 Found 34075 hits with Last Name = 'shi' and Initial = 's'
Found 34075 hits with Last Name = 'shi' and Initial = 's' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Dihydrofolate reductase
(Escherichia coli) | BDBM18050

(2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)a...)Show SMILES CN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(cc1)C(=O)N[C@@H](CCC(O)=O)C(O)=O |r| Show InChI InChI=1S/C20H22N8O5/c1-28(9-11-8-23-17-15(24-11)16(21)26-20(22)27-17)12-4-2-10(3-5-12)18(31)25-13(19(32)33)6-7-14(29)30/h2-5,8,13H,6-7,9H2,1H3,(H,25,31)(H,29,30)(H,32,33)(H4,21,22,23,26,27)/t13-/m0/s1 | MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.00100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Connecticut
Curated by ChEMBL
| Assay Description
Inhibition of recombinant Escherichia coli DHFR |
ACS Med Chem Lett 7: 692-6 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00120
BindingDB Entry DOI: 10.7270/Q2DJ5HKK |
More data for this
Ligand-Target Pair | |
Dihydrofolate reductase
(Homo sapiens (Human)) | BDBM18050

(2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)a...)Show SMILES CN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(cc1)C(=O)N[C@@H](CCC(O)=O)C(O)=O |r| Show InChI InChI=1S/C20H22N8O5/c1-28(9-11-8-23-17-15(24-11)16(21)26-20(22)27-17)12-4-2-10(3-5-12)18(31)25-13(19(32)33)6-7-14(29)30/h2-5,8,13H,6-7,9H2,1H3,(H,25,31)(H,29,30)(H,32,33)(H4,21,22,23,26,27)/t13-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Connecticut
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human DHFR assessed as reduction in NADPH oxidation using dihydrofolate as substrate by fluorescence spectrophotometric ana... |
ACS Med Chem Lett 7: 692-6 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00120
BindingDB Entry DOI: 10.7270/Q2DJ5HKK |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cathepsin K
(Homo sapiens (Human)) | BDBM50098576
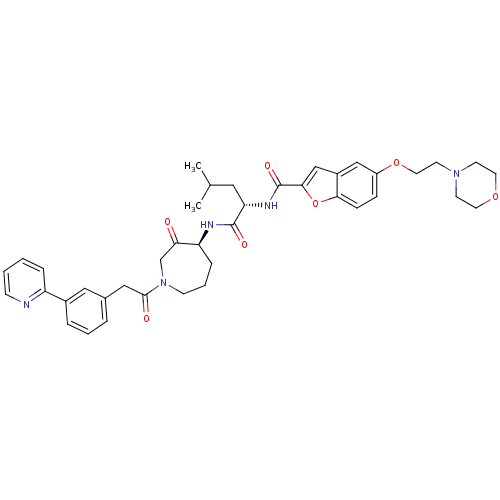
(5-(2-MORPHOLIN-4-YLETHOXY)BENZOFURAN-2-CARBOXYLIC ...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2cc(OCCN3CCOCC3)ccc2o1)C(=O)N[C@H]1CCCN(CC1=O)C(=O)Cc1cccc(c1)-c1ccccn1 Show InChI InChI=1S/C40H47N5O7/c1-27(2)21-34(43-40(49)37-25-30-24-31(11-12-36(30)52-37)51-20-17-44-15-18-50-19-16-44)39(48)42-33-10-6-14-45(26-35(33)46)38(47)23-28-7-5-8-29(22-28)32-9-3-4-13-41-32/h3-5,7-9,11-13,22,24-25,27,33-34H,6,10,14-21,23,26H2,1-2H3,(H,42,48)(H,43,49)/t33-,34-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
PubMed
| 0.00480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of Human cathepsin K |
J Med Chem 44: 1380-95 (2001)
BindingDB Entry DOI: 10.7270/Q2QR4WDC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Melatonin receptor type 1B
(Homo sapiens (Human)) | BDBM50343599
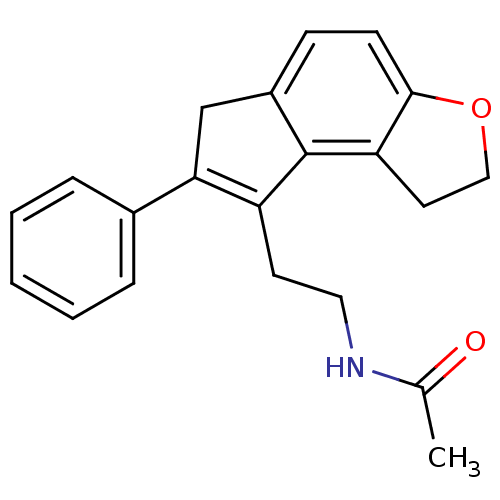
(CHEMBL1774522 | N-[2-(7-Phenyl-1,6-dihydro-2H-inde...)Show SMILES CC(=O)NCCC1=C(Cc2ccc3OCCc3c12)c1ccccc1 |t:6| Show InChI InChI=1S/C21H21NO2/c1-14(23)22-11-9-17-19(15-5-3-2-4-6-15)13-16-7-8-20-18(21(16)17)10-12-24-20/h2-8H,9-13H2,1H3,(H,22,23) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.00650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited
Curated by ChEMBL
| Assay Description
Displacement of 2-[125I]iodomelatonin from human MT2 receptor expressed in CHO cells |
J Med Chem 54: 4207-18 (2011)
Article DOI: 10.1021/jm200385u
BindingDB Entry DOI: 10.7270/Q2HT2PN3 |
More data for this
Ligand-Target Pair | |
Melatonin receptor type 1A
(Homo sapiens (Human)) | BDBM50343599

(CHEMBL1774522 | N-[2-(7-Phenyl-1,6-dihydro-2H-inde...)Show SMILES CC(=O)NCCC1=C(Cc2ccc3OCCc3c12)c1ccccc1 |t:6| Show InChI InChI=1S/C21H21NO2/c1-14(23)22-11-9-17-19(15-5-3-2-4-6-15)13-16-7-8-20-18(21(16)17)10-12-24-20/h2-8H,9-13H2,1H3,(H,22,23) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.00820 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited
Curated by ChEMBL
| Assay Description
Displacement of 2-[125I]iodomelatonin from human MT1 receptor expressed in CHO cells |
J Med Chem 54: 4207-18 (2011)
Article DOI: 10.1021/jm200385u
BindingDB Entry DOI: 10.7270/Q2HT2PN3 |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM19770
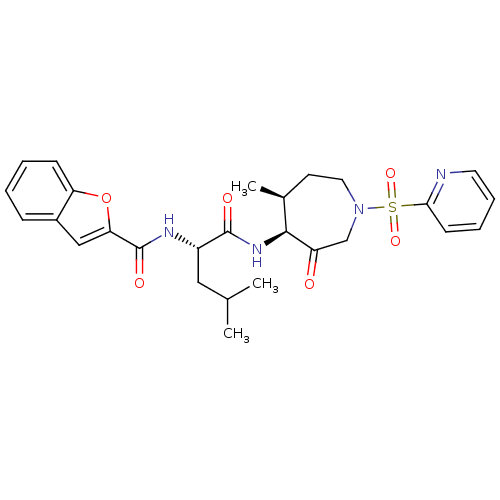
((2S)-2-(1-benzofuran-2-ylformamido)-4-methyl-N-[(4...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2ccccc2o1)C(=O)N[C@H]1[C@@H](C)CCN(CC1=O)S(=O)(=O)c1ccccn1 |r| Show InChI InChI=1S/C27H32N4O6S/c1-17(2)14-20(29-27(34)23-15-19-8-4-5-9-22(19)37-23)26(33)30-25-18(3)11-13-31(16-21(25)32)38(35,36)24-10-6-7-12-28-24/h4-10,12,15,17-18,20,25H,11,13-14,16H2,1-3H3,(H,29,34)(H,30,33)/t18-,20-,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.00990 | -62.2 | n/a | n/a | n/a | n/a | n/a | 5.5 | 22 |
GSK
| Assay Description
Potential inhibitors were evaluated using the progress curve method. Assays were carried out in the presence of variable concentrations of test compo... |
J Med Chem 49: 1597-612 (2006)
Article DOI: 10.1021/jm050915u
BindingDB Entry DOI: 10.7270/Q2SQ8XP5 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Muscarinic acetylcholine receptor M3
(RAT) | BDBM50021919
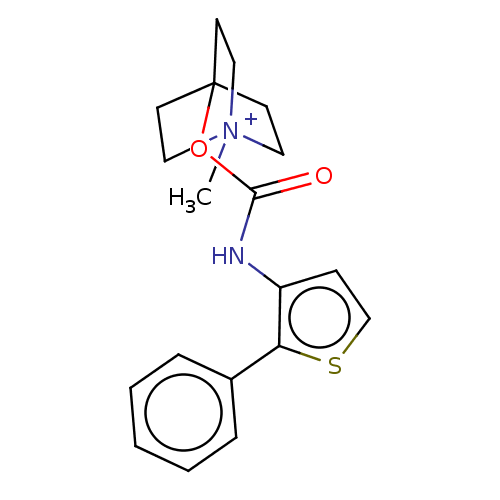
(CHEMBL3298595)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ccsc1-c1ccccc1 Show InChI InChI=1S/C19H22N2O2S.HI/c1-21-11-8-19(9-12-21,10-13-21)23-18(22)20-16-7-14-24-17(16)15-5-3-2-4-6-15;/h2-7,14H,8-13H2,1H3;1H | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M3 receptor in submandibular gland |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021919

