 Found 1920 hits with Last Name = 'andrisano' and Initial = 'v'
Found 1920 hits with Last Name = 'andrisano' and Initial = 'v' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50271367
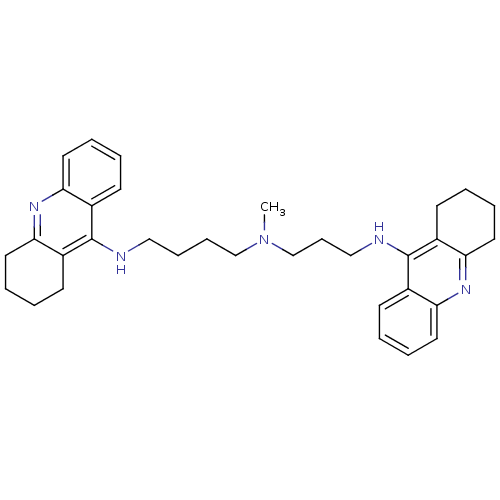
(CHEMBL489454 | N-Methyl-N-(1,2,3,4-tetrahydroacrid...)Show SMILES CN(CCCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H43N5/c1-39(24-12-22-36-34-27-15-4-8-19-31(27)38-32-20-9-5-16-28(32)34)23-11-10-21-35-33-25-13-2-6-17-29(25)37-30-18-7-3-14-26(30)33/h2,4,6,8,13,15,17,19H,3,5,7,9-12,14,16,18,20-24H2,1H3,(H,35,37)(H,36,38) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human acetylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50005193
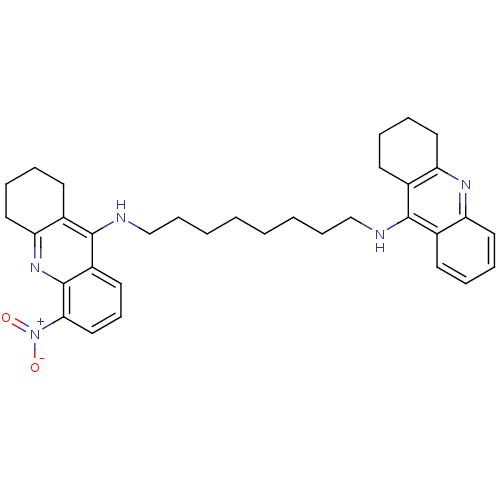
(CHEMBL3099496)Show SMILES [O-][N+](=O)c1cccc2c(NCCCCCCCCNc3c4CCCCc4nc4ccccc34)c3CCCCc3nc12 Show InChI InChI=1S/C34H41N5O2/c40-39(41)31-21-13-17-27-33(26-16-7-10-20-30(26)38-34(27)31)36-23-12-4-2-1-3-11-22-35-32-24-14-5-8-18-28(24)37-29-19-9-6-15-25(29)32/h5,8,13-14,17-18,21H,1-4,6-7,9-12,15-16,19-20,22-23H2,(H,35,37)(H,36,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human butyrylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50067482
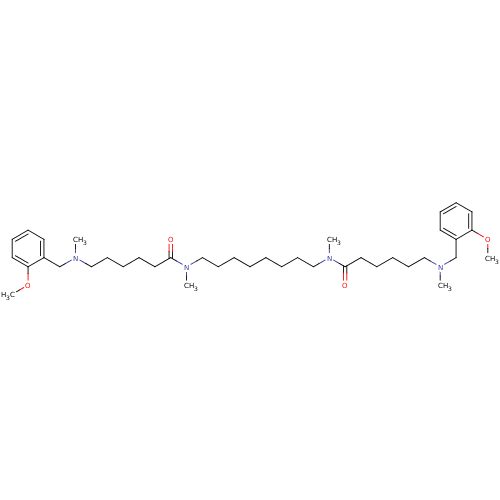
(6-[(2-Methoxy-benzyl)-methyl-amino]-hexanoic acid ...)Show SMILES COc1ccccc1CN(C)CCCCCC(=O)N(C)CCCCCCCCN(C)C(=O)CCCCCN(C)Cc1ccccc1OC Show InChI InChI=1S/C40H66N4O4/c1-41(33-35-23-15-17-25-37(35)47-5)29-19-11-13-27-39(45)43(3)31-21-9-7-8-10-22-32-44(4)40(46)28-14-12-20-30-42(2)34-36-24-16-18-26-38(36)48-6/h15-18,23-26H,7-14,19-22,27-34H2,1-6H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibition constant determined against Acetylcholinesterase (AChE) receptor. |
J Med Chem 41: 4186-9 (1998)
Checked by Author
Article DOI: 10.1021/jm9810452
BindingDB Entry DOI: 10.7270/Q2HH6KRM |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50560224
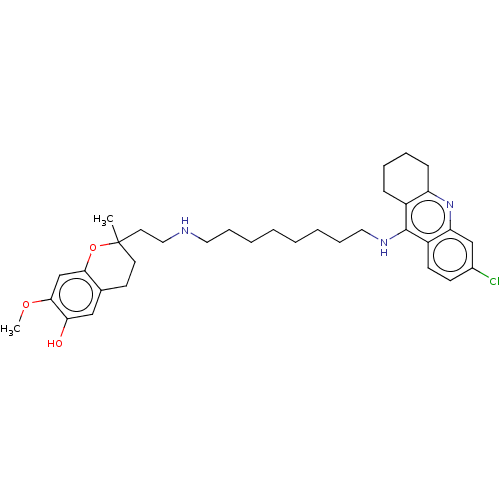
(CHEMBL4751100)Show SMILES COc1cc2OC(C)(CCNCCCCCCCCNc3c4CCCCc4nc4cc(Cl)ccc34)CCc2cc1O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.132 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Mixed inhibition of recombinant human AChE expressed in HEK293 cells assessed as dissociation constant for enzyme-inhibitor complex using varying lev... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00528
BindingDB Entry DOI: 10.7270/Q23N2733 |
More data for this
Ligand-Target Pair | |
Choline O-acetyltransferase
(RAT) | BDBM8961

(1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...)Show InChI InChI=1S/C13H14N2/c14-13-9-5-1-3-7-11(9)15-12-8-4-2-6-10(12)13/h1,3,5,7H,2,4,6,8H2,(H2,14,15) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibition constant determined against Acetylcholinesterase (AChE) receptor. |
J Med Chem 41: 4186-9 (1998)
Checked by Author
Article DOI: 10.1021/jm9810452
BindingDB Entry DOI: 10.7270/Q2HH6KRM |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50005192
(CHEMBL3099497)Show SMILES Nc1cccc2c(NCCCCCCCCNc3c4CCCCc4nc4ccccc34)c3CCCCc3nc12 Show InChI InChI=1S/C34H43N5/c35-28-18-13-17-27-33(26-16-7-10-21-31(26)39-34(27)28)37-23-12-4-2-1-3-11-22-36-32-24-14-5-8-19-29(24)38-30-20-9-6-15-25(30)32/h5,8,13-14,17-19H,1-4,6-7,9-12,15-16,20-23,35H2,(H,36,38)(H,37,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human acetylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50560224

