| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 6 |
|---|
| Ligand | BDBM50236844 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1659425 (CHEMBL4009037) |
|---|
| Ki | 1.6±n/a nM |
|---|
| Citation |  Nirogi, R; Shinde, A; Kambhampati, RS; Mohammed, AR; Saraf, SK; Badange, RK; Bandyala, TR; Bhatta, V; Bojja, K; Reballi, V; Subramanian, R; Benade, V; Palacharla, RC; Bhyrapuneni, G; Jayarajan, P; Goyal, V; Jasti, V Discovery and Development of 1-[(2-Bromophenyl)sulfonyl]-5-methoxy-3-[(4-methyl-1-piperazinyl)methyl]-1H-indole Dimesylate Monohydrate (SUVN-502): A Novel, Potent, Selective and Orally Active Serotonin 6 (5-HT J Med Chem60:1843-1859 (2017) [PubMed] Article Nirogi, R; Shinde, A; Kambhampati, RS; Mohammed, AR; Saraf, SK; Badange, RK; Bandyala, TR; Bhatta, V; Bojja, K; Reballi, V; Subramanian, R; Benade, V; Palacharla, RC; Bhyrapuneni, G; Jayarajan, P; Goyal, V; Jasti, V Discovery and Development of 1-[(2-Bromophenyl)sulfonyl]-5-methoxy-3-[(4-methyl-1-piperazinyl)methyl]-1H-indole Dimesylate Monohydrate (SUVN-502): A Novel, Potent, Selective and Orally Active Serotonin 6 (5-HT J Med Chem60:1843-1859 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 6 |
|---|
| Name: | 5-hydroxytryptamine receptor 6 |
|---|
| Synonyms: | 5-HT-6 | 5-HT6 | 5-hydroxytryptamine receptor 6 (5-HT-6) | 5-hydroxytryptamine receptor 6 (5-HT6R) | 5-hydroxytryptamine receptor 6 (5HT6) | 5HT6R_HUMAN | HTR6 | Serotonin (5-HT3) receptor | Serotonin 6 (5-HT6) receptor | Serotonin Receptor 6 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 46968.43 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P50406 |
|---|
| Residue: | 440 |
|---|
| Sequence: | MVPEPGPTANSTPAWGAGPPSAPGGSGWVAAALCVVIALTAAANSLLIALICTQPALRNT
SNFFLVSLFTSDLMVGLVVMPPAMLNALYGRWVLARGLCLLWTAFDVMCCSASILNLCLI
SLDRYLLILSPLRYKLRMTPLRALALVLGAWSLAALASFLPLLLGWHELGHARPPVPGQC
RLLASLPFVLVASGLTFFLPSGAICFTYCRILLAARKQAVQVASLTTGMASQASETLQVP
RTPRPGVESADSRRLATKHSRKALKASLTLGILLGMFFVTWLPFFVANIVQAVCDCISPG
LFDVLTWLGYCNSTMNPIIYPLFMRDFKRALGRFLPCPRCPRERQASLASPSLRTSHSGP
RPGLSLQQVLPLPLPPDSDSDSDAGSGGSSGLRLTAQLLLPGEATQDPPLPTRAAAAVNF
FNIDPAEPELRPHPLGIPTN
|
|
|
|---|
| BDBM50236844 |
|---|
| n/a |
|---|
| Name | BDBM50236844 |
|---|
| Synonyms: | CHEMBL4100484 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H39N3O9S3 |
|---|
| Mol. Mass. | 633.798 |
|---|
| SMILES | CS(O)(=O)=O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccc(cc1)C(C)C |
|---|
| Structure | 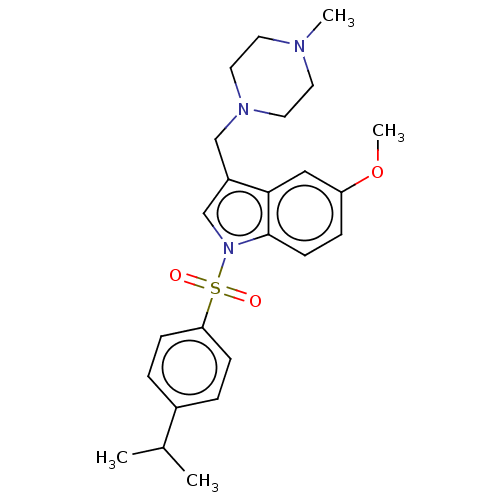
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nirogi, R; Shinde, A; Kambhampati, RS; Mohammed, AR; Saraf, SK; Badange, RK; Bandyala, TR; Bhatta, V; Bojja, K; Reballi, V; Subramanian, R; Benade, V; Palacharla, RC; Bhyrapuneni, G; Jayarajan, P; Goyal, V; Jasti, V Discovery and Development of 1-[(2-Bromophenyl)sulfonyl]-5-methoxy-3-[(4-methyl-1-piperazinyl)methyl]-1H-indole Dimesylate Monohydrate (SUVN-502): A Novel, Potent, Selective and Orally Active Serotonin 6 (5-HT J Med Chem60:1843-1859 (2017) [PubMed] Article
Nirogi, R; Shinde, A; Kambhampati, RS; Mohammed, AR; Saraf, SK; Badange, RK; Bandyala, TR; Bhatta, V; Bojja, K; Reballi, V; Subramanian, R; Benade, V; Palacharla, RC; Bhyrapuneni, G; Jayarajan, P; Goyal, V; Jasti, V Discovery and Development of 1-[(2-Bromophenyl)sulfonyl]-5-methoxy-3-[(4-methyl-1-piperazinyl)methyl]-1H-indole Dimesylate Monohydrate (SUVN-502): A Novel, Potent, Selective and Orally Active Serotonin 6 (5-HT J Med Chem60:1843-1859 (2017) [PubMed] Article