(CHEMBL3298595)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ccsc1-c1ccccc1 Show InChI InChI=1S/C19H22N2O2S.HI/c1-21-11-8-19(9-12-21,10-13-21)23-18(22)20-16-7-14-24-17(16)15-5-3-2-4-6-15;/h2-7,14H,8-13H2,1H3;1H | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Melatonin receptor type 1A
(Homo sapiens (Human)) | BDBM50347590

(CHEMBL1802028)Show InChI InChI=1S/C17H21N3O2/c1-2-15(21)18-8-5-13-16(11-3-4-11)19-20-9-6-14-12(17(13)20)7-10-22-14/h6,9,11H,2-5,7-8,10H2,1H3,(H,18,21) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited
Curated by ChEMBL
| Assay Description
Displacement of 2-[125I]iodomelatonin from human MT1 receptor expressed in CHO cells |
J Med Chem 54: 4207-18 (2011)
Article DOI: 10.1021/jm200385u
BindingDB Entry DOI: 10.7270/Q2HT2PN3 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(RAT) | BDBM50021919

(CHEMBL3298595)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ccsc1-c1ccccc1 Show InChI InChI=1S/C19H22N2O2S.HI/c1-21-11-8-19(9-12-21,10-13-21)23-18(22)20-16-7-14-24-17(16)15-5-3-2-4-6-15;/h2-7,14H,8-13H2,1H3;1H | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M2 receptor in heart |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Melanin-concentrating hormone receptor 1
(Homo sapiens (Human)) | BDBM50295693
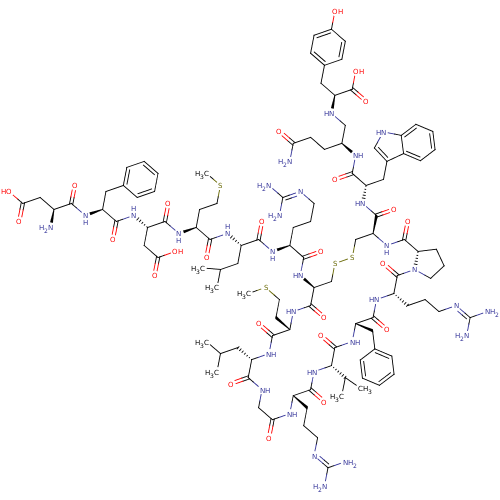
(CHEMBL557629)Show SMILES CSCC[C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@H]1CSSC[C@H](NC(=O)[C@@H]2CCCN2C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@H](CCCN=C(N)N)NC(=O)CNC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCSC)NC1=O)C(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCC(N)=O)CN[C@@H](Cc1ccc(O)cc1)C(O)=O |r,wU:27.28,36.36,71.73,148.153,55.55,60.134,8.12,wD:112.115,4.4,97.100,93.131,82.84,16.24,134.137,44.44,64.69,156.161,120.123,(5.27,-20.83,;6.6,-20.06,;6.6,-18.52,;5.27,-17.75,;5.27,-16.21,;3.93,-15.44,;2.6,-16.21,;2.6,-17.75,;1.27,-15.44,;1.27,-13.9,;2.6,-13.13,;3.93,-13.9,;2.6,-11.59,;-.07,-16.21,;-1.4,-15.44,;-1.4,-13.9,;-2.73,-16.21,;-2.73,-17.75,;-1.4,-18.52,;-.07,-17.74,;1.27,-18.51,;1.27,-20.05,;-.08,-20.82,;-1.4,-20.05,;-4.07,-15.44,;-5.4,-16.21,;-5.4,-17.75,;-6.74,-15.44,;-8.07,-16.21,;-6.74,-13.9,;-8.07,-13.13,;-8.07,-11.59,;-9.4,-13.9,;6.6,-15.44,;6.6,-13.9,;7.93,-16.21,;9.27,-15.44,;9.27,-13.9,;10.6,-13.13,;10.6,-11.59,;11.94,-13.9,;10.6,-16.21,;10.6,-17.75,;11.94,-15.44,;13.27,-16.21,;13.27,-17.75,;11.94,-18.52,;11.94,-20.06,;10.6,-20.83,;10.6,-22.37,;9.27,-23.14,;11.94,-23.14,;14.6,-15.44,;14.6,-13.9,;15.94,-16.21,;17.27,-15.44,;17.27,-13.9,;15.94,-13.13,;15.71,-11.29,;14.63,-10.2,;15.04,-8.71,;16.59,-8.74,;17.38,-7.41,;18.92,-7.43,;16.62,-6.07,;15.22,-6.04,;14.79,-4.38,;16.14,-3.62,;17.27,-4.66,;18.6,-3.89,;18.6,-2.35,;19.94,-4.66,;19.94,-6.2,;21.27,-6.97,;21.27,-8.51,;19.94,-9.28,;19.94,-10.82,;18.6,-11.59,;21.27,-11.59,;21.27,-3.89,;22.61,-4.66,;22.61,-6.2,;23.94,-3.89,;23.94,-2.35,;22.61,-1.58,;22.61,-.04,;21.28,.73,;19.95,-.04,;19.95,-1.59,;21.27,-2.35,;25.27,-4.66,;26.61,-3.89,;26.61,-2.35,;27.94,-4.66,;27.94,-6.2,;26.61,-6.97,;25.27,-6.2,;26.61,-8.51,;27.94,-9.28,;27.94,-10.82,;29.27,-11.59,;29.27,-13.13,;30.61,-13.9,;31.94,-13.13,;30.61,-15.44,;25.27,-9.28,;25.27,-10.82,;23.94,-11.59,;26.61,-11.59,;26.61,-13.13,;25.27,-13.9,;23.94,-13.13,;25.27,-15.44,;26.61,-16.21,;26.61,-17.75,;27.94,-18.52,;25.27,-18.52,;23.94,-16.21,;22.61,-15.44,;22.61,-13.9,;21.27,-16.21,;21.27,-17.75,;19.94,-18.52,;19.94,-20.06,;18.6,-20.83,;19.94,-15.44,;18.6,-16.21,;18.6,-17.75,;29.27,-3.89,;30.61,-4.66,;29.27,-2.35,;13.71,-7.94,;13.71,-6.4,;12.38,-8.71,;11.04,-7.94,;11.05,-6.4,;9.71,-5.63,;8.29,-6.29,;7.28,-5.11,;8.05,-3.78,;7.58,-2.33,;8.6,-1.2,;10.11,-1.52,;10.57,-2.98,;9.54,-4.11,;9.71,-8.71,;9.71,-10.25,;8.38,-7.93,;7.04,-8.7,;7.04,-10.24,;5.71,-11.01,;5.7,-12.55,;7.19,-12.94,;5.3,-14.03,;5.71,-7.93,;4.38,-8.7,;3.04,-7.93,;3.04,-6.39,;1.71,-5.62,;1.72,-4.07,;.39,-3.3,;-.95,-4.07,;-2.28,-3.3,;-.94,-5.62,;.39,-6.38,;1.71,-8.7,;.38,-7.92,;1.71,-10.24,)| Show InChI InChI=1S/C109H162N30O25S4/c1-58(2)45-75-91(148)123-55-86(142)125-70(27-17-39-118-107(112)113)95(152)138-89(60(5)6)104(161)135-78(48-62-23-13-10-14-24-62)98(155)129-74(29-19-41-120-109(116)117)105(162)139-42-20-30-84(139)103(160)137-83(102(159)133-79(50-64-53-121-69-26-16-15-25-67(64)69)96(153)124-65(33-36-85(111)141)54-122-81(106(163)164)49-63-31-34-66(140)35-32-63)57-168-167-56-82(101(158)128-73(38-44-166-8)93(150)131-75)136-92(149)71(28-18-40-119-108(114)115)126-97(154)76(46-59(3)4)132-94(151)72(37-43-165-7)127-100(157)80(52-88(145)146)134-99(156)77(47-61-21-11-9-12-22-61)130-90(147)68(110)51-87(143)144/h9-16,21-26,31-32,34-35,53,58-60,65,68,70-84,89,121-122,140H,17-20,27-30,33,36-52,54-57,110H2,1-8H3,(H2,111,141)(H,123,148)(H,124,153)(H,125,142)(H,126,154)(H,127,157)(H,128,158)(H,129,155)(H,130,147)(H,131,150)(H,132,151)(H,133,159)(H,134,156)(H,135,161)(H,136,149)(H,137,160)(H,138,152)(H,143,144)(H,145,146)(H,163,164)(H4,112,113,118)(H4,114,115,119)(H4,116,117,120)/t65-,68-,70-,71-,72-,73-,74-,75-,76-,77-,78-,79-,80-,81-,82-,83-,84-,89-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute
Curated by ChEMBL
| Assay Description
Displacement of [35S] 4-(1-(3,4-difluorophenyl)-2-(ethyl(3-(6-fluoro-3H-spiro[isobenzofuran-1,4'-piperidine]-1'-yl)propyl)amino)-2-oxoethyl)-3-oxopip... |
Bioorg Med Chem Lett 19: 2835-9 (2009)
Article DOI: 10.1016/j.bmcl.2009.03.102
BindingDB Entry DOI: 10.7270/Q2R49QS7 |
More data for this
Ligand-Target Pair | |
Melatonin receptor type 1A
(Homo sapiens (Human)) | BDBM50347593
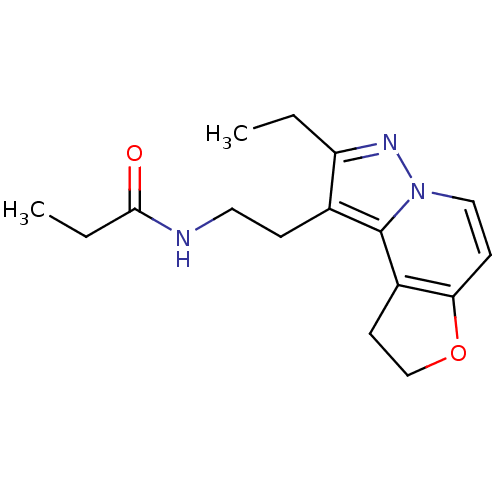
(CHEMBL1802026)Show InChI InChI=1S/C16H21N3O2/c1-3-13-11(5-8-17-15(20)4-2)16-12-7-10-21-14(12)6-9-19(16)18-13/h6,9H,3-5,7-8,10H2,1-2H3,(H,17,20) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited
Curated by ChEMBL
| Assay Description
Displacement of 2-[125I]iodomelatonin from human MT1 receptor expressed in CHO cells |
J Med Chem 54: 4207-18 (2011)
Article DOI: 10.1021/jm200385u
BindingDB Entry DOI: 10.7270/Q2HT2PN3 |
More data for this
Ligand-Target Pair | |
Procathepsin L
(Homo sapiens (Human)) | BDBM19770