(CHEMBL4751100)Show SMILES COc1cc2OC(C)(CCNCCCCCCCCNc3c4CCCCc4nc4cc(Cl)ccc34)CCc2cc1O | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.258 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Mixed inhibition of recombinant human AChE expressed in HEK293 cells assessed as dissociation constant for enzyme-substrate-inhibitor complex using v... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00528
BindingDB Entry DOI: 10.7270/Q23N2733 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50556952
(CHEMBL4754487)Show SMILES COc1cc(CNC(=O)CCCCCCNc2c3C4CC(Cc3nc3cc(Cl)ccc23)C=C(C)C4)ccc1O |t:34,TLB:23:22:19:34.31.32,THB:16:17:19:34.31.32| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 0.680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Mixed-type inhibition of recombinant human AChE assessed as inhibition constant using acetylthiocholine iodide as substrate by Cornish-Bowden plot an... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01775
BindingDB Entry DOI: 10.7270/Q2K077XJ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50138279
(CHEMBL3752467)Show SMILES C(CCCNCc1cc2ccccc2o1)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C29H35N3O/c1(2-10-18-30-21-23-20-22-12-4-9-17-28(22)33-23)3-11-19-31-29-24-13-5-7-15-26(24)32-27-16-8-6-14-25(27)29/h4-5,7,9,12-13,15,17,20,30H,1-3,6,8,10-11,14,16,18-19,21H2,(H,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.720 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Mixed-type inhibition of recombinant human AChE using acetylthiocholine iodide as substrate assessed as enzyme-inhibitor complex by Lineweaver-Burk d... |
J Med Chem 59: 114-31 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01119
BindingDB Entry DOI: 10.7270/Q2S75J5X |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50005193

(CHEMBL3099496)Show SMILES [O-][N+](=O)c1cccc2c(NCCCCCCCCNc3c4CCCCc4nc4ccccc34)c3CCCCc3nc12 Show InChI InChI=1S/C34H41N5O2/c40-39(41)31-21-13-17-27-33(26-16-7-10-20-30(26)38-34(27)31)36-23-12-4-2-1-3-11-22-35-32-24-14-5-8-18-28(24)37-29-19-9-6-15-25(29)32/h5,8,13-14,17-18,21H,1-4,6-7,9-12,15-16,19-20,22-23H2,(H,35,37)(H,36,38) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human acetylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50271367

(CHEMBL489454 | N-Methyl-N-(1,2,3,4-tetrahydroacrid...)Show SMILES CN(CCCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H43N5/c1-39(24-12-22-36-34-27-15-4-8-19-31(27)38-32-20-9-5-16-28(32)34)23-11-10-21-35-33-25-13-2-6-17-29(25)37-30-18-7-3-14-26(30)33/h2,4,6,8,13,15,17,19H,3,5,7,9-12,14,16,18,20-24H2,1H3,(H,35,37)(H,36,38) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.820 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human butyrylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50556952

(CHEMBL4754487)Show SMILES COc1cc(CNC(=O)CCCCCCNc2c3C4CC(Cc3nc3cc(Cl)ccc23)C=C(C)C4)ccc1O |t:34,TLB:23:22:19:34.31.32,THB:16:17:19:34.31.32| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 0.940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Mixed-type inhibition of recombinant human AChE assessed as dissociation constant for protein-substrate-compound complex using acetylthiocholine iodi... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c01775
BindingDB Entry DOI: 10.7270/Q2K077XJ |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50351015
(CHEMBL1819176)Show SMILES Clc1ccc2c(NCCCCCCCNc3c4Cc5ccccc5-c4nc4ccccc34)c3CCCCc3nc2c1 Show InChI InChI=1S/C36H37ClN4/c37-25-18-19-29-33(23-25)40-31-16-8-6-14-27(31)34(29)38-20-10-2-1-3-11-21-39-35-28-15-7-9-17-32(28)41-36-26-13-5-4-12-24(26)22-30(35)36/h4-5,7,9,12-13,15,17-19,23H,1-3,6,8,10-11,14,16,20-22H2,(H,38,40)(H,39,41) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Institut f£r Molekulare Physiologie
Curated by ChEMBL
| Assay Description
Mixed-type inhibition of human AChE assessed as hydrolysis of acetylthiocholine by Lineweaver-Burk plot analysis |
Eur J Med Chem 46: 4336-43 (2011)
Article DOI: 10.1016/j.ejmech.2011.07.004
BindingDB Entry DOI: 10.7270/Q2NP24TK |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50138279

(CHEMBL3752467)Show SMILES C(CCCNCc1cc2ccccc2o1)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C29H35N3O/c1(2-10-18-30-21-23-20-22-12-4-9-17-28(22)33-23)3-11-19-31-29-24-13-5-7-15-26(24)32-27-16-8-6-14-25(27)29/h4-5,7,9,12-13,15,17,20,30H,1-3,6,8,10-11,14,16,18-19,21H2,(H,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Mixed-type inhibition of recombinant human AChE using acetylthiocholine iodide as substrate assessed as enzyme-substrate-inhibitor complex by Linewea... |
J Med Chem 59: 114-31 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01119
BindingDB Entry DOI: 10.7270/Q2S75J5X |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50158582
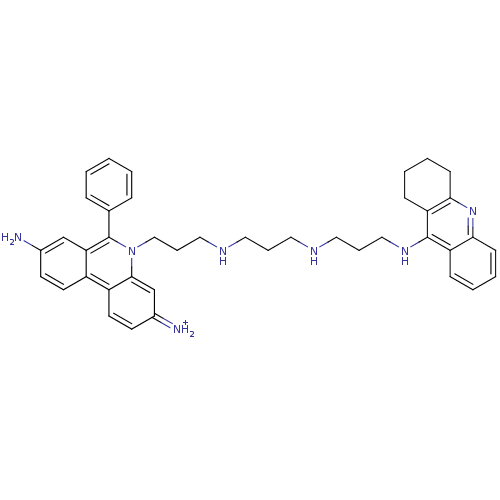
(3,8-Diamino-6-phenyl-5-(3-{3-[3-(1,2,3,4-tetrahydr...)Show SMILES Nc1ccc2c(c1)c(-c1ccccc1)n(CCCNCCCNCCCNc1c3CCCCc3nc3ccccc13)c1cc(=[NH2+])ccc21 Show InChI InChI=1S/C41H47N7/c42-30-17-19-32-33-20-18-31(43)28-39(33)48(41(36(32)27-30)29-11-2-1-3-12-29)26-10-24-45-22-8-21-44-23-9-25-46-40-34-13-4-6-15-37(34)47-38-16-7-5-14-35(38)40/h1-4,6,11-13,15,17-20,27-28,43-45H,5,7-10,14,16,21-26,42H2,(H,46,47)/p+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.49 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Competitive inhibition constant for Acetylcholinesterase |
J Med Chem 48: 24-7 (2005)
Article DOI: 10.1021/jm049156q
BindingDB Entry DOI: 10.7270/Q2P84CPM |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50379268

