| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50268936 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_519907 (CHEMBL964396) |
|---|
| IC50 | 2100±n/a nM |
|---|
| Citation |  Bailey, N; Bamford, MJ; Brissy, D; Brookfield, J; Demont, E; Elliott, R; Garton, N; Farre-Gutierrez, I; Hayhow, T; Hutley, G; Naylor, A; Panchal, TA; Seow, HX; Spalding, D; Takle, AK Orally active C-6 heteroaryl- and heterocyclyl-substituted imidazo[1,2-a]pyridine acid pump antagonists (APAs). Bioorg Med Chem Lett19:3602-6 (2009) [PubMed] Article Bailey, N; Bamford, MJ; Brissy, D; Brookfield, J; Demont, E; Elliott, R; Garton, N; Farre-Gutierrez, I; Hayhow, T; Hutley, G; Naylor, A; Panchal, TA; Seow, HX; Spalding, D; Takle, AK Orally active C-6 heteroaryl- and heterocyclyl-substituted imidazo[1,2-a]pyridine acid pump antagonists (APAs). Bioorg Med Chem Lett19:3602-6 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50268936 |
|---|
| n/a |
|---|
| Name | BDBM50268936 |
|---|
| Synonyms: | 3-(8-(2,6-dimethylbenzylamino)-2,3-dimethylimidazo[1,2-a]pyridin-6-yl)pyrimidin-4(3H)-one | CHEMBL523563 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H23N5O |
|---|
| Mol. Mass. | 373.4509 |
|---|
| SMILES | Cc1nc2c(NCc3c(C)cccc3C)cc(cn2c1C)-n1cnccc1=O |
|---|
| Structure | 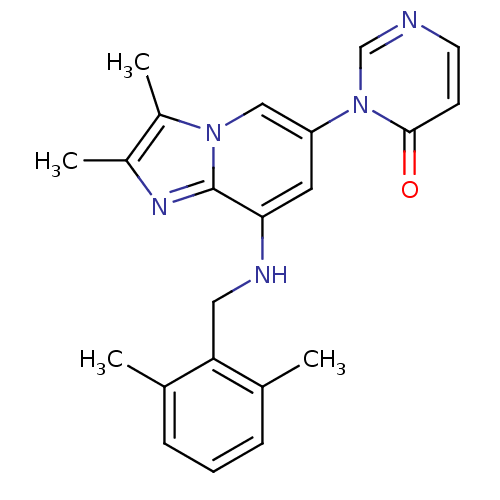
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bailey, N; Bamford, MJ; Brissy, D; Brookfield, J; Demont, E; Elliott, R; Garton, N; Farre-Gutierrez, I; Hayhow, T; Hutley, G; Naylor, A; Panchal, TA; Seow, HX; Spalding, D; Takle, AK Orally active C-6 heteroaryl- and heterocyclyl-substituted imidazo[1,2-a]pyridine acid pump antagonists (APAs). Bioorg Med Chem Lett19:3602-6 (2009) [PubMed] Article
Bailey, N; Bamford, MJ; Brissy, D; Brookfield, J; Demont, E; Elliott, R; Garton, N; Farre-Gutierrez, I; Hayhow, T; Hutley, G; Naylor, A; Panchal, TA; Seow, HX; Spalding, D; Takle, AK Orally active C-6 heteroaryl- and heterocyclyl-substituted imidazo[1,2-a]pyridine acid pump antagonists (APAs). Bioorg Med Chem Lett19:3602-6 (2009) [PubMed] Article