| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor X |
|---|
| Ligand | BDBM50096098 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_48791 (CHEMBL662444) |
|---|
| Ki | 0.01±n/a nM |
|---|
| Citation |  Pinto, DJ; Orwat, MJ; Wang, S; Fevig, JM; Quan, ML; Amparo, E; Cacciola, J; Rossi, KA; Alexander, RS; Smallwood, AM; Luettgen, JM; Liang, L; Aungst, BJ; Wright, MR; Knabb, RM; Wong, PC; Wexler, RR; Lam, PY Discovery of 1-[3-(aminomethyl)phenyl]-N-3-fluoro-2'-(methylsulfonyl)-[1,1'-biphenyl]-4-yl]-3-(trifluoromethyl)-1H-pyrazole-5-carboxamide (DPC423), a highly potent, selective, and orally bioavailable inhibitor of blood coagulation factor Xa. J Med Chem44:566-78 (2001) [PubMed] Pinto, DJ; Orwat, MJ; Wang, S; Fevig, JM; Quan, ML; Amparo, E; Cacciola, J; Rossi, KA; Alexander, RS; Smallwood, AM; Luettgen, JM; Liang, L; Aungst, BJ; Wright, MR; Knabb, RM; Wong, PC; Wexler, RR; Lam, PY Discovery of 1-[3-(aminomethyl)phenyl]-N-3-fluoro-2'-(methylsulfonyl)-[1,1'-biphenyl]-4-yl]-3-(trifluoromethyl)-1H-pyrazole-5-carboxamide (DPC423), a highly potent, selective, and orally bioavailable inhibitor of blood coagulation factor Xa. J Med Chem44:566-78 (2001) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor X |
|---|
| Name: | Coagulation factor X |
|---|
| Synonyms: | Activated coagulation factor X (FXa) | Activated factor Xa heavy chain | Coagulation factor X precursor | Coagulation factor Xa | F10 | FA10_HUMAN | Factor X heavy chain | Factor X light chain | Factor Xa | Stuart factor | Stuart-Prower factor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 54726.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 488 |
|---|
| Sequence: | MGRPLHLVLLSASLAGLLLLGESLFIRREQANNILARVTRANSFLEEMKKGHLERECMEE
TCSYEEAREVFEDSDKTNEFWNKYKDGDQCETSPCQNQGKCKDGLGEYTCTCLEGFEGKN
CELFTRKLCSLDNGDCDQFCHEEQNSVVCSCARGYTLADNGKACIPTGPYPCGKQTLERR
KRSVAQATSSSGEAPDSITWKPYDAADLDPTENPFDLLDFNQTQPERGDNNLTRIVGGQE
CKDGECPWQALLINEENEGFCGGTILSEFYILTAAHCLYQAKRFKVRVGDRNTEQEEGGE
AVHEVEVVIKHNRFTKETYDFDIAVLRLKTPITFRMNVAPACLPERDWAESTLMTQKTGI
VSGFGRTHEKGRQSTRLKMLEVPYVDRNSCKLSSSFIITQNMFCAGYDTKQEDACQGDSG
GPHVTRFKDTYFVTGIVSWGEGCARKGKYGIYTKVTAFLKWIDRSMKTRGLPKAKSHAPE
VITSSPLK
|
|
|
|---|
| BDBM50096098 |
|---|
| n/a |
|---|
| Name | BDBM50096098 |
|---|
| Synonyms: | 2-(3-Carbamimidoyl-phenyl)-5-methyl-2H-pyrazole-3-carboxylic acid (3-bromo-2'-sulfamoyl-biphenyl-4-yl)-amide | CHEMBL152859 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H21BrN6O3S |
|---|
| Mol. Mass. | 553.431 |
|---|
| SMILES | Cc1cc(C(=O)Nc2ccc(cc2Br)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N |
|---|
| Structure | 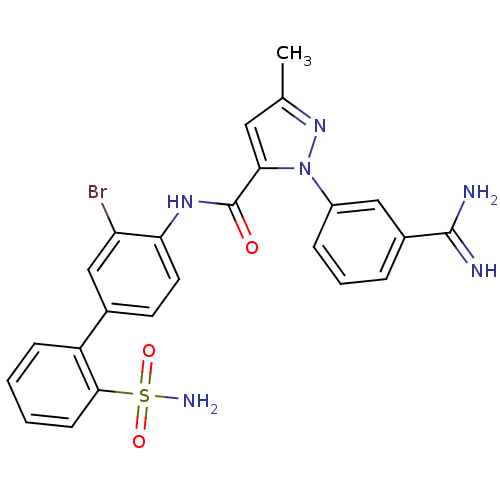
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Pinto, DJ; Orwat, MJ; Wang, S; Fevig, JM; Quan, ML; Amparo, E; Cacciola, J; Rossi, KA; Alexander, RS; Smallwood, AM; Luettgen, JM; Liang, L; Aungst, BJ; Wright, MR; Knabb, RM; Wong, PC; Wexler, RR; Lam, PY Discovery of 1-[3-(aminomethyl)phenyl]-N-3-fluoro-2'-(methylsulfonyl)-[1,1'-biphenyl]-4-yl]-3-(trifluoromethyl)-1H-pyrazole-5-carboxamide (DPC423), a highly potent, selective, and orally bioavailable inhibitor of blood coagulation factor Xa. J Med Chem44:566-78 (2001) [PubMed]
Pinto, DJ; Orwat, MJ; Wang, S; Fevig, JM; Quan, ML; Amparo, E; Cacciola, J; Rossi, KA; Alexander, RS; Smallwood, AM; Luettgen, JM; Liang, L; Aungst, BJ; Wright, MR; Knabb, RM; Wong, PC; Wexler, RR; Lam, PY Discovery of 1-[3-(aminomethyl)phenyl]-N-3-fluoro-2'-(methylsulfonyl)-[1,1'-biphenyl]-4-yl]-3-(trifluoromethyl)-1H-pyrazole-5-carboxamide (DPC423), a highly potent, selective, and orally bioavailable inhibitor of blood coagulation factor Xa. J Med Chem44:566-78 (2001) [PubMed]