| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C19 |
|---|
| Ligand | BDBM50528194 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1992480 (CHEMBL4626215) |
|---|
| IC50 | >30000±n/a nM |
|---|
| Citation |  Smil, D; Wong, JF; Williams, EP; Adamson, RJ; Howarth, A; McLeod, DA; Mamai, A; Kim, S; Wilson, BJ; Kiyota, T; Aman, A; Owen, J; Poda, G; Horiuchi, KY; Kuznetsova, E; Ma, H; Hamblin, JN; Cramp, S; Roberts, OG; Edwards, AM; Uehling, D; Al-Awar, R; Bullock, AN; O'Meara, JA; Isaac, MB Leveraging an Open Science Drug Discovery Model to Develop CNS-Penetrant ALK2 Inhibitors for the Treatment of Diffuse Intrinsic Pontine Glioma. J Med Chem63:10061-10085 (2020) [PubMed] Article Smil, D; Wong, JF; Williams, EP; Adamson, RJ; Howarth, A; McLeod, DA; Mamai, A; Kim, S; Wilson, BJ; Kiyota, T; Aman, A; Owen, J; Poda, G; Horiuchi, KY; Kuznetsova, E; Ma, H; Hamblin, JN; Cramp, S; Roberts, OG; Edwards, AM; Uehling, D; Al-Awar, R; Bullock, AN; O'Meara, JA; Isaac, MB Leveraging an Open Science Drug Discovery Model to Develop CNS-Penetrant ALK2 Inhibitors for the Treatment of Diffuse Intrinsic Pontine Glioma. J Med Chem63:10061-10085 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C19 |
|---|
| Name: | Cytochrome P450 2C19 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | (S)-limonene 7-monooxygenase | CP2CJ_HUMAN | CYP2C19 | CYPIIC17 | CYPIIC19 | Cytochrome P450 2C19 (CYP2C19) | Cytochrome P450 2C19 [I331V] | Cytochrome P450-11A | Cytochrome P450-254C | Fenbendazole monooxygenase (4'-hydroxylating) | Mephenytoin 4-hydroxylase | P450-11A | P450-254C |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55935.47 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P33261 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDPFVVLVLCLSCLLLLSIWRQSSGRGKLPPGPTPLPVIGNILQIDIKDVSKSLTNLSKI
YGPVFTLYFGLERMVVLHGYEVVKEALIDLGEEFSGRGHFPLAERANRGFGIVFSNGKRW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFQKRFDYKDQQFLNLMEKLNENIRIVSTPWIQICNNFPTIIDYFPGTHNKLLKNLAFM
ESDILEKVKEHQESMDINNPRDFIDCFLIKMEKEKQNQQSEFTIENLVITAADLLGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRGHMPYTDAVVHEVQRYID
LIPTSLPHAVTCDVKFRNYLIPKGTTILTSLTSVLHDNKEFPNPEMFDPRHFLDEGGNFK
KSNYFMPFSAGKRICVGEGLARMELFLFLTFILQNFNLKSLIDPKDLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50528194 |
|---|
| n/a |
|---|
| Name | BDBM50528194 |
|---|
| Synonyms: | CHEMBL4517408 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H29N3O3 |
|---|
| Mol. Mass. | 419.5161 |
|---|
| SMILES | COc1cc(cc(OC)c1OC)-c1cncc(c1C)-c1ccc(cc1)N1CCNCC1 |
|---|
| Structure | 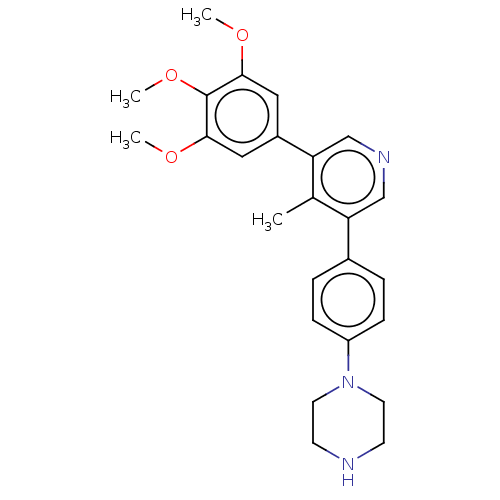
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Smil, D; Wong, JF; Williams, EP; Adamson, RJ; Howarth, A; McLeod, DA; Mamai, A; Kim, S; Wilson, BJ; Kiyota, T; Aman, A; Owen, J; Poda, G; Horiuchi, KY; Kuznetsova, E; Ma, H; Hamblin, JN; Cramp, S; Roberts, OG; Edwards, AM; Uehling, D; Al-Awar, R; Bullock, AN; O'Meara, JA; Isaac, MB Leveraging an Open Science Drug Discovery Model to Develop CNS-Penetrant ALK2 Inhibitors for the Treatment of Diffuse Intrinsic Pontine Glioma. J Med Chem63:10061-10085 (2020) [PubMed] Article
Smil, D; Wong, JF; Williams, EP; Adamson, RJ; Howarth, A; McLeod, DA; Mamai, A; Kim, S; Wilson, BJ; Kiyota, T; Aman, A; Owen, J; Poda, G; Horiuchi, KY; Kuznetsova, E; Ma, H; Hamblin, JN; Cramp, S; Roberts, OG; Edwards, AM; Uehling, D; Al-Awar, R; Bullock, AN; O'Meara, JA; Isaac, MB Leveraging an Open Science Drug Discovery Model to Develop CNS-Penetrant ALK2 Inhibitors for the Treatment of Diffuse Intrinsic Pontine Glioma. J Med Chem63:10061-10085 (2020) [PubMed] Article