

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM22541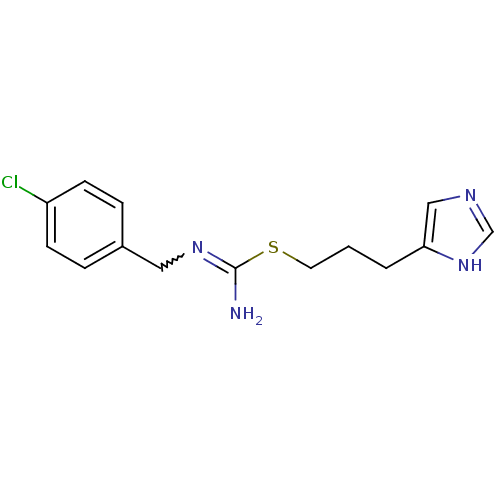 (Clobenpropit | N''-[(4-chlorophenyl)methyl]{[3-(1H...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | PubMed | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gliatech Inc. Curated by ChEMBL | Assay Description Inhibition of [3H]- N-alpha-methylhistamine from histamine H3 receptor | Bioorg Med Chem Lett 8: 1133-8 (1999) BindingDB Entry DOI: 10.7270/Q2GF0SPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50070217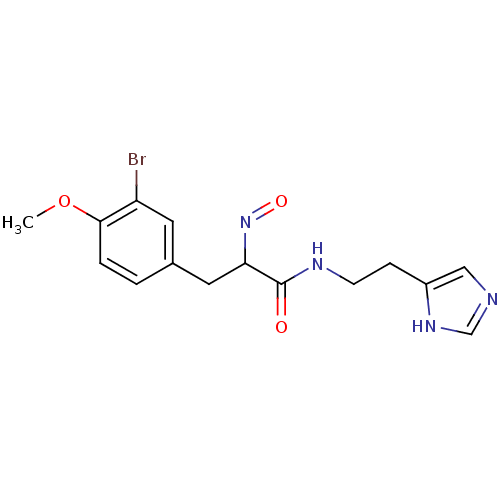 (3-(3-Bromo-4-methoxy-phenyl)-2-[(E)-hydroxyimino]-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gliatech Inc. Curated by ChEMBL | Assay Description Inhibition of [3H]- N-alpha-methylhistamine from histamine H3 receptor | Bioorg Med Chem Lett 8: 1133-8 (1999) BindingDB Entry DOI: 10.7270/Q2GF0SPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50070214 (4-(7,7-Dimethyl-oct-3-ynyl)-1H-imidazole | CHEMBL2...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gliatech Inc. Curated by ChEMBL | Assay Description Inhibition of [3H]- N-alpha-methylhistamine from histamine H3 receptor | Bioorg Med Chem Lett 8: 1133-8 (1999) BindingDB Entry DOI: 10.7270/Q2GF0SPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50070220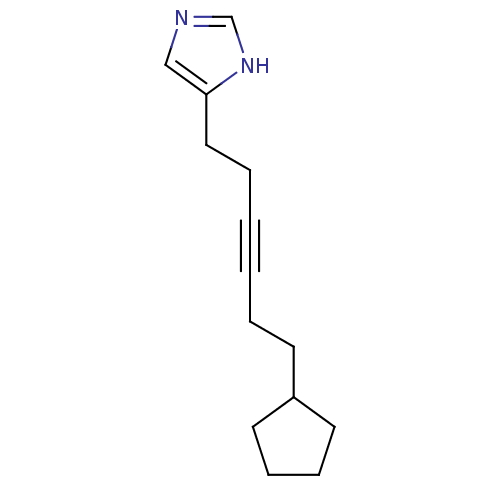 (4-(6-Cyclopentyl-hex-3-ynyl)-1H-imidazole | CHEMBL...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.950 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gliatech Inc. Curated by ChEMBL | Assay Description Inhibition of [3H]- N-alpha-methylhistamine from histamine H3 receptor | Bioorg Med Chem Lett 8: 1133-8 (1999) BindingDB Entry DOI: 10.7270/Q2GF0SPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM7967 (1-methyl-5-(beta-aminoethyl)-imidazole | 2-(1-meth...