| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Smoothened homolog |
|---|
| Ligand | BDBM50268492 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_522936 (CHEMBL995458) |
|---|
| IC50 | 112±n/a nM |
|---|
| Citation |  Miller-Moslin, K; Peukert, S; Jain, RK; McEwan, MA; Karki, R; Llamas, L; Yusuff, N; He, F; Li, Y; Sun, Y; Dai, M; Perez, L; Michael, W; Sheng, T; Lei, H; Zhang, R; Williams, J; Bourret, A; Ramamurthy, A; Yuan, J; Guo, R; Matsumoto, M; Vattay, A; Maniara, W; Amaral, A; Dorsch, M; Kelleher, JF 1-amino-4-benzylphthalazines as orally bioavailable smoothened antagonists with antitumor activity. J Med Chem52:3954-68 (2009) [PubMed] Article Miller-Moslin, K; Peukert, S; Jain, RK; McEwan, MA; Karki, R; Llamas, L; Yusuff, N; He, F; Li, Y; Sun, Y; Dai, M; Perez, L; Michael, W; Sheng, T; Lei, H; Zhang, R; Williams, J; Bourret, A; Ramamurthy, A; Yuan, J; Guo, R; Matsumoto, M; Vattay, A; Maniara, W; Amaral, A; Dorsch, M; Kelleher, JF 1-amino-4-benzylphthalazines as orally bioavailable smoothened antagonists with antitumor activity. J Med Chem52:3954-68 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Smoothened homolog |
|---|
| Name: | Smoothened homolog |
|---|
| Synonyms: | SMO_MOUSE | Smo | Smoh |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 87472.73 |
|---|
| Organism: | Mus musculus |
|---|
| Description: | ChEMBL_1510377 |
|---|
| Residue: | 793 |
|---|
| Sequence: | MAAGRPVRGPELAPRRLLQLLLLVLLGGPGRGAALSGNVTGPGPHSASGSSRRDVPVTSP
PPPLLSHCGRAAHCEPLRYNVCLGSALPYGATTTLLAGDSDSQEEAHGKLVLWSGLRNAP
RCWAVIQPLLCAVYMPKCENDRVELPSRTLCQATRGPCAIVERERGWPDFLRCTPDHFPE
GCPNEVQNIKFNSSGQCEAPLVRTDNPKSWYEDVEGCGIQCQNPLFTEAEHQDMHSYIAA
FGAVTGLCTLFTLATFVADWRNSNRYPAVILFYVNACFFVGSIGWLAQFMDGARREIVCR
ADGTMRFGEPTSSETLSCVIIFVIVYYALMAGVVWFVVLTYAWHTSFKALGTTYQPLSGK
TSYFHLLTWSLPFVLTVAILAVAQVDGDSVSGICFVGYKNYRYRAGFVLAPIGLVLIVGG
YFLIRGVMTLFSIKSNHPGLLSEKAASKINETMLRLGIFGFLAFGFVLITFSCHFYDFFN
QAEWERSFRDYVLCQANVTIGLPTKKPIPDCEIKNRPSLLVEKINLFAMFGTGIAMSTWV
WTKATLLIWRRTWCRLTGHSDDEPKRIKKSKMIAKAFSKRRELLQNPGQELSFSMHTVSH
DGPVAGLAFDLNEPSADVSSAWAQHVTKMVARRGAILPQDVSVTPVATPVPPEEQANMWL
VEAEISPELEKRLGRKKKRRKRKKEVCPLRPAPELHHSAPVPATSAVPRLPQLPRQKCLV
AANAWGTGESCRQGAWTLVSNPFCPEPSPHQDPFLPGASAPRVWAQGRLQGLGSIHSRTN
LMEAEILDADSDF
|
|
|
|---|
| BDBM50268492 |
|---|
| n/a |
|---|
| Name | BDBM50268492 |
|---|
| Synonyms: | 6-{4-[4-(4-Fluorobenzyl)phthalazin-1-yl]piperazin-1-yl}nicotinonitrile | CHEMBL496603 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H21FN6 |
|---|
| Mol. Mass. | 424.4728 |
|---|
| SMILES | Fc1ccc(Cc2nnc(N3CCN(CC3)c3ccc(cn3)C#N)c3ccccc23)cc1 |
|---|
| Structure | 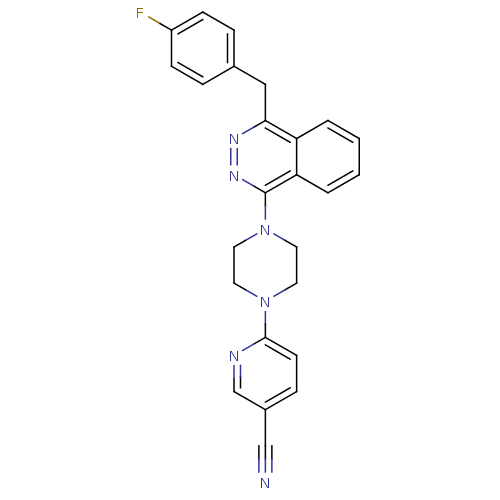
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Miller-Moslin, K; Peukert, S; Jain, RK; McEwan, MA; Karki, R; Llamas, L; Yusuff, N; He, F; Li, Y; Sun, Y; Dai, M; Perez, L; Michael, W; Sheng, T; Lei, H; Zhang, R; Williams, J; Bourret, A; Ramamurthy, A; Yuan, J; Guo, R; Matsumoto, M; Vattay, A; Maniara, W; Amaral, A; Dorsch, M; Kelleher, JF 1-amino-4-benzylphthalazines as orally bioavailable smoothened antagonists with antitumor activity. J Med Chem52:3954-68 (2009) [PubMed] Article
Miller-Moslin, K; Peukert, S; Jain, RK; McEwan, MA; Karki, R; Llamas, L; Yusuff, N; He, F; Li, Y; Sun, Y; Dai, M; Perez, L; Michael, W; Sheng, T; Lei, H; Zhang, R; Williams, J; Bourret, A; Ramamurthy, A; Yuan, J; Guo, R; Matsumoto, M; Vattay, A; Maniara, W; Amaral, A; Dorsch, M; Kelleher, JF 1-amino-4-benzylphthalazines as orally bioavailable smoothened antagonists with antitumor activity. J Med Chem52:3954-68 (2009) [PubMed] Article