| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Beta-secretase 1 |
|---|
| Ligand | BDBM50007801 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1335844 (CHEMBL3238628) |
|---|
| IC50 | 80±n/a nM |
|---|
| Citation |  Viayna, E; Sola, I; Bartolini, M; De Simone, A; Tapia-Rojas, C; Serrano, FG; Sabat�, R; Ju�rez-Jim�nez, J; P�rez, B; Luque, FJ; Andrisano, V; Clos, MV; Inestrosa, NC; Mu�oz-Torrero, D Synthesis and multitarget biological profiling of a novel family of rhein derivatives as disease-modifying anti-Alzheimer agents. J Med Chem57:2549-67 (2014) [PubMed] Article Viayna, E; Sola, I; Bartolini, M; De Simone, A; Tapia-Rojas, C; Serrano, FG; Sabat�, R; Ju�rez-Jim�nez, J; P�rez, B; Luque, FJ; Andrisano, V; Clos, MV; Inestrosa, NC; Mu�oz-Torrero, D Synthesis and multitarget biological profiling of a novel family of rhein derivatives as disease-modifying anti-Alzheimer agents. J Med Chem57:2549-67 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Beta-secretase 1 |
|---|
| Name: | Beta-secretase 1 |
|---|
| Synonyms: | ASP2 | Asp 2 | Aspartyl protease 2 | BACE | BACE1 | BACE1_HUMAN | Beta-secretase (BACE) | Beta-secretase 1 | Beta-secretase 1 (BACE 1) | Beta-secretase 1 (BACE-1) | Beta-secretase 1 (BACE1) | Beta-site APP cleaving enzyme 1 | Beta-site amyloid precursor protein cleaving enzyme 1 | KIAA1149 | Memapsin-2 | Membrane-associated aspartic protease 2 | beta-Secretase (BACE-1) | beta-Secretase (BACE1) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55755.10 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P56817 |
|---|
| Residue: | 501 |
|---|
| Sequence: | MAQALPWLLLWMGAGVLPAHGTQHGIRLPLRSGLGGAPLGLRLPRETDEEPEEPGRRGSF
VEMVDNLRGKSGQGYYVEMTVGSPPQTLNILVDTGSSNFAVGAAPHPFLHRYYQRQLSST
YRDLRKGVYVPYTQGKWEGELGTDLVSIPHGPNVTVRANIAAITESDKFFINGSNWEGIL
GLAYAEIARPDDSLEPFFDSLVKQTHVPNLFSLQLCGAGFPLNQSEVLASVGGSMIIGGI
DHSLYTGSLWYTPIRREWYYEVIIVRVEINGQDLKMDCKEYNYDKSIVDSGTTNLRLPKK
VFEAAVKSIKAASSTEKFPDGFWLGEQLVCWQAGTTPWNIFPVISLYLMGEVTNQSFRIT
ILPQQYLRPVEDVATSQDDCYKFAISQSSTGTVMGAVIMEGFYVVFDRARKRIGFAVSAC
HVHDEFRTAAVEGPFVTLDMEDCGYNIPQTDESTLMTIAYVMAAICALFMLPLCLMVCQW
RCLRCLRQQHDDFADDISLLK
|
|
|
|---|
| BDBM50007801 |
|---|
| n/a |
|---|
| Name | BDBM50007801 |
|---|
| Synonyms: | CHEMBL3233832 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C41H43Cl2N3O5 |
|---|
| Mol. Mass. | 728.703 |
|---|
| SMILES | Cl.[H][C@]12Cc3nc4cc(Cl)ccc4c(NCCCCCCCCCNC(=O)c4cc(O)c5C(=O)c6c(O)cccc6C(=O)c5c4)c3[C@]([H])(CC(C)=C1)C2 |r,c:54| |
|---|
| Structure | 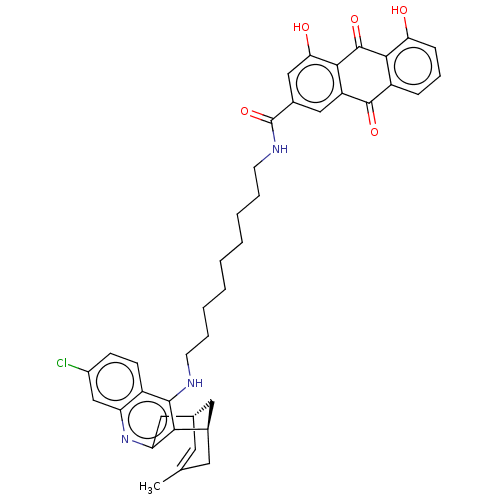
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Viayna, E; Sola, I; Bartolini, M; De Simone, A; Tapia-Rojas, C; Serrano, FG; Sabat�, R; Ju�rez-Jim�nez, J; P�rez, B; Luque, FJ; Andrisano, V; Clos, MV; Inestrosa, NC; Mu�oz-Torrero, D Synthesis and multitarget biological profiling of a novel family of rhein derivatives as disease-modifying anti-Alzheimer agents. J Med Chem57:2549-67 (2014) [PubMed] Article
Viayna, E; Sola, I; Bartolini, M; De Simone, A; Tapia-Rojas, C; Serrano, FG; Sabat�, R; Ju�rez-Jim�nez, J; P�rez, B; Luque, FJ; Andrisano, V; Clos, MV; Inestrosa, NC; Mu�oz-Torrero, D Synthesis and multitarget biological profiling of a novel family of rhein derivatives as disease-modifying anti-Alzheimer agents. J Med Chem57:2549-67 (2014) [PubMed] Article