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents | PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gliatech Inc. Curated by ChEMBL | Assay Description Inhibition of [3H]- N-alpha-methylhistamine from histamine H3 receptor | Bioorg Med Chem Lett 8: 1133-8 (1999) BindingDB Entry DOI: 10.7270/Q2GF0SPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM22914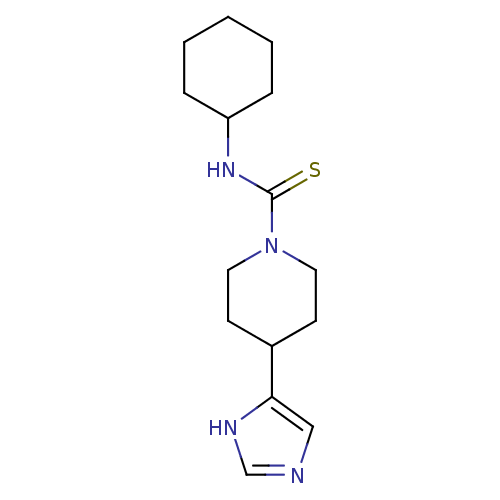 (CHEMBL260374 | N-cyclohexyl-4-(1H-imidazol-5-yl)pi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gliatech Inc. Curated by ChEMBL | Assay Description Antagonistic activity of the compound was evaluated against histamine H3 receptor using [3H]- N-alpha-methylhistamine as radioligand in experiment 2 | Bioorg Med Chem Lett 8: 1133-8 (1999) BindingDB Entry DOI: 10.7270/Q2GF0SPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50070211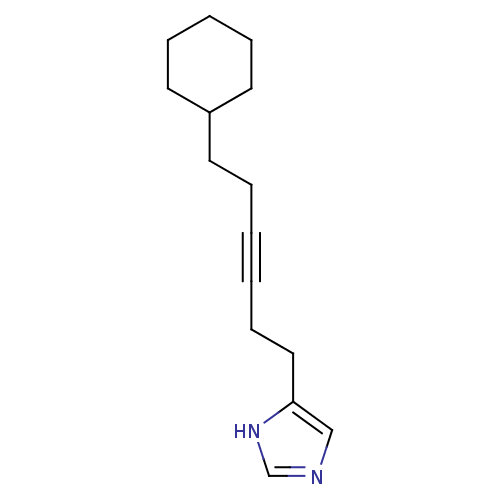 (4-(6-Cyclohexyl-hex-3-ynyl)-1H-imidazole | CHEMBL1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gliatech Inc. Curated by ChEMBL | Assay Description Inhibition of [3H]- N-alpha-methylhistamine from histamine H3 receptor | Bioorg Med Chem Lett 8: 1133-8 (1999) BindingDB Entry DOI: 10.7270/Q2GF0SPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50070216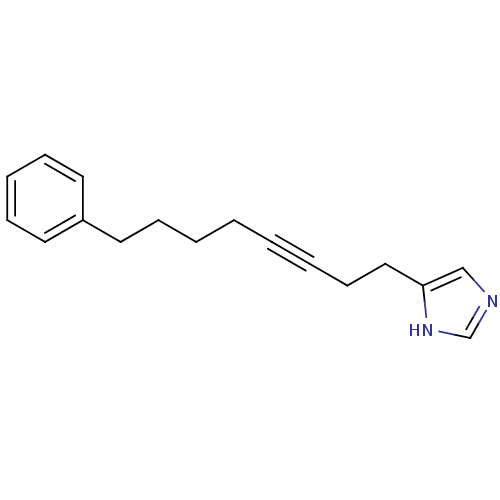 (4-(8-Phenyl-oct-3-ynyl)-1H-imidazole | CHEMBL14811) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | PubMed | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gliatech Inc. Curated by ChEMBL | Assay Description Inhibition of [3H]- N-alpha-methylhistamine from histamine H3 receptor | Bioorg Med Chem Lett 8: 1133-8 (1999) BindingDB Entry DOI: 10.7270/Q2GF0SPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50070212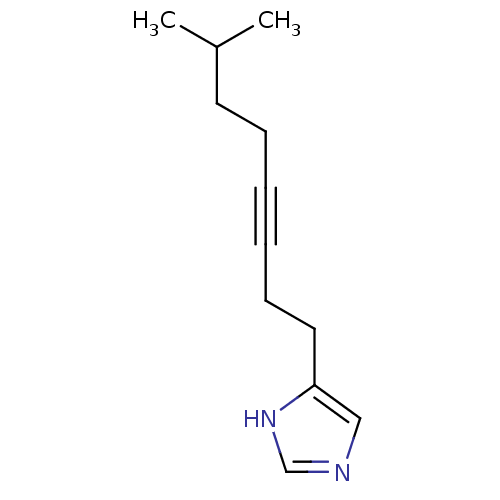 (4-(7-Methyl-oct-3-ynyl)-1H-imidazole | CHEMBL14497) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gliatech Inc. Curated by ChEMBL | Assay Description Inhibition of [3H]- N-alpha-methylhistamine from histamine H3 receptor | Bioorg Med Chem Lett 8: 1133-8 (1999) BindingDB Entry DOI: 10.7270/Q2GF0SPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM22914 (CHEMBL260374 | N-cyclohexyl-4-(1H-imidazol-5-yl)pi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 4.