(CHEMBL3216556)Show SMILES Cl.Cl.Cl.Cl.[H][C@]12Cc3nc4cc(Cl)ccc4c(NCCCCCCCNc4c5CCCCc5nc5cc(Cl)ccc45)c3[C@]([H])(CC(C)=C1)C2 |r,c:47| Show InChI InChI=1S/C37H42Cl2N4/c1-23-17-24-19-25(18-23)35-34(20-24)43-33-22-27(39)12-14-30(33)37(35)41-16-8-4-2-3-7-15-40-36-28-9-5-6-10-31(28)42-32-21-26(38)11-13-29(32)36/h11-14,17,21-22,24-25H,2-10,15-16,18-20H2,1H3,(H,40,42)(H,41,43)/t24-,25+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona
Curated by ChEMBL
| Assay Description
Competitive inhibition of human recombinant AChE by Lineweaver-Burk plot analysis |
J Med Chem 55: 661-9 (2012)
Article DOI: 10.1021/jm200840c
BindingDB Entry DOI: 10.7270/Q26974KH |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50005188
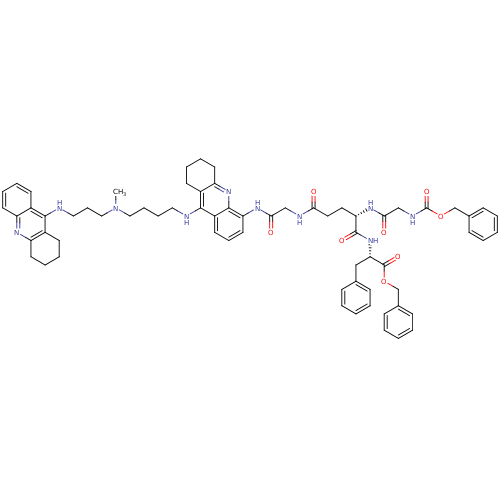
(CHEMBL3099500)Show SMILES CN(CCCCNc1c2CCCCc2nc2c(NC(=O)CNC(=O)CC[C@H](NC(=O)CNC(=O)OCc3ccccc3)C(=O)N[C@@H](Cc3ccccc3)C(=O)OCc3ccccc3)cccc12)CCCNc1c2CCCCc2nc2ccccc12 |r| Show InChI InChI=1S/C67H78N10O8/c1-77(40-20-38-69-62-49-27-11-14-31-53(49)72-54-32-15-12-28-50(54)62)39-18-17-37-68-63-51-29-13-16-33-55(51)75-64-52(63)30-19-34-56(64)73-60(79)42-70-59(78)36-35-57(74-61(80)43-71-67(83)85-45-48-25-9-4-10-26-48)65(81)76-58(41-46-21-5-2-6-22-46)66(82)84-44-47-23-7-3-8-24-47/h2-11,14,19,21-27,30-31,34,57-58H,12-13,15-18,20,28-29,32-33,35-45H2,1H3,(H,68,75)(H,69,72)(H,70,78)(H,71,83)(H,73,79)(H,74,80)(H,76,81)/t57-,58-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human acetylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM120262
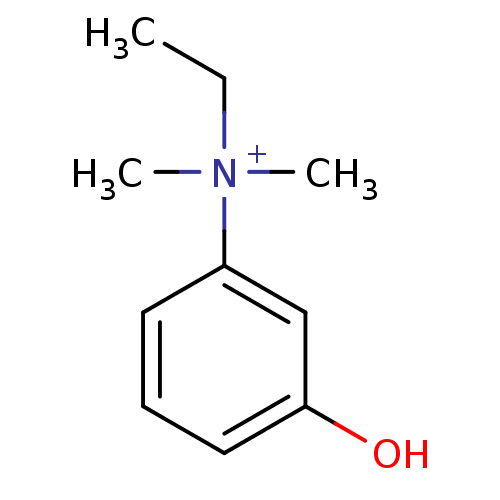
(EDROPHONIUM BROMIDE | EDROPHONIUM CHLORIDE | Edrop...)Show InChI InChI=1S/C10H15NO/c1-4-11(2,3)9-6-5-7-10(12)8-9/h5-8H,4H2,1-3H3/p+1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibition constant determined against Acetylcholinesterase (AChE) receptor. |
J Med Chem 41: 4186-9 (1998)
Checked by Author
Article DOI: 10.1021/jm9810452
BindingDB Entry DOI: 10.7270/Q2HH6KRM |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM8971
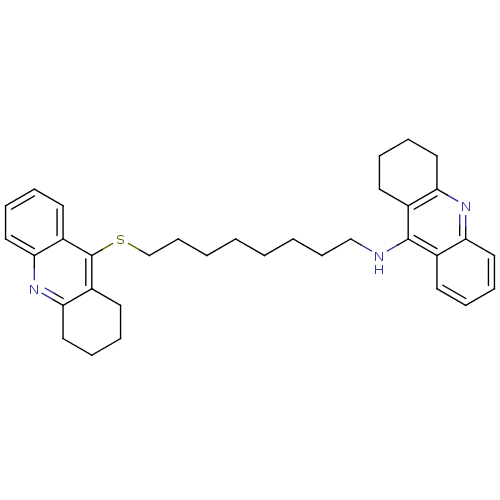
(CHEMBL129108 | N-[8-(1,2,3,4-tetrahydroacridin-9-y...)Show SMILES C(CCCCSc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H41N3S/c1(3-13-23-35-33-25-15-5-9-19-29(25)36-30-20-10-6-16-26(30)33)2-4-14-24-38-34-27-17-7-11-21-31(27)37-32-22-12-8-18-28(32)34/h5,7,9,11,15,17,19,21H,1-4,6,8,10,12-14,16,18,20,22-24H2,(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human butyrylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50005191

