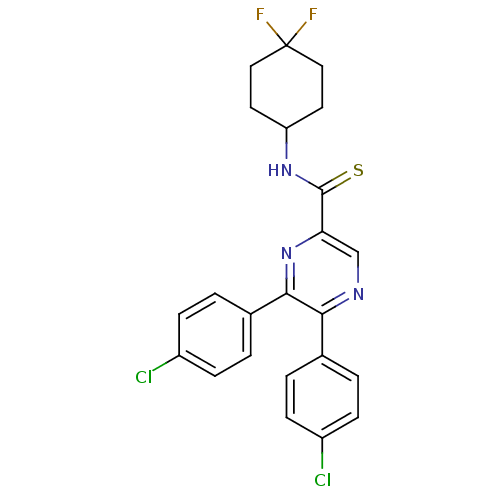 (5,6-bis(4-chlorophenyl)-N-(4,4-difluorocyclohexyl)...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D M£lndal Curated by ChEMBL | Assay Description Agonist activity at human CB1 receptor assessed as stimulation of [35S]GTPgammaS binding | Bioorg Med Chem Lett 20: 479-82 (2010) Article DOI: 10.1016/j.bmcl.2009.11.125 BindingDB Entry DOI: 10.7270/Q2X34XKB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM50305988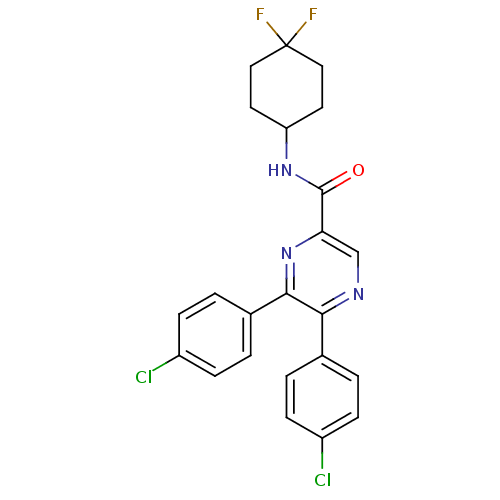 (5,6-bis(4-chlorophenyl)-N-(4,4-difluorocyclohexyl)...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D M£lndal Curated by ChEMBL | Assay Description Agonist activity at human CB1 receptor assessed as stimulation of [35S]GTPgammaS binding | Bioorg Med Chem Lett 20: 479-82 (2010) Article DOI: 10.1016/j.bmcl.2009.11.125 BindingDB Entry DOI: 10.7270/Q2X34XKB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM50305986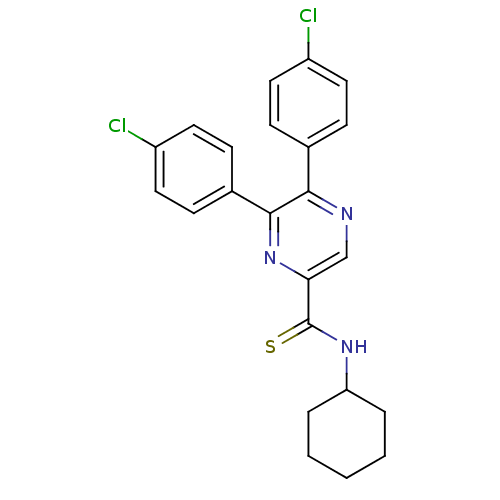 (5,6-bis(4-chlorophenyl)-N-cyclohexylpyrazine-2-car...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.80 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D M£lndal Curated by ChEMBL | Assay Description Agonist activity at human CB1 receptor assessed as stimulation of [35S]GTPgammaS binding | Bioorg Med Chem Lett 20: 479-82 (2010) Article DOI: 10.1016/j.bmcl.2009.11.125 BindingDB Entry DOI: 10.7270/Q2X34XKB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM35878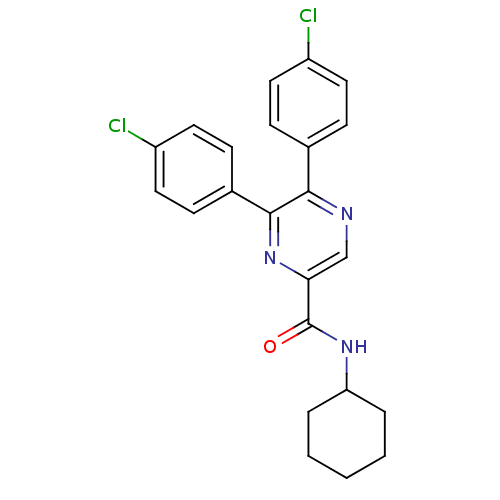 (5,6-bis(4-chlorophenyl)-N-cyclohexylpyrazine-2-car...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 16.6 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D M£lndal Curated by ChEMBL | Assay Description Agonist activity at human CB1 receptor assessed as stimulation of [35S]GTPgammaS binding | Bioorg Med Chem Lett 20: 479-82 (2010) Article DOI: 10.1016/j.bmcl.2009.11.125 BindingDB Entry DOI: 10.7270/Q2X34XKB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||