

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| REST corepressor 1 [4-485] (Homo sapiens (Human)) | BDBM50158884 (CHEMBL3785550) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University Curated by ChEMBL | Assay Description Inhibition of recombinant human His-tagged LSD1 (171 to 836 residues)/GST-tagged CoREST (308 to 440 residues) complex using H3K4 peptide substrate by... | Bioorg Med Chem 25: 1227-1234 (2017) Article DOI: 10.1016/j.bmc.2016.12.033 BindingDB Entry DOI: 10.7270/Q2X92D9V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50210048 (CHEMBL3884919) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University Curated by ChEMBL | Assay Description Inhibition of human LSD1 assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincubated for 10 mins foll... | Bioorg Med Chem 25: 1227-1234 (2017) Article DOI: 10.1016/j.bmc.2016.12.033 BindingDB Entry DOI: 10.7270/Q2X92D9V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50158884 (CHEMBL3785550) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University Curated by ChEMBL | Assay Description Inhibition of human LSD1 assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincubated for 10 mins foll... | Bioorg Med Chem 25: 1227-1234 (2017) Article DOI: 10.1016/j.bmc.2016.12.033 BindingDB Entry DOI: 10.7270/Q2X92D9V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50210048 (CHEMBL3884919) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University Curated by ChEMBL | Assay Description Inhibition of human LSD1 assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincubated for 10 mins foll... | Bioorg Med Chem 25: 1227-1234 (2017) Article DOI: 10.1016/j.bmc.2016.12.033 BindingDB Entry DOI: 10.7270/Q2X92D9V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50210048 (CHEMBL3884919) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University Curated by ChEMBL | Assay Description Inhibition of human LSD1 assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincubated for 10 mins foll... | Bioorg Med Chem 25: 1227-1234 (2017) Article DOI: 10.1016/j.bmc.2016.12.033 BindingDB Entry DOI: 10.7270/Q2X92D9V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50210049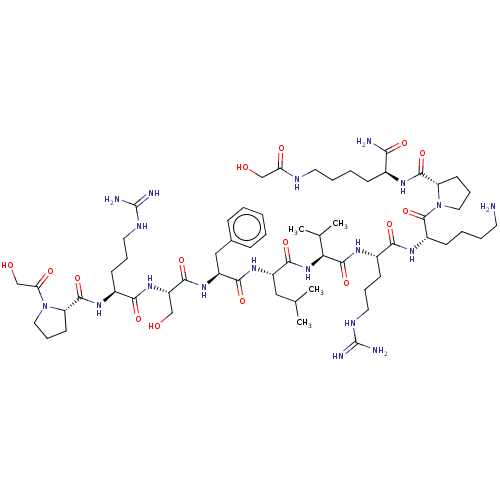 (CHEMBL3885101) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.90E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University Curated by ChEMBL | Assay Description Inhibition of human LSD1 assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincubated for 10 mins foll... | Bioorg Med Chem 25: 1227-1234 (2017) Article DOI: 10.1016/j.bmc.2016.12.033 BindingDB Entry DOI: 10.7270/Q2X92D9V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50210050 (CHEMBL3884529) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University Curated by ChEMBL | Assay Description Inhibition of human LSD1 assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincubated for 10 mins foll... | Bioorg Med Chem 25: 1227-1234 (2017) Article DOI: 10.1016/j.bmc.2016.12.033 BindingDB Entry DOI: 10.7270/Q2X92D9V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lysine-specific histone demethylase 1A (Homo sapiens (Human)) | BDBM50210050 (CHEMBL3884529) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University Curated by ChEMBL | Assay Description Inhibition of human LSD1 assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincubated for 10 mins foll... | Bioorg Med Chem 25: 1227-1234 (2017) Article DOI: 10.1016/j.bmc.2016.12.033 BindingDB Entry DOI: 10.7270/Q2X92D9V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||