(CHEMBL3099498)Show SMILES O=C(CC[C@H](NC(=O)CNC(=O)OCc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)OCc1ccccc1)NCC(=O)Nc1cccc2c(NCCCCCCCCNc3c4CCCCc4nc4ccccc34)c3CCCCc3nc12 |r| Show InChI InChI=1S/C67H77N9O8/c77-59(38-37-57(74-61(79)43-71-67(82)84-45-48-27-12-7-13-28-48)65(80)76-58(41-46-23-8-5-9-24-46)66(81)83-44-47-25-10-6-11-26-47)70-42-60(78)73-56-36-22-32-52-63(51-31-16-19-35-55(51)75-64(52)56)69-40-21-4-2-1-3-20-39-68-62-49-29-14-17-33-53(49)72-54-34-18-15-30-50(54)62/h5-14,17,22-29,32-33,36,57-58H,1-4,15-16,18-21,30-31,34-35,37-45H2,(H,68,72)(H,69,75)(H,70,77)(H,71,82)(H,73,78)(H,74,79)(H,76,80)/t57-,58-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human acetylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Tetronarce californica (Pacific electric ray) (Tor...) | BDBM50138279

(CHEMBL3752467)Show SMILES C(CCCNCc1cc2ccccc2o1)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C29H35N3O/c1(2-10-18-30-21-23-20-22-12-4-9-17-28(22)33-23)3-11-19-31-29-24-13-5-7-15-26(24)32-27-16-8-6-14-25(27)29/h4-5,7,9,12-13,15,17,20,30H,1-3,6,8,10-11,14,16,18-19,21H2,(H,31,32) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Noncompetitive inhibition of Torpedo californica AChE using ATCh as substrate preincubated for 90 mins followed by substrate addition by potentiometr... |
J Med Chem 59: 114-31 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01119
BindingDB Entry DOI: 10.7270/Q2S75J5X |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50580216
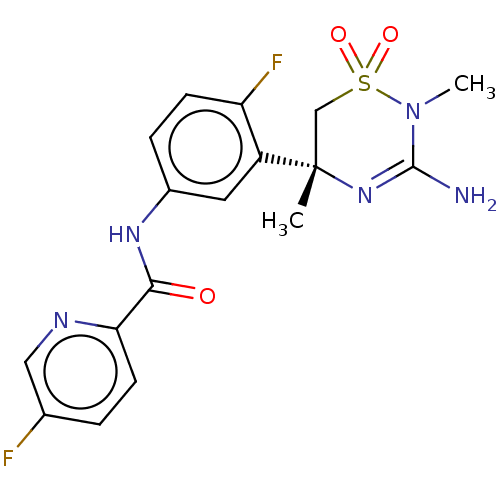
(MK-8931 | SCH 900931 | SCH-900931 | SCH900931 | VE...)Show SMILES CN1C(N)=N[C@@](C)(CS1(=O)=O)c1cc(NC(=O)c2ccc(F)cn2)ccc1F |r,c:3| | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00931
BindingDB Entry DOI: 10.7270/Q2CF9V25 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Tetronarce californica (Pacific electric ray) (Tor...) | BDBM50138279

(CHEMBL3752467)Show SMILES C(CCCNCc1cc2ccccc2o1)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C29H35N3O/c1(2-10-18-30-21-23-20-22-12-4-9-17-28(22)33-23)3-11-19-31-29-24-13-5-7-15-26(24)32-27-16-8-6-14-25(27)29/h4-5,7,9,12-13,15,17,20,30H,1-3,6,8,10-11,14,16,18-19,21H2,(H,31,32) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University
Curated by ChEMBL
| Assay Description
Competitive inhibition of Torpedo californica AChE using ATCh as substrate preincubated for 90 mins followed by substrate addition by potentiometric ... |
J Med Chem 59: 114-31 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01119
BindingDB Entry DOI: 10.7270/Q2S75J5X |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50007801
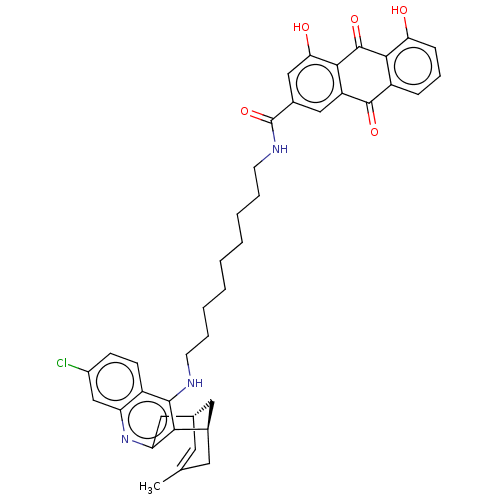
(CHEMBL3233832)Show SMILES Cl.[H][C@]12Cc3nc4cc(Cl)ccc4c(NCCCCCCCCCNC(=O)c4cc(O)c5C(=O)c6c(O)cccc6C(=O)c5c4)c3[C@]([H])(CC(C)=C1)C2 |r,c:54| Show InChI InChI=1S/C41H42ClN3O5.ClH/c1-23-16-24-18-25(17-23)35-32(19-24)45-31-22-27(42)12-13-28(31)38(35)43-14-7-5-3-2-4-6-8-15-44-41(50)26-20-30-37(34(47)21-26)40(49)36-29(39(30)48)10-9-11-33(36)46;/h9-13,16,20-22,24-25,46-47H,2-8,14-15,17-19H2,1H3,(H,43,45)(H,44,50);1H/t24-,25+;/m0./s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant AChE using acetylthiocholine iodide as substrate assessed as dissociation constant for enzyme-inhibitor complex by Li... |
J Med Chem 57: 2549-67 (2014)
Article DOI: 10.1021/jm401824w
BindingDB Entry DOI: 10.7270/Q2FX7BZ9 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50007801

(CHEMBL3233832)Show SMILES Cl.[H][C@]12Cc3nc4cc(Cl)ccc4c(NCCCCCCCCCNC(=O)c4cc(O)c5C(=O)c6c(O)cccc6C(=O)c5c4)c3[C@]([H])(CC(C)=C1)C2 |r,c:54| Show InChI InChI=1S/C41H42ClN3O5.ClH/c1-23-16-24-18-25(17-23)35-32(19-24)45-31-22-27(42)12-13-28(31)38(35)43-14-7-5-3-2-4-6-8-15-44-41(50)26-20-30-37(34(47)21-26)40(49)36-29(39(30)48)10-9-11-33(36)46;/h9-13,16,20-22,24-25,46-47H,2-8,14-15,17-19H2,1H3,(H,43,45)(H,44,50);1H/t24-,25+;/m0./s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universitat de Barcelona
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant AChE using acetylthiocholine iodide as substrate assessed as dissociation constant for enzyme-substrate-inhibitor com... |
J Med Chem 57: 2549-67 (2014)
Article DOI: 10.1021/jm401824w
BindingDB Entry DOI: 10.7270/Q2FX7BZ9 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50005189

