| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Potassium voltage-gated channel subfamily A member 5 |
|---|
| Ligand | BDBM50306905 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_608671 (CHEMBL1067155) |
|---|
| IC50 | 90±n/a nM |
|---|
| Citation |  Lloyd, J; Finlay, HJ; Vacarro, W; Hyunh, T; Kover, A; Bhandaru, R; Yan, L; Atwal, K; Conder, ML; Jenkins-West, T; Shi, H; Huang, C; Li, D; Sun, H; Levesque, P Pyrrolidine amides of pyrazolodihydropyrimidines as potent and selective KV1.5 blockers. Bioorg Med Chem Lett20:1436-9 (2010) [PubMed] Article Lloyd, J; Finlay, HJ; Vacarro, W; Hyunh, T; Kover, A; Bhandaru, R; Yan, L; Atwal, K; Conder, ML; Jenkins-West, T; Shi, H; Huang, C; Li, D; Sun, H; Levesque, P Pyrrolidine amides of pyrazolodihydropyrimidines as potent and selective KV1.5 blockers. Bioorg Med Chem Lett20:1436-9 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Potassium voltage-gated channel subfamily A member 5 |
|---|
| Name: | Potassium voltage-gated channel subfamily A member 5 |
|---|
| Synonyms: | HK2 | HPCN1 | KCNA5 | KCNA5_HUMAN | Potassium voltage-gated channel subfamily A member 5 | Voltage-gated potassium channel | Voltage-gated potassium channel subunit Kv1.5 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 67222.47 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P22460 |
|---|
| Residue: | 613 |
|---|
| Sequence: | MEIALVPLENGGAMTVRGGDEARAGCGQATGGELQCPPTAGLSDGPKEPAPKGRGAQRDA
DSGVRPLPPLPDPGVRPLPPLPEELPRPRRPPPEDEEEEGDPGLGTVEDQALGTASLHHQ
RVHINISGLRFETQLGTLAQFPNTLLGDPAKRLRYFDPLRNEYFFDRNRPSFDGILYYYQ
SGGRLRRPVNVSLDVFADEIRFYQLGDEAMERFREDEGFIKEEEKPLPRNEFQRQVWLIF
EYPESSGSARAIAIVSVLVILISIITFCLETLPEFRDERELLRHPPAPHQPPAPAPGANG
SGVMAPPSGPTVAPLLPRTLADPFFIVETTCVIWFTFELLVRFFACPSKAGFSRNIMNII
DVVAIFPYFITLGTELAEQQPGGGGGGQNGQQAMSLAILRVIRLVRVFRIFKLSRHSKGL
QILGKTLQASMRELGLLIFFLFIGVILFSSAVYFAEADNQGTHFSSIPDAFWWAVVTMTT
VGYGDMRPITVGGKIVGSLCAIAGVLTIALPVPVIVSNFNYFYHRETDHEEPAVLKEEQG
TQSQGPGLDRGVQRKVSGSRGSFCKAGGTLENADSARRGSCPLEKCNVKAKSNVDLRRSL
YALCLDTSRETDL
|
|
|
|---|
| BDBM50306905 |
|---|
| n/a |
|---|
| Name | BDBM50306905 |
|---|
| Synonyms: | (7-(3,4-dichlorophenyl)-5-methyl-4,7-dihydropyrazolo[1,5-a]pyrimidin-6-yl)((S)-2-(5-methylisoxazol-3-yl)pyrrolidin-1-yl)methanone | CHEMBL602854 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H21Cl2N5O2 |
|---|
| Mol. Mass. | 458.34 |
|---|
| SMILES | Cc1cc(no1)[C@@H]1CCCN1C(=O)C1C(c2ccc(Cl)c(Cl)c2)n2nccc2N=C1C |r,c:32| |
|---|
| Structure | 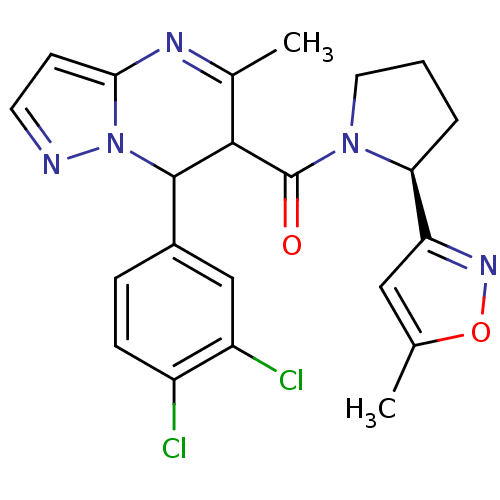
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lloyd, J; Finlay, HJ; Vacarro, W; Hyunh, T; Kover, A; Bhandaru, R; Yan, L; Atwal, K; Conder, ML; Jenkins-West, T; Shi, H; Huang, C; Li, D; Sun, H; Levesque, P Pyrrolidine amides of pyrazolodihydropyrimidines as potent and selective KV1.5 blockers. Bioorg Med Chem Lett20:1436-9 (2010) [PubMed] Article
Lloyd, J; Finlay, HJ; Vacarro, W; Hyunh, T; Kover, A; Bhandaru, R; Yan, L; Atwal, K; Conder, ML; Jenkins-West, T; Shi, H; Huang, C; Li, D; Sun, H; Levesque, P Pyrrolidine amides of pyrazolodihydropyrimidines as potent and selective KV1.5 blockers. Bioorg Med Chem Lett20:1436-9 (2010) [PubMed] Article