((2S)-2-(1-benzofuran-2-ylformamido)-4-methyl-N-[(4...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2ccccc2o1)C(=O)N[C@H]1[C@@H](C)CCN(CC1=O)S(=O)(=O)c1ccccn1 |r| Show InChI InChI=1S/C27H32N4O6S/c1-17(2)14-20(29-27(34)23-15-19-8-4-5-9-22(19)37-23)26(33)30-25-18(3)11-13-31(16-21(25)32)38(35,36)24-10-6-7-12-28-24/h4-10,12,15,17-18,20,25H,11,13-14,16H2,1-3H3,(H,29,34)(H,30,33)/t18-,20-,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0400 | -58.8 | n/a | n/a | n/a | n/a | n/a | 5.5 | 22 |
GSK
| Assay Description
Potential inhibitors were evaluated using the progress curve method. Assays were carried out in the presence of variable concentrations of test compo... |
J Med Chem 49: 1597-612 (2006)
Article DOI: 10.1021/jm050915u
BindingDB Entry DOI: 10.7270/Q2SQ8XP5 |
More data for this
Ligand-Target Pair | |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50278693
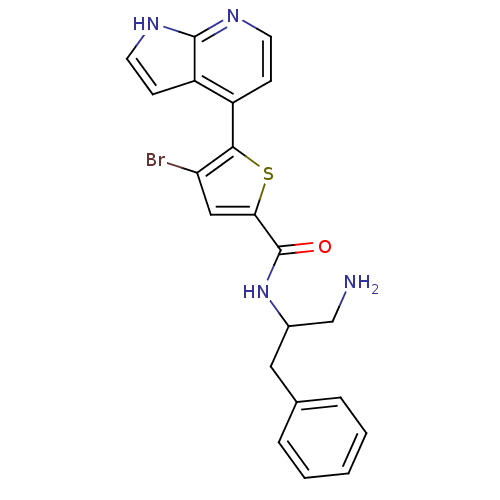
((+/-)-N-(1-amino-3-phenylpropan-2-yl)-4-bromo-5-(1...)Show SMILES NCC(Cc1ccccc1)NC(=O)c1cc(Br)c(s1)-c1ccnc2[nH]ccc12 Show InChI InChI=1S/C21H19BrN4OS/c22-17-11-18(21(27)26-14(12-23)10-13-4-2-1-3-5-13)28-19(17)15-6-8-24-20-16(15)7-9-25-20/h1-9,11,14H,10,12,23H2,(H,24,25)(H,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of AKT1 |
Bioorg Med Chem Lett 19: 2244-8 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.094
BindingDB Entry DOI: 10.7270/Q24X57PX |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM19778
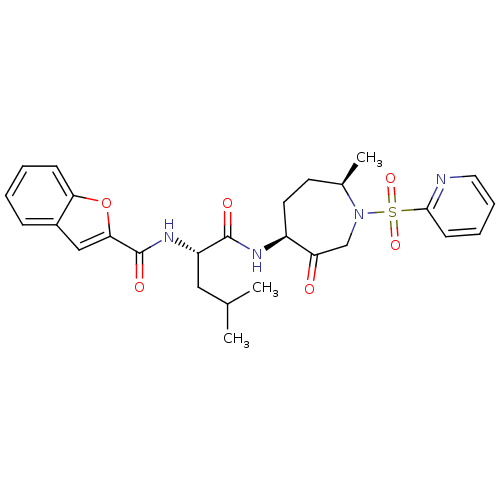
((2S)-2-(1-benzofuran-2-ylformamido)-4-methyl-N-[(4...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2ccccc2o1)C(=O)N[C@H]1CC[C@@H](C)N(CC1=O)S(=O)(=O)c1ccccn1 |r| Show InChI InChI=1S/C27H32N4O6S/c1-17(2)14-21(30-27(34)24-15-19-8-4-5-9-23(19)37-24)26(33)29-20-12-11-18(3)31(16-22(20)32)38(35,36)25-10-6-7-13-28-25/h4-10,13,15,17-18,20-21H,11-12,14,16H2,1-3H3,(H,29,33)(H,30,34)/t18-,20+,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0410 | -58.7 | n/a | n/a | n/a | n/a | n/a | 5.5 | 22 |
GSK
| Assay Description
Potential inhibitors were evaluated using the progress curve method. Assays were carried out in the presence of variable concentrations of test compo... |
J Med Chem 49: 1597-612 (2006)
Article DOI: 10.1021/jm050915u
BindingDB Entry DOI: 10.7270/Q2SQ8XP5 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021928
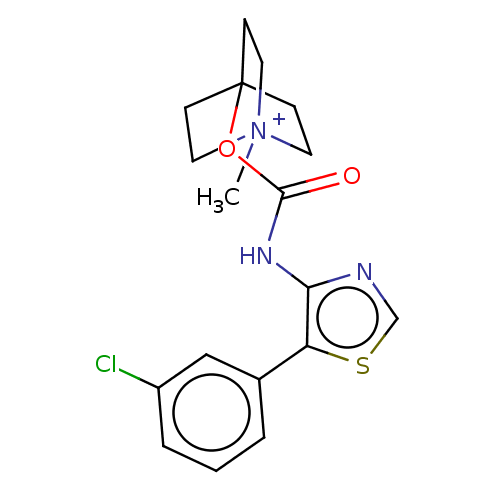
(CHEMBL3298599)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ncsc1-c1cccc(Cl)c1 Show InChI InChI=1S/C18H20ClN3O2S.HI/c1-22-8-5-18(6-9-22,7-10-22)24-17(23)21-16-15(25-12-20-16)13-3-2-4-14(19)11-13;/h2-4,11-12H,5-10H2,1H3;1H | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Melatonin receptor type 1A
(Homo sapiens (Human)) | BDBM50347589
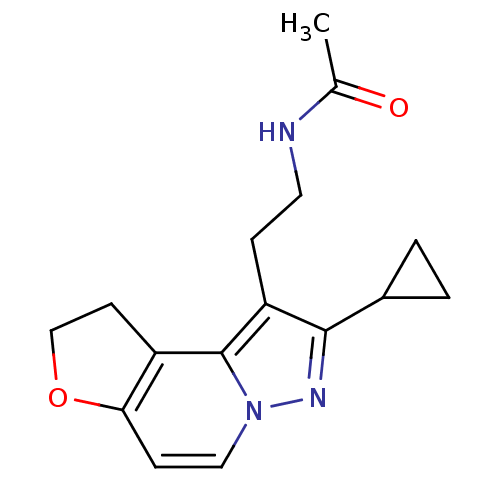
(CHEMBL1802027)Show InChI InChI=1S/C16H19N3O2/c1-10(20)17-7-4-13-15(11-2-3-11)18-19-8-5-14-12(16(13)19)6-9-21-14/h5,8,11H,2-4,6-7,9H2,1H3,(H,17,20) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited
Curated by ChEMBL
| Assay Description
Displacement of 2-[125I]iodomelatonin from human MT1 receptor expressed in CHO cells |
J Med Chem 54: 4207-18 (2011)
Article DOI: 10.1021/jm200385u
BindingDB Entry DOI: 10.7270/Q2HT2PN3 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(RAT) | BDBM50021928