(CHEMBL3099499)Show SMILES CN(CCCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2c(NC(=O)CNC(=O)CC[C@H](NC(=O)CNC(=O)OCc3ccccc3)C(=O)N[C@@H](Cc3ccccc3)C(=O)OCc3ccccc3)cccc12 |r| Show InChI InChI=1S/C67H78N10O8/c1-77(39-18-17-37-68-62-49-27-11-14-31-53(49)72-54-32-15-12-28-50(54)62)40-20-38-69-63-51-29-13-16-33-55(51)75-64-52(63)30-19-34-56(64)73-60(79)42-70-59(78)36-35-57(74-61(80)43-71-67(83)85-45-48-25-9-4-10-26-48)65(81)76-58(41-46-21-5-2-6-22-46)66(82)84-44-47-23-7-3-8-24-47/h2-11,14,19,21-27,30-31,34,57-58H,12-13,15-18,20,28-29,32-33,35-45H2,1H3,(H,68,72)(H,69,75)(H,70,78)(H,71,83)(H,73,79)(H,74,80)(H,76,81)/t57-,58-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human acetylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50157597
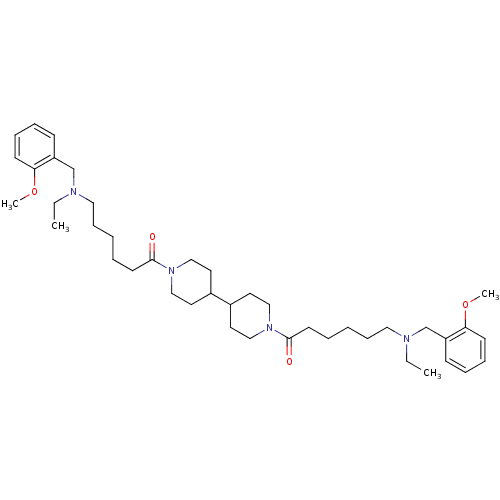
(6-[ethyl-(2-methoxy-benzyl)-amino]-1-(1'-{6-[ethyl...)Show SMILES CCN(CCCCCC(=O)N1CCC(CC1)C1CCN(CC1)C(=O)CCCCCN(CC)Cc1ccccc1OC)Cc1ccccc1OC Show InChI InChI=1S/C42H66N4O4/c1-5-43(33-37-17-11-13-19-39(37)49-3)27-15-7-9-21-41(47)45-29-23-35(24-30-45)36-25-31-46(32-26-36)42(48)22-10-8-16-28-44(6-2)34-38-18-12-14-20-40(38)50-4/h11-14,17-20,35-36H,5-10,15-16,21-34H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.67 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant AChE |
J Med Chem 47: 6490-8 (2004)
Article DOI: 10.1021/jm0494366
BindingDB Entry DOI: 10.7270/Q2JD4W91 |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50005192

(CHEMBL3099497)Show SMILES Nc1cccc2c(NCCCCCCCCNc3c4CCCCc4nc4ccccc34)c3CCCCc3nc12 Show InChI InChI=1S/C34H43N5/c35-28-18-13-17-27-33(26-16-7-10-21-31(26)39-34(27)28)37-23-12-4-2-1-3-11-22-36-32-24-14-5-8-19-29(24)38-30-20-9-6-15-25(30)32/h5,8,13-14,17-19H,1-4,6-7,9-12,15-16,20-23,35H2,(H,36,38)(H,37,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 8.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human butyrylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50124570
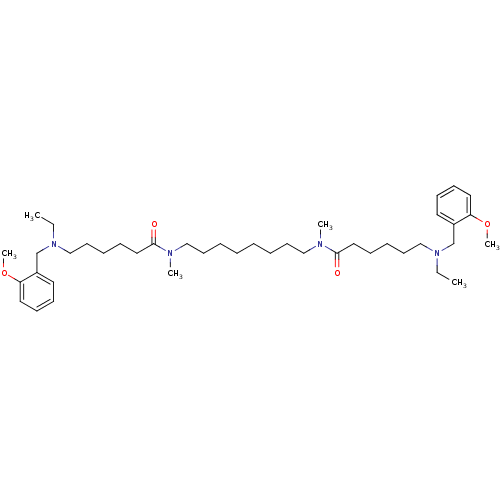
(6-[Ethyl-(2-methoxy-benzyl)-amino]-hexanoic acid [...)Show SMILES CCN(CCCCCC(=O)N(C)CCCCCCCCN(C)C(=O)CCCCCN(CC)Cc1ccccc1OC)Cc1ccccc1OC Show InChI InChI=1S/C42H70N4O4/c1-7-45(35-37-25-17-19-27-39(37)49-5)33-23-13-15-29-41(47)43(3)31-21-11-9-10-12-22-32-44(4)42(48)30-16-14-24-34-46(8-2)36-38-26-18-20-28-40(38)50-6/h17-20,25-28H,7-16,21-24,29-36H2,1-6H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12.2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant AChE |
J Med Chem 47: 6490-8 (2004)
Article DOI: 10.1021/jm0494366
BindingDB Entry DOI: 10.7270/Q2JD4W91 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50124570

(6-[Ethyl-(2-methoxy-benzyl)-amino]-hexanoic acid [...)Show SMILES CCN(CCCCCC(=O)N(C)CCCCCCCCN(C)C(=O)CCCCCN(CC)Cc1ccccc1OC)Cc1ccccc1OC Show InChI InChI=1S/C42H70N4O4/c1-7-45(35-37-25-17-19-27-39(37)49-5)33-23-13-15-29-41(47)43(3)31-21-11-9-10-12-22-32-44(4)42(48)30-16-14-24-34-46(8-2)36-38-26-18-20-28-40(38)50-6/h17-20,25-28H,7-16,21-24,29-36H2,1-6H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12.2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibitory activity of compound against acetylcholinesterase from human erythrocytes |
J Med Chem 46: 954-66 (2003)
Article DOI: 10.1021/jm021055+
BindingDB Entry DOI: 10.7270/Q2RF5TD4 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8960

((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...)Show SMILES COc1cc2CC(CC3CCN(Cc4ccccc4)CC3)C(=O)c2cc1OC Show InChI InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 20.5 | -45.6 | 23.1 | n/a | n/a | n/a | n/a | 8.0 | 37 |
University of Bologna
| Assay Description
The cholinesterase assays were performed using colorimetric method reported by Ellman. Estimates of the competitive inhibition constants (Ki) were ob... |
J Med Chem 46: 2279-82 (2003)
Article DOI: 10.1021/jm0340602
BindingDB Entry DOI: 10.7270/Q29Z9332 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8960

