| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Chondroitin sulfate N-acetylgalactosaminyltransferase 1 |
|---|
| Ligand | BDBM50386148 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_824644 (CHEMBL2045702) |
|---|
| IC50 | 69000±n/a nM |
|---|
| Citation |  Jadhav, RD; Kadam, KS; Kandre, S; Guha, T; Reddy, MM; Brahma, MK; Deshmukh, NJ; Dixit, A; Doshi, L; Potdar, N; Enose, AA; Vishwakarma, RA; Sivaramakrishnan, H; Srinivasan, S; Nemmani, KV; Gupte, A; Gangopadhyay, AK; Sharma, R Synthesis and biological evaluation of isoxazole, oxazole, and oxadiazole containing heteroaryl analogs of biaryl ureas as DGAT1 inhibitors. Eur J Med Chem54:324-42 (2012) [PubMed] Article Jadhav, RD; Kadam, KS; Kandre, S; Guha, T; Reddy, MM; Brahma, MK; Deshmukh, NJ; Dixit, A; Doshi, L; Potdar, N; Enose, AA; Vishwakarma, RA; Sivaramakrishnan, H; Srinivasan, S; Nemmani, KV; Gupte, A; Gangopadhyay, AK; Sharma, R Synthesis and biological evaluation of isoxazole, oxazole, and oxadiazole containing heteroaryl analogs of biaryl ureas as DGAT1 inhibitors. Eur J Med Chem54:324-42 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Chondroitin sulfate N-acetylgalactosaminyltransferase 1 |
|---|
| Name: | Chondroitin sulfate N-acetylgalactosaminyltransferase 1 |
|---|
| Synonyms: | Beta4GalNAcT-1 | CGAT1_HUMAN | CHGN | CSGALNACT1 | Chondroitin beta-1,4-N-acetylgalactosaminyltransferase 1 | CsGalNAcT-1 | GALNACT1 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 61307.78 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_824644 |
|---|
| Residue: | 532 |
|---|
| Sequence: | MMMVRRGLLAWISRVVVLLVLLCCAISVLYMLACTPKGDEEQLALPRANSPTGKEGYQAV
LQEWEEQHRNYVSSLKRQIAQLKEELQERSEQLRNGQYQASDAAGLGLDRSPPEKTQADL
LAFLHSQVDKAEVNAGVKLATEYAAVPFDSFTLQKVYQLETGLTRHPEEKPVRKDKRDEL
VEAIESALETLNSPAENSPNHRPYTASDFIEGIYRTERDKGTLYELTFKGDHKHEFKRLI
LFRPFGPIMKVKNEKLNMANTLINVIVPLAKRVDKFRQFMQNFREMCIEQDGRVHLTVVY
FGKEEINEVKGILENTSKAANFRNFTFIQLNGEFSRGKGLDVGARFWKGSNVLLFFCDVD
IYFTSEFLNTCRLNTQPGKKVFYPVLFSQYNPGIIYGHHDAVPPLEQQLVIKKETGFWRD
FGFGMTCQYRSDFINIGGFDLDIKGWGGEDVHLYRKYLHSNLIVVRTPVRGLFHLWHEKR
CMDELTPEQYKMCMQSKAMNEASHGQLGMLVFRHEIEAHLRKQKQKTSSKKT
|
|
|
|---|
| BDBM50386148 |
|---|
| n/a |
|---|
| Name | BDBM50386148 |
|---|
| Synonyms: | CHEMBL2042350 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H23FN4O5 |
|---|
| Mol. Mass. | 454.4509 |
|---|
| SMILES | CC(C)[C@H](NC(=O)c1cc(no1)-c1ccc(NC(=O)Nc2cc(F)ccc2C)cc1)C(O)=O |r| |
|---|
| Structure | 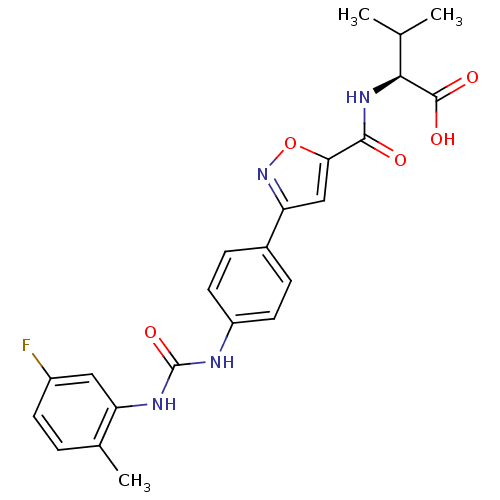
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Jadhav, RD; Kadam, KS; Kandre, S; Guha, T; Reddy, MM; Brahma, MK; Deshmukh, NJ; Dixit, A; Doshi, L; Potdar, N; Enose, AA; Vishwakarma, RA; Sivaramakrishnan, H; Srinivasan, S; Nemmani, KV; Gupte, A; Gangopadhyay, AK; Sharma, R Synthesis and biological evaluation of isoxazole, oxazole, and oxadiazole containing heteroaryl analogs of biaryl ureas as DGAT1 inhibitors. Eur J Med Chem54:324-42 (2012) [PubMed] Article
Jadhav, RD; Kadam, KS; Kandre, S; Guha, T; Reddy, MM; Brahma, MK; Deshmukh, NJ; Dixit, A; Doshi, L; Potdar, N; Enose, AA; Vishwakarma, RA; Sivaramakrishnan, H; Srinivasan, S; Nemmani, KV; Gupte, A; Gangopadhyay, AK; Sharma, R Synthesis and biological evaluation of isoxazole, oxazole, and oxadiazole containing heteroaryl analogs of biaryl ureas as DGAT1 inhibitors. Eur J Med Chem54:324-42 (2012) [PubMed] Article