(CHEMBL3298599)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ncsc1-c1cccc(Cl)c1 Show InChI InChI=1S/C18H20ClN3O2S.HI/c1-22-8-5-18(6-9-22,7-10-22)24-17(23)21-16-15(25-12-20-16)13-3-2-4-14(19)11-13;/h2-4,11-12H,5-10H2,1H3;1H | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M3 receptor in submandibular gland |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50278098
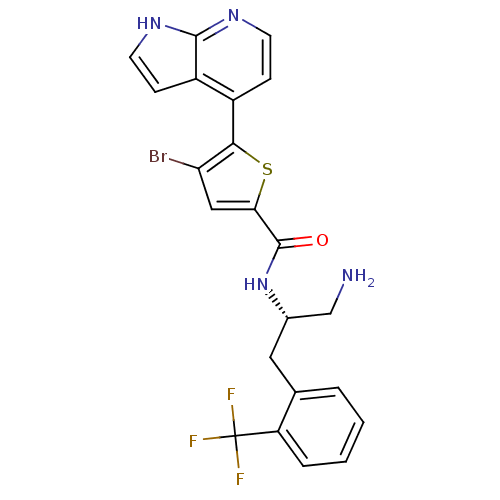
(CHEMBL520788 | N-((S)-1-amino-3-(2-(trifluoromethy...)Show SMILES NC[C@H](Cc1ccccc1C(F)(F)F)NC(=O)c1cc(Br)c(s1)-c1ccnc2[nH]ccc12 |r| Show InChI InChI=1S/C22H18BrF3N4OS/c23-17-10-18(32-19(17)14-5-7-28-20-15(14)6-8-29-20)21(31)30-13(11-27)9-12-3-1-2-4-16(12)22(24,25)26/h1-8,10,13H,9,11,27H2,(H,28,29)(H,30,31)/t13-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of AKT1 |
Bioorg Med Chem Lett 19: 2244-8 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.094
BindingDB Entry DOI: 10.7270/Q24X57PX |
More data for this
Ligand-Target Pair | |
Melanin-concentrating hormone receptor 1
(Mus musculus) | BDBM50295693

(CHEMBL557629)Show SMILES CSCC[C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@@H](N)CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@H]1CSSC[C@H](NC(=O)[C@@H]2CCCN2C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](NC(=O)[C@H](CCCN=C(N)N)NC(=O)CNC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCSC)NC1=O)C(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCC(N)=O)CN[C@@H](Cc1ccc(O)cc1)C(O)=O |r,wU:27.28,36.36,71.73,148.153,55.55,60.134,8.12,wD:112.115,4.4,97.100,93.131,82.84,16.24,134.137,44.44,64.69,156.161,120.123,(5.27,-20.83,;6.6,-20.06,;6.6,-18.52,;5.27,-17.75,;5.27,-16.21,;3.93,-15.44,;2.6,-16.21,;2.6,-17.75,;1.27,-15.44,;1.27,-13.9,;2.6,-13.13,;3.93,-13.9,;2.6,-11.59,;-.07,-16.21,;-1.4,-15.44,;-1.4,-13.9,;-2.73,-16.21,;-2.73,-17.75,;-1.4,-18.52,;-.07,-17.74,;1.27,-18.51,;1.27,-20.05,;-.08,-20.82,;-1.4,-20.05,;-4.07,-15.44,;-5.4,-16.21,;-5.4,-17.75,;-6.74,-15.44,;-8.07,-16.21,;-6.74,-13.9,;-8.07,-13.13,;-8.07,-11.59,;-9.4,-13.9,;6.6,-15.44,;6.6,-13.9,;7.93,-16.21,;9.27,-15.44,;9.27,-13.9,;10.6,-13.13,;10.6,-11.59,;11.94,-13.9,;10.6,-16.21,;10.6,-17.75,;11.94,-15.44,;13.27,-16.21,;13.27,-17.75,;11.94,-18.52,;11.94,-20.06,;10.6,-20.83,;10.6,-22.37,;9.27,-23.14,;11.94,-23.14,;14.6,-15.44,;14.6,-13.9,;15.94,-16.21,;17.27,-15.44,;17.27,-13.9,;15.94,-13.13,;15.71,-11.29,;14.63,-10.2,;15.04,-8.71,;16.59,-8.74,;17.38,-7.41,;18.92,-7.43,;16.62,-6.07,;15.22,-6.04,;14.79,-4.38,;16.14,-3.62,;17.27,-4.66,;18.6,-3.89,;18.6,-2.35,;19.94,-4.66,;19.94,-6.2,;21.27,-6.97,;21.27,-8.51,;19.94,-9.28,;19.94,-10.82,;18.6,-11.59,;21.27,-11.59,;21.27,-3.89,;22.61,-4.66,;22.61,-6.2,;23.94,-3.89,;23.94,-2.35,;22.61,-1.58,;22.61,-.04,;21.28,.73,;19.95,-.04,;19.95,-1.59,;21.27,-2.35,;25.27,-4.66,;26.61,-3.89,;26.61,-2.35,;27.94,-4.66,;27.94,-6.2,;26.61,-6.97,;25.27,-6.2,;26.61,-8.51,;27.94,-9.28,;27.94,-10.82,;29.27,-11.59,;29.27,-13.13,;30.61,-13.9,;31.94,-13.13,;30.61,-15.44,;25.27,-9.28,;25.27,-10.82,;23.94,-11.59,;26.61,-11.59,;26.61,-13.13,;25.27,-13.9,;23.94,-13.13,;25.27,-15.44,;26.61,-16.21,;26.61,-17.75,;27.94,-18.52,;25.27,-18.52,;23.94,-16.21,;22.61,-15.44,;22.61,-13.9,;21.27,-16.21,;21.27,-17.75,;19.94,-18.52,;19.94,-20.06,;18.6,-20.83,;19.94,-15.44,;18.6,-16.21,;18.6,-17.75,;29.27,-3.89,;30.61,-4.66,;29.27,-2.35,;13.71,-7.94,;13.71,-6.4,;12.38,-8.71,;11.04,-7.94,;11.05,-6.4,;9.71,-5.63,;8.29,-6.29,;7.28,-5.11,;8.05,-3.78,;7.58,-2.33,;8.6,-1.2,;10.11,-1.52,;10.57,-2.98,;9.54,-4.11,;9.71,-8.71,;9.71,-10.25,;8.38,-7.93,;7.04,-8.7,;7.04,-10.24,;5.71,-11.01,;5.7,-12.55,;7.19,-12.94,;5.3,-14.03,;5.71,-7.93,;4.38,-8.7,;3.04,-7.93,;3.04,-6.39,;1.71,-5.62,;1.72,-4.07,;.39,-3.3,;-.95,-4.07,;-2.28,-3.3,;-.94,-5.62,;.39,-6.38,;1.71,-8.7,;.38,-7.92,;1.71,-10.24,)| Show InChI InChI=1S/C109H162N30O25S4/c1-58(2)45-75-91(148)123-55-86(142)125-70(27-17-39-118-107(112)113)95(152)138-89(60(5)6)104(161)135-78(48-62-23-13-10-14-24-62)98(155)129-74(29-19-41-120-109(116)117)105(162)139-42-20-30-84(139)103(160)137-83(102(159)133-79(50-64-53-121-69-26-16-15-25-67(64)69)96(153)124-65(33-36-85(111)141)54-122-81(106(163)164)49-63-31-34-66(140)35-32-63)57-168-167-56-82(101(158)128-73(38-44-166-8)93(150)131-75)136-92(149)71(28-18-40-119-108(114)115)126-97(154)76(46-59(3)4)132-94(151)72(37-43-165-7)127-100(157)80(52-88(145)146)134-99(156)77(47-61-21-11-9-12-22-61)130-90(147)68(110)51-87(143)144/h9-16,21-26,31-32,34-35,53,58-60,65,68,70-84,89,121-122,140H,17-20,27-30,33,36-52,54-57,110H2,1-8H3,(H2,111,141)(H,123,148)(H,124,153)(H,125,142)(H,126,154)(H,127,157)(H,128,158)(H,129,155)(H,130,147)(H,131,150)(H,132,151)(H,133,159)(H,134,156)(H,135,161)(H,136,149)(H,137,160)(H,138,152)(H,143,144)(H,145,146)(H,163,164)(H4,112,113,118)(H4,114,115,119)(H4,116,117,120)/t65-,68-,70-,71-,72-,73-,74-,75-,76-,77-,78-,79-,80-,81-,82-,83-,84-,89-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute
Curated by ChEMBL
| Assay Description
Displacement of [35S] 4-(1-(3,4-difluorophenyl)-2-(ethyl(3-(6-fluoro-3H-spiro[isobenzofuran-1,4'-piperidine]-1'-yl)propyl)amino)-2-oxoethyl)-3-oxopip... |
Bioorg Med Chem Lett 19: 2835-9 (2009)
Article DOI: 10.1016/j.bmcl.2009.03.102
BindingDB Entry DOI: 10.7270/Q2R49QS7 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(RAT) | BDBM50021928