((+/-)-2-[(1-benzylpiperidin-4-yl)methyl]-5,6-dimet...)Show SMILES COc1cc2CC(CC3CCN(Cc4ccccc4)CC3)C(=O)c2cc1OC Show InChI InChI=1S/C24H29NO3/c1-27-22-14-19-13-20(24(26)21(19)15-23(22)28-2)12-17-8-10-25(11-9-17)16-18-6-4-3-5-7-18/h3-7,14-15,17,20H,8-13,16H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 20.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant AChE |
J Med Chem 47: 6490-8 (2004)
Article DOI: 10.1021/jm0494366
BindingDB Entry DOI: 10.7270/Q2JD4W91 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cholinesterase
(Homo sapiens (Human)) | BDBM50005188

(CHEMBL3099500)Show SMILES CN(CCCCNc1c2CCCCc2nc2c(NC(=O)CNC(=O)CC[C@H](NC(=O)CNC(=O)OCc3ccccc3)C(=O)N[C@@H](Cc3ccccc3)C(=O)OCc3ccccc3)cccc12)CCCNc1c2CCCCc2nc2ccccc12 |r| Show InChI InChI=1S/C67H78N10O8/c1-77(40-20-38-69-62-49-27-11-14-31-53(49)72-54-32-15-12-28-50(54)62)39-18-17-37-68-63-51-29-13-16-33-55(51)75-64-52(63)30-19-34-56(64)73-60(79)42-70-59(78)36-35-57(74-61(80)43-71-67(83)85-45-48-25-9-4-10-26-48)65(81)76-58(41-46-21-5-2-6-22-46)66(82)84-44-47-23-7-3-8-24-47/h2-11,14,19,21-27,30-31,34,57-58H,12-13,15-18,20,28-29,32-33,35-45H2,1H3,(H,68,75)(H,69,72)(H,70,78)(H,71,83)(H,73,79)(H,74,80)(H,76,81)/t57-,58-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human butyrylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM10949
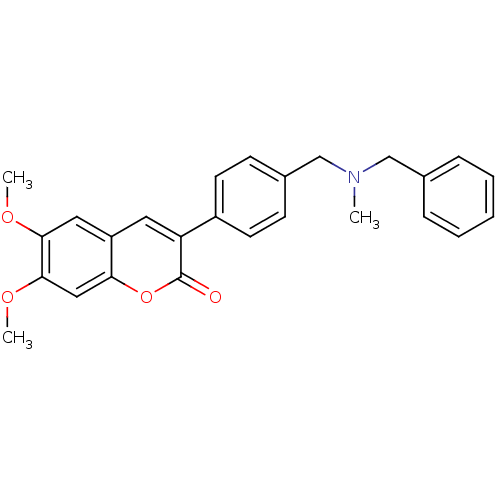
(3-(4-{[Benzyl(methyl)amino]methyl}-phenyl)-6,7-dim...)Show SMILES COc1cc2cc(-c3ccc(CN(C)Cc4ccccc4)cc3)c(=O)oc2cc1OC Show InChI InChI=1S/C26H25NO4/c1-27(16-18-7-5-4-6-8-18)17-19-9-11-20(12-10-19)22-13-21-14-24(29-2)25(30-3)15-23(21)31-26(22)28/h4-15H,16-17H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 21.7 | -45.5 | 44.5 | n/a | n/a | n/a | n/a | 8.0 | 37 |
University of Bologna
| Assay Description
The cholinesterase assays were performed using colorimetric method reported by Ellman. Estimates of the competitive inhibition constants (Ki) were ob... |
J Med Chem 46: 2279-82 (2003)
Article DOI: 10.1021/jm0340602
BindingDB Entry DOI: 10.7270/Q29Z9332 |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50510823
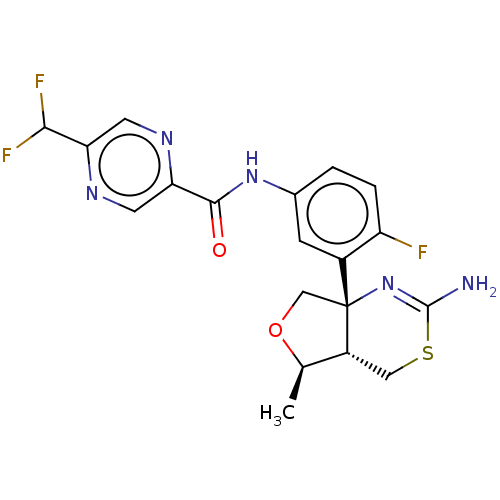
(E-2609 | E2609 | Elenbecestat)Show SMILES [H][C@]12CSC(N)=N[C@]1(CO[C@@H]2C)c1cc(NC(=O)c2cnc(cn2)C(F)F)ccc1F |r,c:5| Show InChI InChI=1S/C19H18F3N5O2S/c1-9-12-7-30-18(23)27-19(12,8-29-9)11-4-10(2-3-13(11)20)26-17(28)15-6-24-14(5-25-15)16(21)22/h2-6,9,12,16H,7-8H2,1H3,(H2,23,27)(H,26,28)/t9-,12-,19-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00931
BindingDB Entry DOI: 10.7270/Q2CF9V25 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8971

(CHEMBL129108 | N-[8-(1,2,3,4-tetrahydroacridin-9-y...)Show SMILES C(CCCCSc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2ccccc12 Show InChI InChI=1S/C34H41N3S/c1(3-13-23-35-33-25-15-5-9-19-29(25)36-30-20-10-6-16-26(30)33)2-4-14-24-38-34-27-17-7-11-21-31(27)37-32-22-12-8-18-28(32)34/h5,7,9,11,15,17,19,21H,1-4,6,8,10,12-14,16,18,20,22-24H2,(H,35,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 28 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human acetylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50278990

(CHEMBL4164524)Show InChI InChI=1S/C18H21Cl2N3O2/c19-16-11-15(12-17(20)23-16)22-18(24)25-10-6-2-5-9-21-13-14-7-3-1-4-8-14/h1,3-4,7-8,11-12,21H,2,5-6,9-10,13H2,(H,22,23,24) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 39 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sapienza University of Rome
Curated by ChEMBL
| Assay Description
Mixed inhibition of electric eel AChE using acetylthiocholine iodide as substrate measured from 0.5 to 1.5 mins by Dixon plot analysis |
Eur J Med Chem 141: 197-210 (2017)
Article DOI: 10.1016/j.ejmech.2017.09.022
BindingDB Entry DOI: 10.7270/Q27083ZM |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50005191