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gliatech Inc. Curated by ChEMBL | Assay Description Antagonistic activity of the compound was evaluated against histamine H3 receptor using [3H]- N-alpha-methylhistamine as radioligand in experiment 1 | Bioorg Med Chem Lett 8: 1133-8 (1999) BindingDB Entry DOI: 10.7270/Q2GF0SPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50070213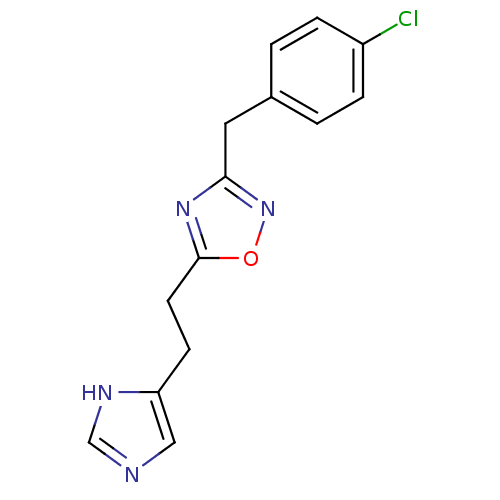 (3-(4-Chloro-benzyl)-5-[2-(1H-imidazol-4-yl)-ethyl]...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gliatech Inc. Curated by ChEMBL | Assay Description Inhibition of [3H]- N-alpha-methylhistamine from histamine H3 receptor | Bioorg Med Chem Lett 8: 1133-8 (1999) BindingDB Entry DOI: 10.7270/Q2GF0SPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50070215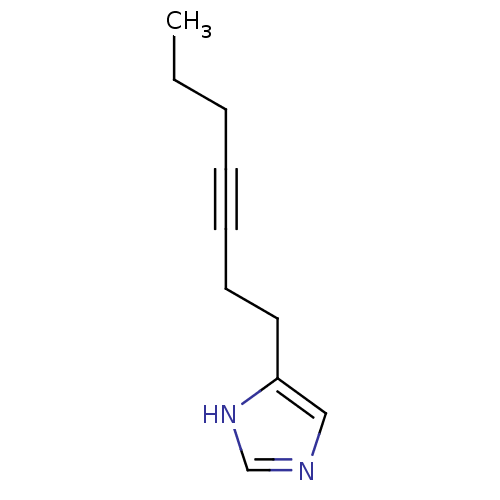 (4-Hept-3-ynyl-1H-imidazole | CHEMBL276804) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gliatech Inc. Curated by ChEMBL | Assay Description Inhibition of [3H]- N-alpha-methylhistamine from histamine H3 receptor | Bioorg Med Chem Lett 8: 1133-8 (1999) BindingDB Entry DOI: 10.7270/Q2GF0SPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM86490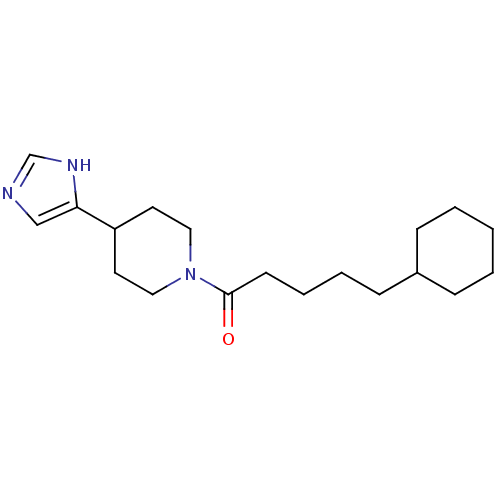 (CAS_0 | CHEMBL14812 | GT 2016 | NSC_0) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gliatech Inc. Curated by ChEMBL | Assay Description Inhibition of [3H]- N-alpha-methylhistamine from histamine H3 receptor | Bioorg Med Chem Lett 8: 1133-8 (1999) BindingDB Entry DOI: 10.7270/Q2GF0SPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50070219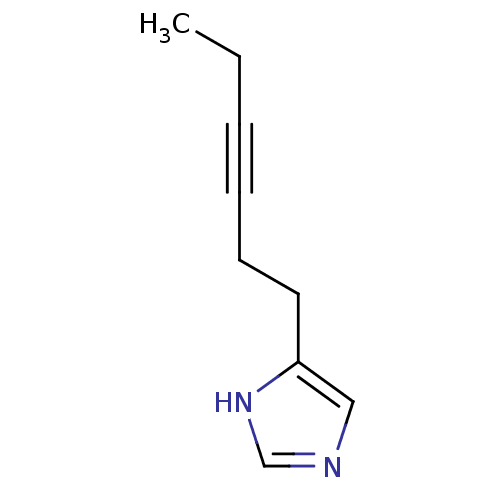 (4-Hex-3-ynyl-1H-imidazole | CHEMBL14721) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 79 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gliatech Inc. Curated by ChEMBL | Assay Description Inhibition of [3H]- N-alpha-methylhistamine from histamine H3 receptor | Bioorg Med Chem Lett 8: 1133-8 (1999) BindingDB Entry DOI: 10.7270/Q2GF0SPX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||