(CHEMBL3298599)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ncsc1-c1cccc(Cl)c1 Show InChI InChI=1S/C18H20ClN3O2S.HI/c1-22-8-5-18(6-9-22,7-10-22)24-17(23)21-16-15(25-12-20-16)13-3-2-4-14(19)11-13;/h2-4,11-12H,5-10H2,1H3;1H | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M2 receptor in heart |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021922
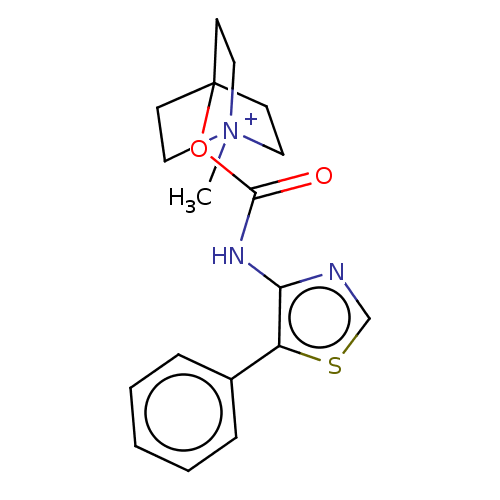
(CHEMBL3298596)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ncsc1-c1ccccc1 Show InChI InChI=1S/C18H21N3O2S.HI/c1-21-10-7-18(8-11-21,9-12-21)23-17(22)20-16-15(24-13-19-16)14-5-3-2-4-6-14;/h2-6,13H,7-12H2,1H3;1H | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(RAT) | BDBM50021904
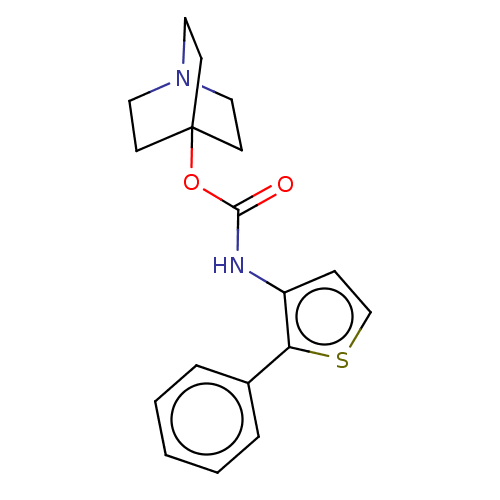
(CHEMBL3298588)Show InChI InChI=1S/C18H20N2O2S.ClH/c21-17(22-18-7-10-20(11-8-18)12-9-18)19-15-6-13-23-16(15)14-4-2-1-3-5-14;/h1-6,13H,7-12H2,(H,19,21);1H | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M3 receptor in submandibular gland |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50131385

((2R,3R,4R,5S)-3,4,5-Trihydroxy-1-(4-phenoxy-benzen...)Show SMILES ONC(=O)[C@H]1[C@@H](O)[C@H](O)[C@@H](O)CN1S(=O)(=O)c1ccc(Oc2ccccc2)cc1 |r| Show InChI InChI=1S/C18H20N2O8S/c21-14-10-20(15(18(24)19-25)17(23)16(14)22)29(26,27)13-8-6-12(7-9-13)28-11-4-2-1-3-5-11/h1-9,14-17,21-23,25H,10H2,(H,19,24)/t14-,15+,16+,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokkaido Collaboration Center
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human matrix metalloproteinase-9 |
Bioorg Med Chem Lett 13: 2737-40 (2003)
BindingDB Entry DOI: 10.7270/Q2PC31SG |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50131385

((2R,3R,4R,5S)-3,4,5-Trihydroxy-1-(4-phenoxy-benzen...)Show SMILES ONC(=O)[C@H]1[C@@H](O)[C@H](O)[C@@H](O)CN1S(=O)(=O)c1ccc(Oc2ccccc2)cc1 |r| Show InChI InChI=1S/C18H20N2O8S/c21-14-10-20(15(18(24)19-25)17(23)16(14)22)29(26,27)13-8-6-12(7-9-13)28-11-4-2-1-3-5-11/h1-9,14-17,21-23,25H,10H2,(H,19,24)/t14-,15+,16+,17+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokkaido Collaboration Center N-21
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant matrix metalloprotease 9 |
J Med Chem 47: 1930-8 (2004)
Article DOI: 10.1021/jm0304313
BindingDB Entry DOI: 10.7270/Q2ZS2VXF |
More data for this
Ligand-Target Pair | |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50278837
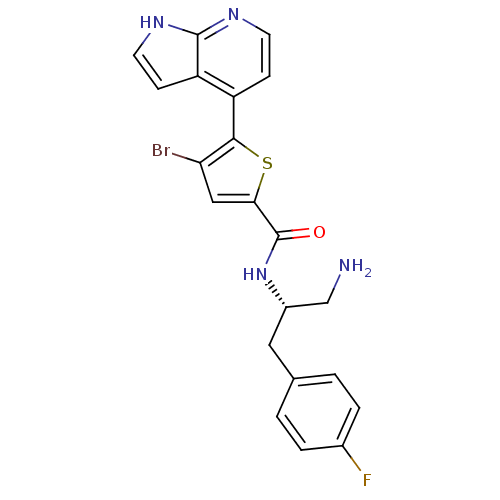
(CHEMBL496690 | N-((S)-1-amino-3-(4-fluorophenyl)pr...)Show SMILES NC[C@H](Cc1ccc(F)cc1)NC(=O)c1cc(Br)c(s1)-c1ccnc2[nH]ccc12 |r| Show InChI InChI=1S/C21H18BrFN4OS/c22-17-10-18(29-19(17)15-5-7-25-20-16(15)6-8-26-20)21(28)27-14(11-24)9-12-1-3-13(23)4-2-12/h1-8,10,14H,9,11,24H2,(H,25,26)(H,27,28)/t14-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of AKT1 |
Bioorg Med Chem Lett 19: 2244-8 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.094
BindingDB Entry DOI: 10.7270/Q24X57PX |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50073254
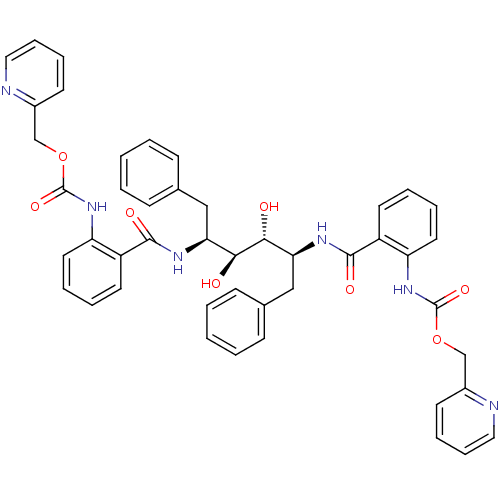
(Anthranilamide derivative | CHEMBL408110)Show SMILES O[C@@H]([C@H](Cc1ccccc1)NC(=O)c1ccccc1NC(=O)OCc1ccccn1)[C@H](O)[C@H](Cc1ccccc1)NC(=O)c1ccccc1NC(=O)OCc1ccccn1 Show InChI InChI=1S/C46H44N6O8/c53-41(39(27-31-15-3-1-4-16-31)49-43(55)35-21-7-9-23-37(35)51-45(57)59-29-33-19-11-13-25-47-33)42(54)40(28-32-17-5-2-6-18-32)50-44(56)36-22-8-10-24-38(36)52-46(58)60-30-34-20-12-14-26-48-34/h1-26,39-42,53-54H,27-30H2,(H,49,55)(H,50,56)(H,51,57)(H,52,58)/t39-,40-,41-,42+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Tested for inhibition of HIV protease using fluorogenic substrate |
Bioorg Med Chem Lett 5: 2557-2562 (1995)
Article DOI: 10.1016/0960-894X(95)00449-4
BindingDB Entry DOI: 10.7270/Q2RB74KT |
More data for this
Ligand-Target Pair | |
Melatonin receptor type 1A
(Homo sapiens (Human)) | BDBM50347588
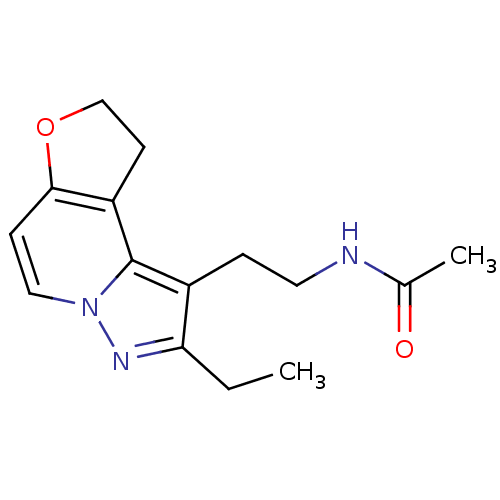
(CHEMBL1802025)Show InChI InChI=1S/C15H19N3O2/c1-3-13-11(4-7-16-10(2)19)15-12-6-9-20-14(12)5-8-18(15)17-13/h5,8H,3-4,6-7,9H2,1-2H3,(H,16,19) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited
Curated by ChEMBL
| Assay Description
Displacement of 2-[125I]iodomelatonin from human MT1 receptor expressed in CHO cells |
J Med Chem 54: 4207-18 (2011)
Article DOI: 10.1021/jm200385u
BindingDB Entry DOI: 10.7270/Q2HT2PN3 |
More data for this
Ligand-Target Pair | |
Cathepsin L2
(Homo sapiens (Human)) | BDBM19778

