

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Prothrombin | ||
| Ligand | BDBM50215301 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_225420 (CHEMBL847455) | ||
| IC50 | 33±n/a nM | ||
| Citation |  Plummer, JS; Berryman, KA; Cai, C; Cody, WL; DiMaio, J; Doherty, AM; Edmunds, JJ; He, JX; Holland, DR; Levesque, S; Kent, DR; Narasimhan, LS; Rubin, JR; Rapundalo, ST; Siddiqui, MA; Susser, AJ; St-Denis, Y; Winocour, PD Potent and selective bicyclic lactam inhibitors of thrombin: Part 2: P1 modifications. Bioorg Med Chem Lett8:3409-14 (1999) [PubMed] Plummer, JS; Berryman, KA; Cai, C; Cody, WL; DiMaio, J; Doherty, AM; Edmunds, JJ; He, JX; Holland, DR; Levesque, S; Kent, DR; Narasimhan, LS; Rubin, JR; Rapundalo, ST; Siddiqui, MA; Susser, AJ; St-Denis, Y; Winocour, PD Potent and selective bicyclic lactam inhibitors of thrombin: Part 2: P1 modifications. Bioorg Med Chem Lett8:3409-14 (1999) [PubMed] | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Prothrombin | |||
| Name: | Prothrombin | ||
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain | ||
| Type: | Protein | ||
| Mol. Mass.: | 70029.57 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P00734 | ||
| Residue: | 622 | ||
| Sequence: |
| ||
| BDBM50215301 | |||
| n/a | |||
| Name | BDBM50215301 | ||
| Synonyms: | CHEMBL3349354 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C28H35N5O4S | ||
| Mol. Mass. | 537.674 | ||
| SMILES | [H][C@@]12CC[C@H](N1C(=O)CN(C2)C(=O)CCc1ccccc1)C(=O)N[C@H](C(=O)c1nccs1)[C@@]1([H])CC[C@H](N)CC1 |wU:4.23,36.40,1.0,32.36,wD:24.26,(11.74,-.27,;11.55,-2.13,;13.01,-1.64,;13.92,-2.88,;13.02,-4.12,;11.56,-3.66,;10.22,-4.44,;10.22,-5.99,;8.88,-3.68,;8.88,-2.14,;10.2,-1.36,;7.54,-1.36,;7.54,.18,;6.2,-2.14,;4.87,-1.38,;3.54,-2.16,;2.2,-1.39,;.87,-2.16,;.87,-3.7,;2.2,-4.47,;3.54,-3.7,;13.5,-5.6,;12.49,-6.74,;15.02,-5.9,;15.5,-7.37,;17.01,-7.69,;18.04,-6.53,;17.5,-9.15,;16.6,-10.4,;17.52,-11.64,;18.97,-11.16,;18.97,-9.62,;14.43,-8.42,;13.98,-6.94,;12.91,-8.14,;11.93,-9.31,;12.45,-10.76,;11.46,-11.95,;13.97,-11.04,;14.96,-9.85,)| | ||
| Structure | 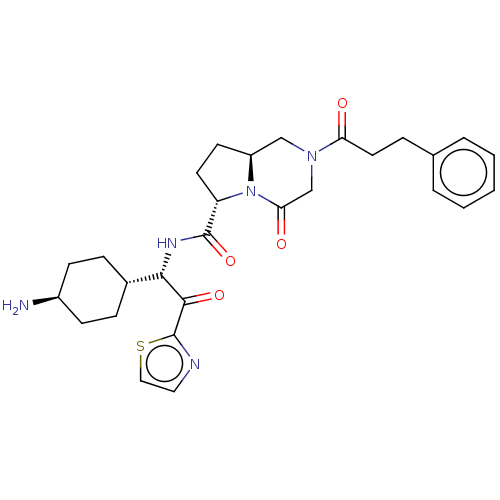
| ||