| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 11B2, mitochondrial |
|---|
| Ligand | BDBM50125050 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1518969 (CHEMBL3626121) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Martin, RE; Aebi, JD; Hornsperger, B; Krebs, HJ; Kuhn, B; Kuglstatter, A; Alker, AM; M�rki, HP; M�ller, S; Burger, D; Ottaviani, G; Riboulet, W; Verry, P; Tan, X; Amrein, K; Mayweg, AV Discovery of 4-Aryl-5,6,7,8-tetrahydroisoquinolines as Potent, Selective, and Orally Active Aldosterone Synthase (CYP11B2) Inhibitors: In Vivo Evaluation in Rodents and Cynomolgus Monkeys. J Med Chem58:8054-65 (2015) [PubMed] Article Martin, RE; Aebi, JD; Hornsperger, B; Krebs, HJ; Kuhn, B; Kuglstatter, A; Alker, AM; M�rki, HP; M�ller, S; Burger, D; Ottaviani, G; Riboulet, W; Verry, P; Tan, X; Amrein, K; Mayweg, AV Discovery of 4-Aryl-5,6,7,8-tetrahydroisoquinolines as Potent, Selective, and Orally Active Aldosterone Synthase (CYP11B2) Inhibitors: In Vivo Evaluation in Rodents and Cynomolgus Monkeys. J Med Chem58:8054-65 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 11B2, mitochondrial |
|---|
| Name: | Cytochrome P450 11B2, mitochondrial |
|---|
| Synonyms: | Aldosterone synthase | C11B2_MOUSE | CYPXIB2 | Cyp11b-2 | Cyp11b2 | Cytochrome P450 11B2, mitochondrial | Cytochrome P450C11 | Steroid 11-beta-hydroxylase |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 57397.51 |
|---|
| Organism: | Mus musculus |
|---|
| Description: | ChEMBL_109884 |
|---|
| Residue: | 500 |
|---|
| Sequence: | MALRVTADVWLARPWQCLHRTRALGTTATLAPKTLQPFEAIPQYSRNKWLKMIQILREQG
QENLHLEMHQVFRELGPIFRHSVGKTQIVSVMLPEDAEKLHQVESMLPRRMHLEPWVAHR
ELRGLRRGVFLLNGPEWRLNRLRLNRNVLSPKAVQKFVPMVDMVARDFLETLKEKVLQNA
RGSLTMDVQQSLFNYTIEASNFALFGERLGLLGHDLSPGSLKFIHALHSMFKSTSQLLFL
PKSLTRWTSTRVWKEHFDAWDVISEYANRCIWKVHQELRLGSSQTYSGIVAELISQGSLP
LDAIKANSMELTAGSVDTTAIPLVMTLFELARNPDVQKALRQESLAAEASIAANPQKAMS
DLPLLRAALKETLRLYPVGGFLERILSSDLVLQNYHVPAGTLVLLYLYSMGRNPAVFPRP
ERYMPQRWLERKRSFQHLAFGFGVRQCLGRRLAEVEMMLLLHHILKTFQVETLRQEDVQM
AYRFVLMPSSSPVLTFRPVS
|
|
|
|---|
| BDBM50125050 |
|---|
| n/a |
|---|
| Name | BDBM50125050 |
|---|
| Synonyms: | CHEMBL3623831 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H19N3O |
|---|
| Mol. Mass. | 305.3737 |
|---|
| SMILES | CCC(=O)N[C@H]1CCCc2c1cncc2-c1ccc(cc1)C#N |r| |
|---|
| Structure | 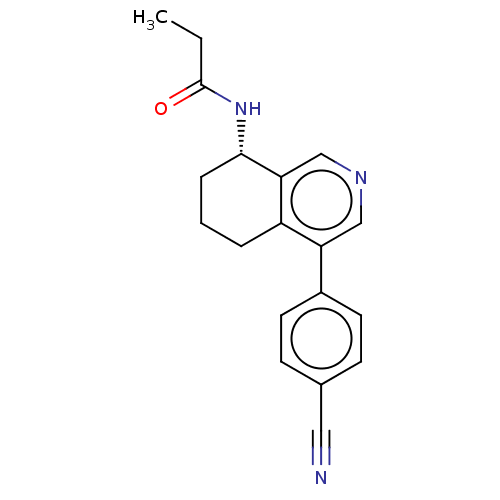
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Martin, RE; Aebi, JD; Hornsperger, B; Krebs, HJ; Kuhn, B; Kuglstatter, A; Alker, AM; M�rki, HP; M�ller, S; Burger, D; Ottaviani, G; Riboulet, W; Verry, P; Tan, X; Amrein, K; Mayweg, AV Discovery of 4-Aryl-5,6,7,8-tetrahydroisoquinolines as Potent, Selective, and Orally Active Aldosterone Synthase (CYP11B2) Inhibitors: In Vivo Evaluation in Rodents and Cynomolgus Monkeys. J Med Chem58:8054-65 (2015) [PubMed] Article
Martin, RE; Aebi, JD; Hornsperger, B; Krebs, HJ; Kuhn, B; Kuglstatter, A; Alker, AM; M�rki, HP; M�ller, S; Burger, D; Ottaviani, G; Riboulet, W; Verry, P; Tan, X; Amrein, K; Mayweg, AV Discovery of 4-Aryl-5,6,7,8-tetrahydroisoquinolines as Potent, Selective, and Orally Active Aldosterone Synthase (CYP11B2) Inhibitors: In Vivo Evaluation in Rodents and Cynomolgus Monkeys. J Med Chem58:8054-65 (2015) [PubMed] Article