((2S)-2-(1-benzofuran-2-ylformamido)-4-methyl-N-[(4...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2ccccc2o1)C(=O)N[C@H]1CC[C@@H](C)N(CC1=O)S(=O)(=O)c1ccccn1 |r| Show InChI InChI=1S/C27H32N4O6S/c1-17(2)14-21(30-27(34)24-15-19-8-4-5-9-23(19)37-24)26(33)29-20-12-11-18(3)31(16-22(20)32)38(35,36)25-10-6-7-13-28-25/h4-10,13,15,17-18,20-21H,11-12,14,16H2,1-3H3,(H,29,33)(H,30,34)/t18-,20+,21+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0630 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK
| Assay Description
Potential inhibitors were evaluated using the progress curve method. Assays were carried out in the presence of variable concentrations of test compo... |
J Med Chem 49: 1597-612 (2006)
Article DOI: 10.1021/jm050915u
BindingDB Entry DOI: 10.7270/Q2SQ8XP5 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(RAT) | BDBM50021922

(CHEMBL3298596)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ncsc1-c1ccccc1 Show InChI InChI=1S/C18H21N3O2S.HI/c1-21-10-7-18(8-11-21,9-12-21)23-17(22)20-16-15(24-13-19-16)14-5-3-2-4-6-14;/h2-6,13H,7-12H2,1H3;1H | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0640 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M3 receptor in submandibular gland |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Procathepsin L
(Homo sapiens (Human)) | BDBM19778

((2S)-2-(1-benzofuran-2-ylformamido)-4-methyl-N-[(4...)Show SMILES CC(C)C[C@H](NC(=O)c1cc2ccccc2o1)C(=O)N[C@H]1CC[C@@H](C)N(CC1=O)S(=O)(=O)c1ccccn1 |r| Show InChI InChI=1S/C27H32N4O6S/c1-17(2)14-21(30-27(34)24-15-19-8-4-5-9-23(19)37-24)26(33)29-20-12-11-18(3)31(16-22(20)32)38(35,36)25-10-6-7-13-28-25/h4-10,13,15,17-18,20-21H,11-12,14,16H2,1-3H3,(H,29,33)(H,30,34)/t18-,20+,21+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0680 | -57.4 | n/a | n/a | n/a | n/a | n/a | 5.5 | 22 |
GSK
| Assay Description
Potential inhibitors were evaluated using the progress curve method. Assays were carried out in the presence of variable concentrations of test compo... |
J Med Chem 49: 1597-612 (2006)
Article DOI: 10.1021/jm050915u
BindingDB Entry DOI: 10.7270/Q2SQ8XP5 |
More data for this
Ligand-Target Pair | |
Alpha-2C adrenergic receptor
(Homo sapiens (Human)) | BDBM81811
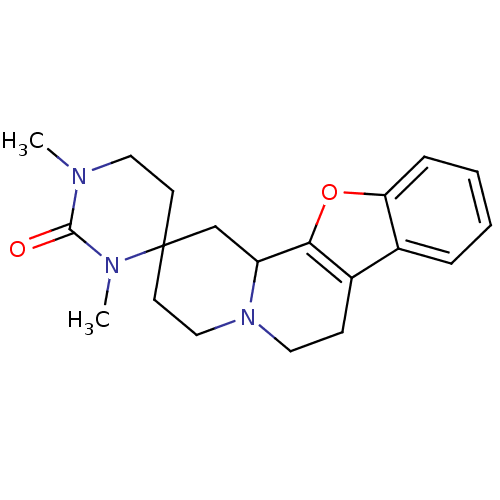
(CAS_123679 | L-657,743 | MK-912 | NSC_123679)Show InChI InChI=1S/C20H25N3O2/c1-21-11-8-20(22(2)19(21)24)9-12-23-10-7-15-14-5-3-4-6-17(14)25-18(15)16(23)13-20/h3-6,16H,7-13H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Bioscience
Curated by PDSP Ki Database
| |
Biochem Pharmacol 55: 1035-43 (1998)
Article DOI: 10.1016/s0006-2952(97)00631-x
BindingDB Entry DOI: 10.7270/Q2T72G01 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50285701
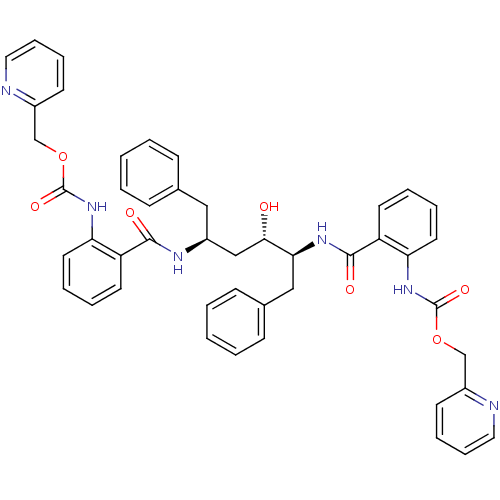
(Anthranilamide derivative | CHEMBL313767)Show SMILES O[C@@H](C[C@H](Cc1ccccc1)NC(=O)c1ccccc1NC(=O)OCc1ccccn1)[C@H](Cc1ccccc1)NC(=O)c1ccccc1NC(=O)OCc1ccccn1 Show InChI InChI=1S/C46H44N6O7/c53-42(41(28-33-17-5-2-6-18-33)50-44(55)38-22-8-10-24-40(38)52-46(57)59-31-35-20-12-14-26-48-35)29-36(27-32-15-3-1-4-16-32)49-43(54)37-21-7-9-23-39(37)51-45(56)58-30-34-19-11-13-25-47-34/h1-26,36,41-42,53H,27-31H2,(H,49,54)(H,50,55)(H,51,56)(H,52,57)/t36-,41-,42-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Tested for inhibition of HIV protease using fluorogenic substrate |
Bioorg Med Chem Lett 5: 2557-2562 (1995)
Article DOI: 10.1016/0960-894X(95)00449-4
BindingDB Entry DOI: 10.7270/Q2RB74KT |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021935
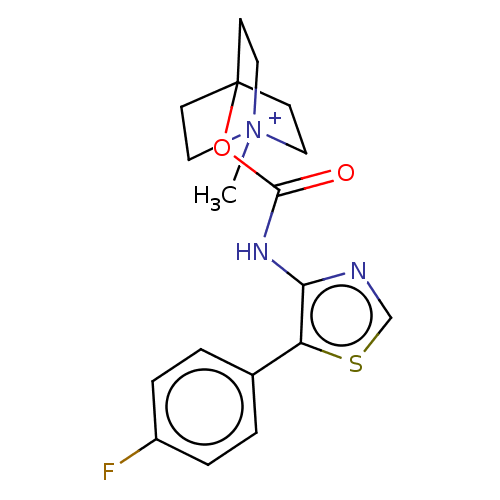
(CHEMBL3298600)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ncsc1-c1ccc(F)cc1 Show InChI InChI=1S/C18H20FN3O2S.HI/c1-22-9-6-18(7-10-22,8-11-22)24-17(23)21-16-15(25-12-20-16)13-2-4-14(19)5-3-13;/h2-5,12H,6-11H2,1H3;1H | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0730 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Translocator protein
(Rattus norvegicus (rat)) | BDBM50122294
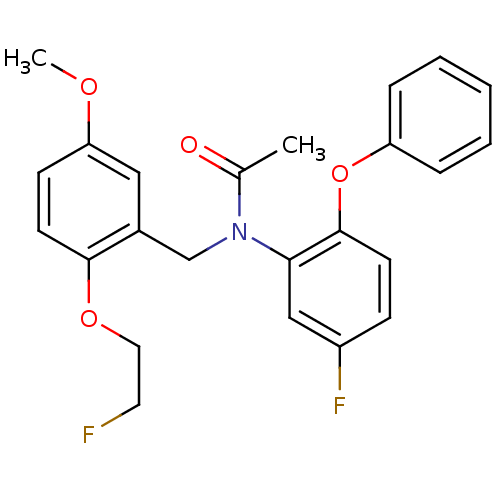
(CHEMBL292092 | N-(5-fluoro-2-phenoxyphenyl)-N-(2-(...)Show SMILES COc1ccc(OCCF)c(CN(C(C)=O)c2cc(F)ccc2Oc2ccccc2)c1 Show InChI InChI=1S/C24H23F2NO4/c1-17(28)27(16-18-14-21(29-2)9-11-23(18)30-13-12-25)22-15-19(26)8-10-24(22)31-20-6-4-3-5-7-20/h3-11,14-15H,12-13,16H2,1-2H3 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Radiological Sciences
Curated by ChEMBL
| Assay Description
In vitro inhibition of [11C]2 binding to Peripheral benzodiazepine receptor (PBR) in rat brain |
J Med Chem 47: 2228-35 (2004)
Article DOI: 10.1021/jm0304919
BindingDB Entry DOI: 10.7270/Q20G3KXQ |
More data for this
Ligand-Target Pair | |
Melatonin receptor type 1A
(Homo sapiens (Human)) | BDBM50347585
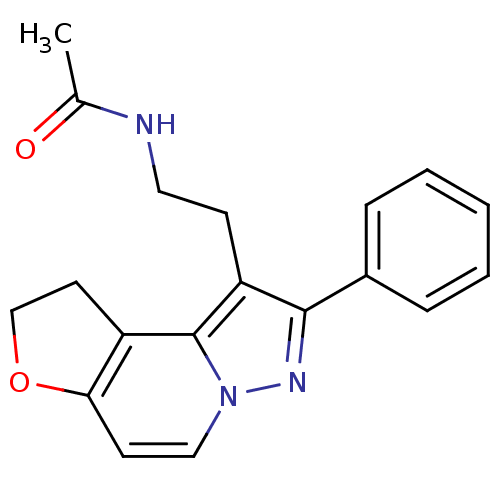
(CHEMBL1802022)Show InChI InChI=1S/C19H19N3O2/c1-13(23)20-10-7-16-18(14-5-3-2-4-6-14)21-22-11-8-17-15(19(16)22)9-12-24-17/h2-6,8,11H,7,9-10,12H2,1H3,(H,20,23) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0820 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited
Curated by ChEMBL
| Assay Description
Displacement of 2-[125I]iodomelatonin from human MT1 receptor expressed in CHO cells |
J Med Chem 54: 4207-18 (2011)
Article DOI: 10.1021/jm200385u
BindingDB Entry DOI: 10.7270/Q2HT2PN3 |
More data for this
Ligand-Target Pair | |
Melatonin receptor type 1A
(Homo sapiens (Human)) | BDBM50347587
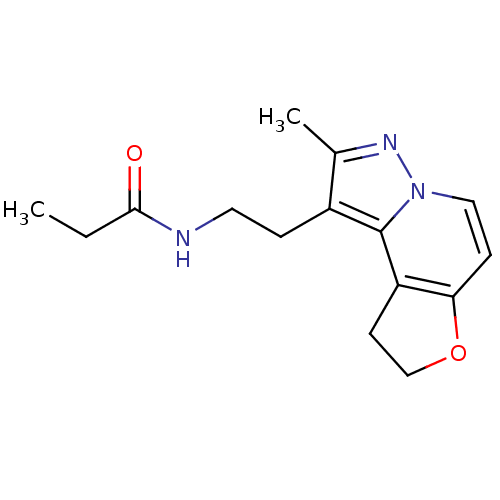
(CHEMBL1802024)Show InChI InChI=1S/C15H19N3O2/c1-3-14(19)16-7-4-11-10(2)17-18-8-5-13-12(15(11)18)6-9-20-13/h5,8H,3-4,6-7,9H2,1-2H3,(H,16,19) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0830 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited
Curated by ChEMBL
| Assay Description
Displacement of 2-[125I]iodomelatonin from human MT1 receptor expressed in CHO cells |
J Med Chem 54: 4207-18 (2011)
Article DOI: 10.1021/jm200385u
BindingDB Entry DOI: 10.7270/Q2HT2PN3 |
More data for this
Ligand-Target Pair | |
Melatonin receptor type 1B
(Homo sapiens (Human)) | BDBM50347585