(CHEMBL3099498)Show SMILES O=C(CC[C@H](NC(=O)CNC(=O)OCc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)OCc1ccccc1)NCC(=O)Nc1cccc2c(NCCCCCCCCNc3c4CCCCc4nc4ccccc34)c3CCCCc3nc12 |r| Show InChI InChI=1S/C67H77N9O8/c77-59(38-37-57(74-61(79)43-71-67(82)84-45-48-27-12-7-13-28-48)65(80)76-58(41-46-23-8-5-9-24-46)66(81)83-44-47-25-10-6-11-26-47)70-42-60(78)73-56-36-22-32-52-63(51-31-16-19-35-55(51)75-64(52)56)69-40-21-4-2-1-3-20-39-68-62-49-29-14-17-33-53(49)72-54-34-18-15-30-50(54)62/h5-14,17,22-29,32-33,36,57-58H,1-4,15-16,18-21,30-31,34-35,37-45H2,(H,68,72)(H,69,75)(H,70,77)(H,71,82)(H,73,78)(H,74,79)(H,76,80)/t57-,58-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 41 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human butyrylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50278986

(CHEMBL4161860)Show InChI InChI=1S/C19H23Cl2N3O/c20-17-12-16(13-18(21)24-17)19(25)23-11-7-2-1-6-10-22-14-15-8-4-3-5-9-15/h3-5,8-9,12-13,22H,1-2,6-7,10-11,14H2,(H,23,25) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 53 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sapienza University of Rome
Curated by ChEMBL
| Assay Description
Mixed inhibition of electric eel AChE using acetylthiocholine iodide as substrate measured from 0.5 to 1.5 mins by Dixon plot analysis |
Eur J Med Chem 141: 197-210 (2017)
Article DOI: 10.1016/j.ejmech.2017.09.022
BindingDB Entry DOI: 10.7270/Q27083ZM |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50308280
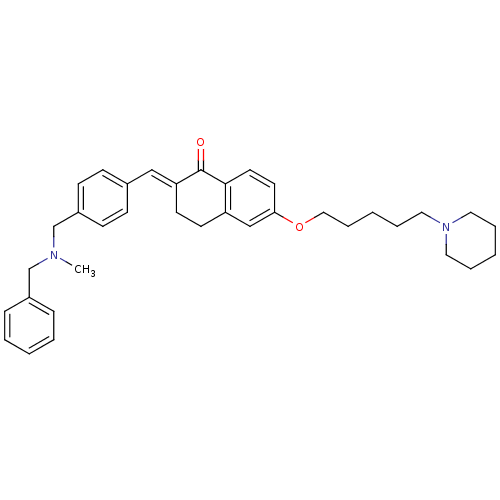
(2-{4-[(Benzylmethylamino)methyl]benzylidene}-6-(5-...)Show SMILES CN(Cc1ccccc1)Cc1ccc(\C=C2/CCc3cc(OCCCCCN4CCCCC4)ccc3C2=O)cc1 Show InChI InChI=1S/C36H44N2O2/c1-37(27-30-11-5-2-6-12-30)28-31-15-13-29(14-16-31)25-33-18-17-32-26-34(19-20-35(32)36(33)39)40-24-10-4-9-23-38-21-7-3-8-22-38/h2,5-6,11-16,19-20,25-26H,3-4,7-10,17-18,21-24,27-28H2,1H3/b33-25+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 61.1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant AChE-mediated hydrolysis of acetylcholine by Lineweaver-Burk plot analysis |
Bioorg Med Chem 18: 1749-60 (2010)
Article DOI: 10.1016/j.bmc.2010.01.071
BindingDB Entry DOI: 10.7270/Q2P26Z7H |
More data for this
Ligand-Target Pair | |
Cholinesterase
(Homo sapiens (Human)) | BDBM50005189

(CHEMBL3099499)Show SMILES CN(CCCCNc1c2CCCCc2nc2ccccc12)CCCNc1c2CCCCc2nc2c(NC(=O)CNC(=O)CC[C@H](NC(=O)CNC(=O)OCc3ccccc3)C(=O)N[C@@H](Cc3ccccc3)C(=O)OCc3ccccc3)cccc12 |r| Show InChI InChI=1S/C67H78N10O8/c1-77(39-18-17-37-68-62-49-27-11-14-31-53(49)72-54-32-15-12-28-50(54)62)40-20-38-69-63-51-29-13-16-33-55(51)75-64-52(63)30-19-34-56(64)73-60(79)42-70-59(78)36-35-57(74-61(80)43-71-67(83)85-45-48-25-9-4-10-26-48)65(81)76-58(41-46-21-5-2-6-22-46)66(82)84-44-47-23-7-3-8-24-47/h2-11,14,19,21-27,30-31,34,57-58H,12-13,15-18,20,28-29,32-33,35-45H2,1H3,(H,68,72)(H,69,75)(H,70,78)(H,71,83)(H,73,79)(H,74,80)(H,76,81)/t57-,58-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 62 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Siena
Curated by ChEMBL
| Assay Description
Inhibition of human butyrylcholine esterase |
ACS Med Chem Lett 4: 1178-82 (2013)
Article DOI: 10.1021/ml4002908
BindingDB Entry DOI: 10.7270/Q2TQ6318 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50381952

(CHEMBL2022933)Show InChI InChI=1S/C19H20N6/c1-2-6-24-9-5-16-15(12-24)17(21)14-10-13(11-20)19(23-18(14)22-16)25-7-3-4-8-25/h1,10H,3-9,12H2,(H2,21,22,23) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 65 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratorio de Radicales Libres y Qu�mica Computacional (IQOG, CSIC)
Curated by ChEMBL
| Assay Description
Non-competitive inhibition of electric eel AChE using acetylthiocholine as substrate by Lineweaver-Burk plot analysis |
Eur J Med Chem 52: 251-62 (2012)
Article DOI: 10.1016/j.ejmech.2012.03.022
BindingDB Entry DOI: 10.7270/Q25H7H97 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50067482

(6-[(2-Methoxy-benzyl)-methyl-amino]-hexanoic acid ...)Show SMILES COc1ccccc1CN(C)CCCCCC(=O)N(C)CCCCCCCCN(C)C(=O)CCCCCN(C)Cc1ccccc1OC Show InChI InChI=1S/C40H66N4O4/c1-41(33-35-23-15-17-25-37(35)47-5)29-19-11-13-27-39(45)43(3)31-21-9-7-8-10-22-32-44(4)40(46)28-14-12-20-30-42(2)34-36-24-16-18-26-38(36)48-6/h15-18,23-26H,7-14,19-22,27-34H2,1-6H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 104 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Competitive inhibition constant for Acetylcholinesterase |
J Med Chem 48: 24-7 (2005)
Article DOI: 10.1021/jm049156q
BindingDB Entry DOI: 10.7270/Q2P84CPM |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50067482

