

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | C-C chemokine receptor type 5 | ||
| Ligand | BDBM50319441 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_634191 (CHEMBL1119773) | ||
| IC50 | 25±n/a nM | ||
| Citation |  Rotstein, DM; Gabriel, SD; Manser, N; Filonova, L; Padilla, F; Sankuratri, S; Ji, C; deRosier, A; Dioszegi, M; Heilek, G; Jekle, A; Weller, P; Berry, P Synthesis, SAR and evaluation of [1,4']-bipiperidinyl-4-yl-imidazolidin-2-one derivatives as novel CCR5 antagonists. Bioorg Med Chem Lett20:3219-22 (2010) [PubMed] Article Rotstein, DM; Gabriel, SD; Manser, N; Filonova, L; Padilla, F; Sankuratri, S; Ji, C; deRosier, A; Dioszegi, M; Heilek, G; Jekle, A; Weller, P; Berry, P Synthesis, SAR and evaluation of [1,4']-bipiperidinyl-4-yl-imidazolidin-2-one derivatives as novel CCR5 antagonists. Bioorg Med Chem Lett20:3219-22 (2010) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| C-C chemokine receptor type 5 | |||
| Name: | C-C chemokine receptor type 5 | ||
| Synonyms: | C-C CKR-5 | C-C chemokine receptor type 5 | CC-CKR-5 | CCR-5 | CCR5 | CCR5/mu opioid receptor complex | CCR5_HUMAN | CD_antigen=CD195 | CHEMR13 | CMKBR5 | HIV-1 fusion coreceptor | ||
| Type: | Enzyme | ||
| Mol. Mass.: | 40540.21 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P51681 | ||
| Residue: | 352 | ||
| Sequence: |
| ||
| BDBM50319441 | |||
| n/a | |||
| Name | BDBM50319441 | ||
| Synonyms: | 5-(4-((R)-3-((trans-4-ethoxycyclohexyl)methyl)-2-oxo-5-(pyridin-2-yl)imidazolidin-1-yl)-4'-methyl-1,4'-bipiperidine-1'-carbonyl)-4,6-dimethylpicolinonitrile | CHEMBL1085722 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C37H51N7O3 | ||
| Mol. Mass. | 641.8459 | ||
| SMILES | CCO[C@H]1CC[C@H](CN2C[C@@H](N(C3CCN(CC3)C3(C)CCN(CC3)C(=O)c3c(C)cc(nc3C)C#N)C2=O)c2ccccn2)CC1 |r,wU:6.6,wD:10.42,3.2,(15.1,-8.75,;15.11,-7.21,;16.45,-6.45,;16.46,-4.91,;17.8,-4.14,;17.81,-2.6,;16.47,-1.82,;16.47,-.28,;17.81,.48,;17.74,2.02,;19.18,2.56,;20.14,1.36,;21.68,1.43,;22.39,2.8,;23.94,2.88,;24.77,1.57,;24.05,.2,;22.51,.13,;26.3,1.64,;25.52,2.98,;27.12,.34,;28.65,.4,;29.37,1.76,;28.55,3.06,;27.01,3,;30.91,1.82,;31.63,3.18,;31.73,.52,;33.27,.58,;33.98,1.95,;34.09,-.71,;33.37,-2.08,;31.83,-2.14,;31.01,-.84,;29.47,-.9,;34.2,-3.39,;35.02,-4.7,;19.29,.07,;19.83,-1.37,;19.96,3.89,;19.19,5.22,;19.96,6.55,;21.51,6.55,;22.28,5.21,;21.5,3.88,;15.13,-2.59,;15.12,-4.13,)| | ||
| Structure | 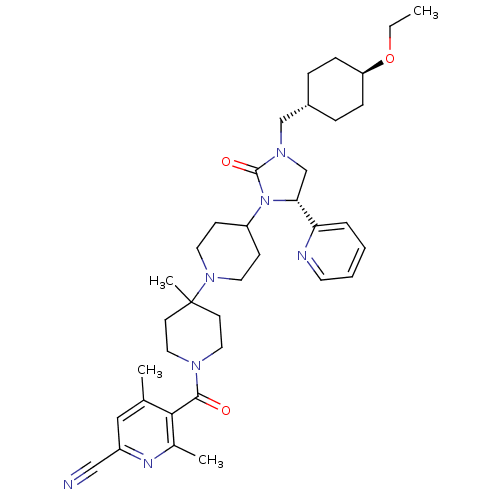
| ||