(CHEMBL1802022)Show InChI InChI=1S/C19H19N3O2/c1-13(23)20-10-7-16-18(14-5-3-2-4-6-14)21-22-11-8-17-15(19(16)22)9-12-24-17/h2-6,8,11H,7,9-10,12H2,1H3,(H,20,23) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Limited
Curated by ChEMBL
| Assay Description
Displacement of 2-[125I]iodomelatonin from human MT2 receptor expressed in CHO cells |
J Med Chem 54: 4207-18 (2011)
Article DOI: 10.1021/jm200385u
BindingDB Entry DOI: 10.7270/Q2HT2PN3 |
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50255850
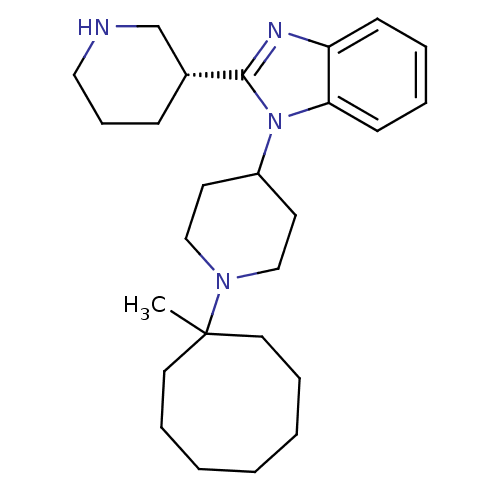
(1-[1-(1-Methylcyclooctyl)-4-piperidinyl]-2-[(3R)-3...)Show SMILES CC1(CCCCCCC1)N1CCC(CC1)n1c(nc2ccccc12)[C@@H]1CCCNC1 |r| Show InChI InChI=1S/C26H40N4/c1-26(15-7-3-2-4-8-16-26)29-18-13-22(14-19-29)30-24-12-6-5-11-23(24)28-25(30)21-10-9-17-27-20-21/h5-6,11-12,21-22,27H,2-4,7-10,13-20H2,1H3/t21-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0858 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]N/OFQ from human NOP receptor expressed in HEK293 cells |
J Med Chem 52: 610-25 (2009)
Article DOI: 10.1021/jm7012979
BindingDB Entry DOI: 10.7270/Q29K4C5K |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021904

(CHEMBL3298588)Show InChI InChI=1S/C18H20N2O2S.ClH/c21-17(22-18-7-10-20(11-8-18)12-9-18)19-15-6-13-23-16(15)14-4-2-1-3-5-14;/h1-6,13H,7-12H2,(H,19,21);1H | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0950 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50143729

((2R,3R,4R,5R)-3,4,5-Trihydroxy-1-(4-phenoxy-benzen...)Show SMILES ONC(=O)[C@H]1[C@@H](O)[C@H](O)[C@H](O)CN1S(=O)(=O)c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C18H20N2O8S/c21-14-10-20(15(18(24)19-25)17(23)16(14)22)29(26,27)13-8-6-12(7-9-13)28-11-4-2-1-3-5-11/h1-9,14-17,21-23,25H,10H2,(H,19,24)/t14-,15-,16-,17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0970 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokkaido Collaboration Center N-21
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant matrix metalloprotease 9 |
J Med Chem 47: 1930-8 (2004)
Article DOI: 10.1021/jm0304313
BindingDB Entry DOI: 10.7270/Q2ZS2VXF |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Rattus norvegicus (rat)) | BDBM194790

(US9206167, 11 | US9206167, 12 | USRE48059, Compoun...)Show SMILES O=C1CCc2ccc(OCCCCN3CCN(CC3)c3cccc4sccc34)cc2N1 Show InChI InChI=1S/C25H29N3O2S/c29-25-9-7-19-6-8-20(18-22(19)26-25)30-16-2-1-11-27-12-14-28(15-13-27)23-4-3-5-24-21(23)10-17-31-24/h3-6,8,10,17-18H,1-2,7,9,11-16H2,(H,26,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.100 | -57.1 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
OTSUKA PHARMACEUTICAL CO., LTD.
US Patent
| Assay Description
The assay was performed according to the method by Kohler et al. (Kohler C, Hall H, Ogren S O and Gawell L, Specific in vitro and in vivo binding of ... |
US Patent US9206167 (2015)
BindingDB Entry DOI: 10.7270/Q2XD10GS |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Rattus norvegicus (rat)) | BDBM194790