(6-[(2-Methoxy-benzyl)-methyl-amino]-hexanoic acid ...)Show SMILES COc1ccccc1CN(C)CCCCCC(=O)N(C)CCCCCCCCN(C)C(=O)CCCCCN(C)Cc1ccccc1OC Show InChI InChI=1S/C40H66N4O4/c1-41(33-35-23-15-17-25-37(35)47-5)29-19-11-13-27-39(45)43(3)31-21-9-7-8-10-22-32-44(4)40(46)28-14-12-20-30-42(2)34-36-24-16-18-26-38(36)48-6/h15-18,23-26H,7-14,19-22,27-34H2,1-6H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 104 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant AChE |
J Med Chem 47: 6490-8 (2004)
Article DOI: 10.1021/jm0494366
BindingDB Entry DOI: 10.7270/Q2JD4W91 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM50067482

(6-[(2-Methoxy-benzyl)-methyl-amino]-hexanoic acid ...)Show SMILES COc1ccccc1CN(C)CCCCCC(=O)N(C)CCCCCCCCN(C)C(=O)CCCCCN(C)Cc1ccccc1OC Show InChI InChI=1S/C40H66N4O4/c1-41(33-35-23-15-17-25-37(35)47-5)29-19-11-13-27-39(45)43(3)31-21-9-7-8-10-22-32-44(4)40(46)28-14-12-20-30-42(2)34-36-24-16-18-26-38(36)48-6/h15-18,23-26H,7-14,19-22,27-34H2,1-6H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 104 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibitory activity of compound against acetylcholinesterase from human erythrocytes |
J Med Chem 46: 954-66 (2003)
Article DOI: 10.1021/jm021055+
BindingDB Entry DOI: 10.7270/Q2RF5TD4 |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8961

(1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...)Show InChI InChI=1S/C13H14N2/c14-13-9-5-1-3-7-11(9)15-12-8-4-2-6-10(12)13/h1,3,5,7H,2,4,6,8H2,(H2,14,15) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 151 | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
| Assay Description
The cholinesterase assays were performed using colorimetric method reported by Ellman. The absorbance changes at 412 nm were recorded for 5 min with ... |
Bioorg Med Chem 8: 497-506 (2000)
Article DOI: 10.1016/s0968-0896(99)00306-5
BindingDB Entry DOI: 10.7270/Q2BG2M6F |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8961

(1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...)Show InChI InChI=1S/C13H14N2/c14-13-9-5-1-3-7-11(9)15-12-8-4-2-6-10(12)13/h1,3,5,7H,2,4,6,8H2,(H2,14,15) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 151 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Inhibitory activity of compound against acetylcholinesterase from human erythrocytes |
J Med Chem 46: 954-66 (2003)
Article DOI: 10.1021/jm021055+
BindingDB Entry DOI: 10.7270/Q2RF5TD4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM8961

(1,2,3,4-tetrahydro-9-acridinamine | 1,2,3,4-tetrah...)Show InChI InChI=1S/C13H14N2/c14-13-9-5-1-3-7-11(9)15-12-8-4-2-6-10(12)13/h1,3,5,7H,2,4,6,8H2,(H2,14,15) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 151 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Competitive inhibition constant for Acetylcholinesterase |
J Med Chem 48: 24-7 (2005)
Article DOI: 10.1021/jm049156q
BindingDB Entry DOI: 10.7270/Q2P84CPM |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Acetylcholinesterase
(Homo sapiens (Human)) | BDBM10512
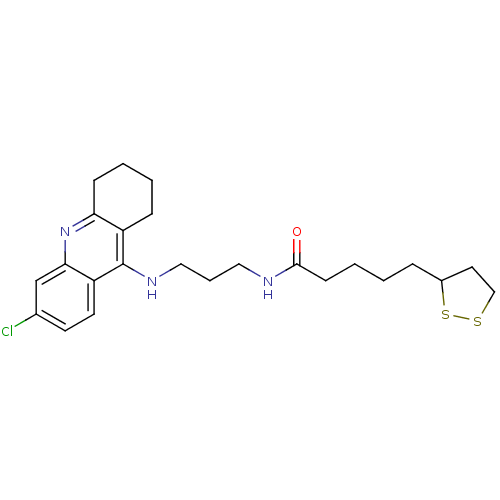
(CHEMBL194823 | N-{3-[(6-chloro-1,2,3,4-tetrahydroa...)Show SMILES Clc1ccc2c(NCCCNC(=O)CCCCC3CCSS3)c3CCCCc3nc2c1 Show InChI InChI=1S/C24H32ClN3OS2/c25-17-10-11-20-22(16-17)28-21-8-3-2-7-19(21)24(20)27-14-5-13-26-23(29)9-4-1-6-18-12-15-30-31-18/h10-11,16,18H,1-9,12-15H2,(H,26,29)(H,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 155 | -40.4 | 0.253 | n/a | n/a | n/a | n/a | 8.0 | 37 |
University of Bologna
| Assay Description
The cholinesterase assays were performed using colorimetric method reported by Ellman. Enzyme activity was determined by measuring the absorbance at... |
J Med Chem 48: 360-3 (2005)
Article DOI: 10.1021/jm049112h
BindingDB Entry DOI: 10.7270/Q2JQ0Z7J |
More data for this
Ligand-Target Pair | |
Acetylcholinesterase
(Electrophorus electricus (Electric eel)) | BDBM50278968
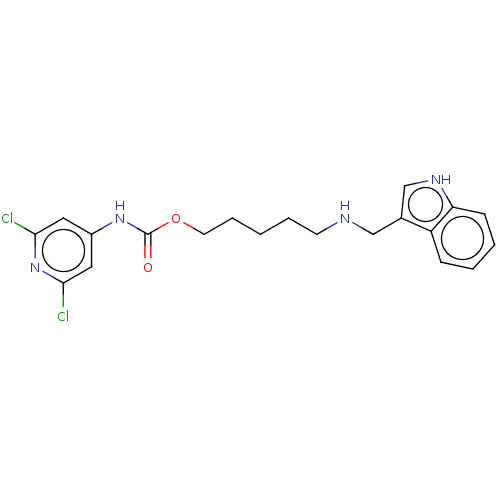
(CHEMBL4164186)Show SMILES Clc1cc(NC(=O)OCCCCCNCc2c[nH]c3ccccc23)cc(Cl)n1 Show InChI InChI=1S/C20H22Cl2N4O2/c21-18-10-15(11-19(22)26-18)25-20(27)28-9-5-1-4-8-23-12-14-13-24-17-7-3-2-6-16(14)17/h2-3,6-7,10-11,13,23-24H,1,4-5,8-9,12H2,(H,25,26,27) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 157 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sapienza University of Rome
Curated by ChEMBL
| Assay Description
Mixed inhibition of electric eel AChE using acetylthiocholine iodide as substrate measured from 0.5 to 1.5 mins by Dixon plot analysis |
Eur J Med Chem 141: 197-210 (2017)
Article DOI: 10.1016/j.ejmech.2017.09.022
BindingDB Entry DOI: 10.7270/Q27083ZM |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data