| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein phosphatase non-receptor type 1 |
|---|
| Ligand | BDBM50355635 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_776872 (CHEMBL1914006) |
|---|
| IC50 | >120000±n/a nM |
|---|
| Citation |  Sheriff, S; Beno, BR; Zhai, W; Kostich, WA; McDonnell, PA; Kish, K; Goldfarb, V; Gao, M; Kiefer, SE; Yanchunas, J; Huang, Y; Shi, S; Zhu, S; Dzierba, C; Bronson, J; Macor, JE; Appiah, KK; Westphal, RS; O'Connell, J; Gerritz, SW Small molecule receptor protein tyrosine phosphatase¿ (RPTP¿) ligands that inhibit phosphatase activity via perturbation of the tryptophan-proline-aspartate (WPD) loop. J Med Chem54:6548-62 (2011) [PubMed] Article Sheriff, S; Beno, BR; Zhai, W; Kostich, WA; McDonnell, PA; Kish, K; Goldfarb, V; Gao, M; Kiefer, SE; Yanchunas, J; Huang, Y; Shi, S; Zhu, S; Dzierba, C; Bronson, J; Macor, JE; Appiah, KK; Westphal, RS; O'Connell, J; Gerritz, SW Small molecule receptor protein tyrosine phosphatase¿ (RPTP¿) ligands that inhibit phosphatase activity via perturbation of the tryptophan-proline-aspartate (WPD) loop. J Med Chem54:6548-62 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein phosphatase non-receptor type 1 |
|---|
| Name: | Tyrosine-protein phosphatase non-receptor type 1 |
|---|
| Synonyms: | PTN1_HUMAN | PTP1B | PTPN1 | Protein tyrosine phosphatase 1B (PTP1B) | Protein tyrosine phosphatase-1B (PTP1B) | Protein-tyrosine phosphatase 1B | Protein-tyrosine phosphatase 1B (PTP1B) | Tyrosine-protein phosphatase non-receptor type 1 | Tyrosine-protein phosphatase non-receptor type 1 (PTP1B) |
|---|
| Type: | Protein phosphatase |
|---|
| Mol. Mass.: | 49963.76 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human recombinant GST-fusion PTP1B (1-435). |
|---|
| Residue: | 435 |
|---|
| Sequence: | MEMEKEFEQIDKSGSWAAIYQDIRHEASDFPCRVAKLPKNKNRNRYRDVSPFDHSRIKLH
QEDNDYINASLIKMEEAQRSYILTQGPLPNTCGHFWEMVWEQKSRGVVMLNRVMEKGSLK
CAQYWPQKEEKEMIFEDTNLKLTLISEDIKSYYTVRQLELENLTTQETREILHFHYTTWP
DFGVPESPASFLNFLFKVRESGSLSPEHGPVVVHCSAGIGRSGTFCLADTCLLLMDKRKD
PSSVDIKKVLLEMRKFRMGLIQTADQLRFSYLAVIEGAKFIMGDSSVQDQWKELSHEDLE
PPPEHIPPPPRPPKRILEPHNGKCREFFPNHQWVKEETQEDKDCPIKEEKGSPLNAAPYG
IESMSQDTEVRSRVVGGSLRGAQAASPAKGEPSLPEKDEDHALSYWKPFLVNMCVATVLT
AGAYLCYRFLFNSNT
|
|
|
|---|
| BDBM50355635 |
|---|
| n/a |
|---|
| Name | BDBM50355635 |
|---|
| Synonyms: | CHEMBL1910882 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C12H8Br2O2S2 |
|---|
| Mol. Mass. | 408.129 |
|---|
| SMILES | OC(=O)c1sccc1SCc1ccc(Br)c(Br)c1 |
|---|
| Structure | 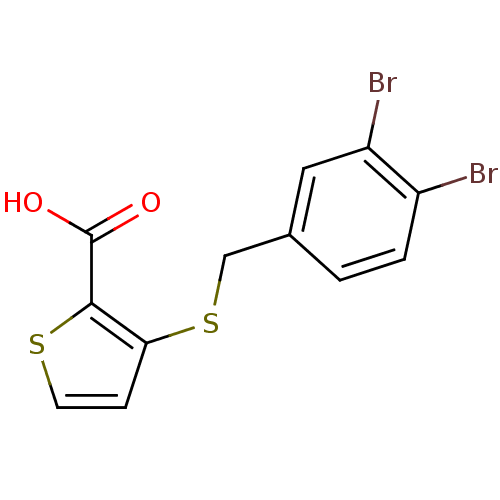
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sheriff, S; Beno, BR; Zhai, W; Kostich, WA; McDonnell, PA; Kish, K; Goldfarb, V; Gao, M; Kiefer, SE; Yanchunas, J; Huang, Y; Shi, S; Zhu, S; Dzierba, C; Bronson, J; Macor, JE; Appiah, KK; Westphal, RS; O'Connell, J; Gerritz, SW Small molecule receptor protein tyrosine phosphatase¿ (RPTP¿) ligands that inhibit phosphatase activity via perturbation of the tryptophan-proline-aspartate (WPD) loop. J Med Chem54:6548-62 (2011) [PubMed] Article
Sheriff, S; Beno, BR; Zhai, W; Kostich, WA; McDonnell, PA; Kish, K; Goldfarb, V; Gao, M; Kiefer, SE; Yanchunas, J; Huang, Y; Shi, S; Zhu, S; Dzierba, C; Bronson, J; Macor, JE; Appiah, KK; Westphal, RS; O'Connell, J; Gerritz, SW Small molecule receptor protein tyrosine phosphatase¿ (RPTP¿) ligands that inhibit phosphatase activity via perturbation of the tryptophan-proline-aspartate (WPD) loop. J Med Chem54:6548-62 (2011) [PubMed] Article