(US9206167, 11 | US9206167, 12 | USRE48059, Compoun...)Show SMILES O=C1CCc2ccc(OCCCCN3CCN(CC3)c3cccc4sccc34)cc2N1 Show InChI InChI=1S/C25H29N3O2S/c29-25-9-7-19-6-8-20(18-22(19)26-25)30-16-2-1-11-27-12-14-28(15-13-27)23-4-3-5-24-21(23)10-17-31-24/h3-6,8,10,17-18H,1-2,7,9,11-16H2,(H,26,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.100 | -57.1 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
OTSUKA PHARMACEUTICAL CO., LTD.
US Patent
| Assay Description
The assay was performed according to the method by Kohler et al. (Kohler C, Hall H, Ogren S O and Gawell L, Specific in vitro and in vivo binding of ... |
US Patent US9206167 (2015)
BindingDB Entry DOI: 10.7270/Q2XD10GS |
More data for this
Ligand-Target Pair | |
Dimer of Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM4227
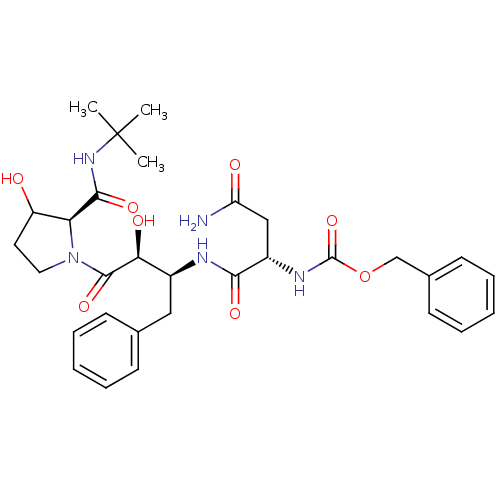
(AHPBA 35a | Z-Asn.(2S,3S)-AHPBA-[3(R)-hydroxy]Pro ...)Show SMILES CC(C)(C)NC(=O)[C@@H]1C(O)CCN1C(=O)[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)OCc1ccccc1 |r| Show InChI InChI=1S/C31H41N5O8/c1-31(2,3)35-28(41)25-23(37)14-15-36(25)29(42)26(39)21(16-19-10-6-4-7-11-19)33-27(40)22(17-24(32)38)34-30(43)44-18-20-12-8-5-9-13-20/h4-13,21-23,25-26,37,39H,14-18H2,1-3H3,(H2,32,38)(H,33,40)(H,34,43)(H,35,41)/t21-,22-,23?,25-,26-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | -59.4 | n/a | n/a | n/a | n/a | n/a | 4.7 | 37 |
Sankyo Co. Ltd.
| Assay Description
The inhibitory activities of the compounds toward HIV-1 PR were determined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) as... |
Bioorg Med Chem 4: 1365-77 (1996)
Article DOI: 10.1016/0968-0896(96)00130-7
BindingDB Entry DOI: 10.7270/Q2V9868K |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(RAT) | BDBM50021935

(CHEMBL3298600)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ncsc1-c1ccc(F)cc1 Show InChI InChI=1S/C18H20FN3O2S.HI/c1-22-9-6-18(7-10-22,8-11-22)24-17(23)21-16-15(25-12-20-16)13-2-4-14(19)5-3-13;/h2-5,12H,6-11H2,1H3;1H | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M3 receptor in submandibular gland |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ITK/TSK
(Homo sapiens (Human)) | BDBM50015266
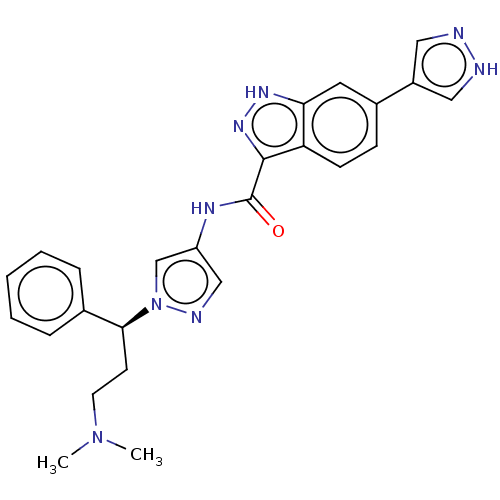
(CHEMBL3263053)Show SMILES CN(C)CC[C@@H](c1ccccc1)n1cc(NC(=O)c2n[nH]c3cc(ccc23)-c2cn[nH]c2)cn1 |r| Show InChI InChI=1S/C25H26N8O/c1-32(2)11-10-23(17-6-4-3-5-7-17)33-16-20(15-28-33)29-25(34)24-21-9-8-18(12-22(21)30-31-24)19-13-26-27-14-19/h3-9,12-16,23H,10-11H2,1-2H3,(H,26,27)(H,29,34)(H,30,31)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full-length ITK (unknown origin) using biotin-EQEDEPEGIYGVLF-NH2 as substrate by plate reader analysis |
Bioorg Med Chem Lett 24: 2448-52 (2014)
Article DOI: 10.1016/j.bmcl.2014.04.023
BindingDB Entry DOI: 10.7270/Q2W097G8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50278099
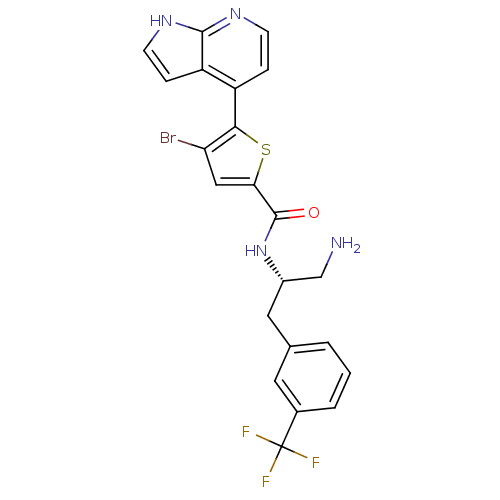
(CHEMBL482536 | N-((S)-1-amino-3-(3-(trifluoromethy...)Show SMILES NC[C@H](Cc1cccc(c1)C(F)(F)F)NC(=O)c1cc(Br)c(s1)-c1ccnc2[nH]ccc12 |r| Show InChI InChI=1S/C22H18BrF3N4OS/c23-17-10-18(32-19(17)15-4-6-28-20-16(15)5-7-29-20)21(31)30-14(11-27)9-12-2-1-3-13(8-12)22(24,25)26/h1-8,10,14H,9,11,27H2,(H,28,29)(H,30,31)/t14-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of AKT1 |
Bioorg Med Chem Lett 19: 2244-8 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.094
BindingDB Entry DOI: 10.7270/Q24X57PX |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase JAK2
(Homo sapiens (Human)) | BDBM50355501
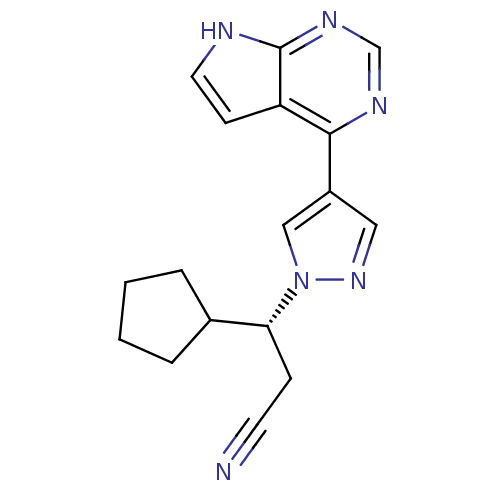
(INCB-018424 | RUXOLITINIB | RUXOLITINIB PHOSPHATE ...)Show SMILES N#CC[C@H](C1CCCC1)n1cc(cn1)-c1ncnc2[nH]ccc12 |r| Show InChI InChI=1S/C17H18N6/c18-7-5-15(12-3-1-2-4-12)23-10-13(9-22-23)16-14-6-8-19-17(14)21-11-20-16/h6,8-12,15H,1-5H2,(H,19,20,21)/t15-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of human JAK2 (828-1132) expressed in baculovirus-infected Sf9 cells using EQEDEPEGDYFEWLE as substrate after 1 hr by HTRF assay |
J Med Chem 56: 4521-36 (2013)
Article DOI: 10.1021/jm400266t
BindingDB Entry DOI: 10.7270/Q2VX0HX0 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase JAK2
(Homo sapiens (Human)) | BDBM50355501

(INCB-018424 | RUXOLITINIB | RUXOLITINIB PHOSPHATE ...)Show SMILES N#CC[C@H](C1CCCC1)n1cc(cn1)-c1ncnc2[nH]ccc12 |r| Show InChI InChI=1S/C17H18N6/c18-7-5-15(12-3-1-2-4-12)23-10-13(9-22-23)16-14-6-8-19-17(14)21-11-20-16/h6,8-12,15H,1-5H2,(H,19,20,21)/t15-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of JAK2 kinase domain using N-terminal 5-carboxyfluorescein-tagged Val-Ala-Leu-Val-Asp-Gly-Tyr-Phe-Arg-Leu-Thr-Thr as substrate after 30 m... |
J Med Chem 55: 6176-93 (2012)
Article DOI: 10.1021/jm300628c
BindingDB Entry DOI: 10.7270/Q25Q4X6S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50255899

(1-[1-(1-Methylcyclooctyl)-4-piperidinyl]-2-[(3S)-3...)Show SMILES CC1(CCCCCCC1)N1CCC(CC1)n1c(nc2ccccc12)[C@H]1CCCNC1 |r| Show InChI InChI=1S/C26H40N4/c1-26(15-7-3-2-4-8-16-26)29-18-13-22(14-19-29)30-24-12-6-5-11-23(24)28-25(30)21-10-9-17-27-20-21/h5-6,11-12,21-22,27H,2-4,7-10,13-20H2,1H3/t21-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]N/OFQ from human NOP receptor expressed in HEK293 cells |
J Med Chem 52: 610-25 (2009)
Article DOI: 10.1021/jm7012979
BindingDB Entry DOI: 10.7270/Q29K4C5K |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data