| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cathepsin D |
|---|
| Ligand | BDBM223395 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1748605 (CHEMBL4183115) |
|---|
| IC50 | >95300±n/a nM |
|---|
| Citation |  O'Neill, BT; Beck, EM; Butler, CR; Nolan, CE; Gonzales, C; Zhang, L; Doran, SD; Lapham, K; Buzon, LM; Dutra, JK; Barreiro, G; Hou, X; Martinez-Alsina, LA; Rogers, BN; Villalobos, A; Murray, JC; Ogilvie, K; LaChapelle, EA; Chang, C; Lanyon, LF; Steppan, CM; Robshaw, A; Hales, K; Boucher, GG; Pandher, K; Houle, C; Ambroise, CW; Karanian, D; Riddell, D; Bales, KR; Brodney, MA Design and Synthesis of Clinical Candidate PF-06751979: A Potent, Brain Penetrant, ?-Site Amyloid Precursor Protein Cleaving Enzyme 1 (BACE1) Inhibitor Lacking Hypopigmentation. J Med Chem61:4476-4504 (2018) [PubMed] Article O'Neill, BT; Beck, EM; Butler, CR; Nolan, CE; Gonzales, C; Zhang, L; Doran, SD; Lapham, K; Buzon, LM; Dutra, JK; Barreiro, G; Hou, X; Martinez-Alsina, LA; Rogers, BN; Villalobos, A; Murray, JC; Ogilvie, K; LaChapelle, EA; Chang, C; Lanyon, LF; Steppan, CM; Robshaw, A; Hales, K; Boucher, GG; Pandher, K; Houle, C; Ambroise, CW; Karanian, D; Riddell, D; Bales, KR; Brodney, MA Design and Synthesis of Clinical Candidate PF-06751979: A Potent, Brain Penetrant, ?-Site Amyloid Precursor Protein Cleaving Enzyme 1 (BACE1) Inhibitor Lacking Hypopigmentation. J Med Chem61:4476-4504 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cathepsin D |
|---|
| Name: | Cathepsin D |
|---|
| Synonyms: | CATD_HUMAN | CPSD | CTSD | Cathepsin D [Precursor] | Cathepsin D heavy chain | Cathepsin D light chain | Cathepsin D precursor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 44551.72 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human proCathepsin D (SwissProt accession number P07339) was expressed in Sf9 cells, purified, and autoactivated. |
|---|
| Residue: | 412 |
|---|
| Sequence: | MQPSSLLPLALCLLAAPASALVRIPLHKFTSIRRTMSEVGGSVEDLIAKGPVSKYSQAVP
AVTEGPIPEVLKNYMDAQYYGEIGIGTPPQCFTVVFDTGSSNLWVPSIHCKLLDIACWIH
HKYNSDKSSTYVKNGTSFDIHYGSGSLSGYLSQDTVSVPCQSASSASALGGVKVERQVFG
EATKQPGITFIAAKFDGILGMAYPRISVNNVLPVFDNLMQQKLVDQNIFSFYLSRDPDAQ
PGGELMLGGTDSKYYKGSLSYLNVTRKAYWQVHLDQVEVASGLTLCKEGCEAIVDTGTSL
MVGPVDEVRELQKAIGAVPLIQGEYMIPCEKVSTLPAITLKLGGKGYKLSPEDYTLKVSQ
AGKTLCLSGFMGMDIPPPSGPLWILGDVFIGRYYTVFDRDNNRVGFAEAARL
|
|
|
|---|
| BDBM223395 |
|---|
| n/a |
|---|
| Name | BDBM223395 |
|---|
| Synonyms: | US9315520, 1 | US9315520, Comparator 1 | US9315520, Example 1 | US9315520, Example 2 | US9605007, Example 1 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H19F2N5O3S2 |
|---|
| Mol. Mass. | 455.502 |
|---|
| SMILES | C[C@H]1C[C@H]2CSC(N)=N[C@]2(CO1)c1nc(NC(=O)c2ccc(OC(F)F)cn2)cs1 |r,c:7| |
|---|
| Structure | 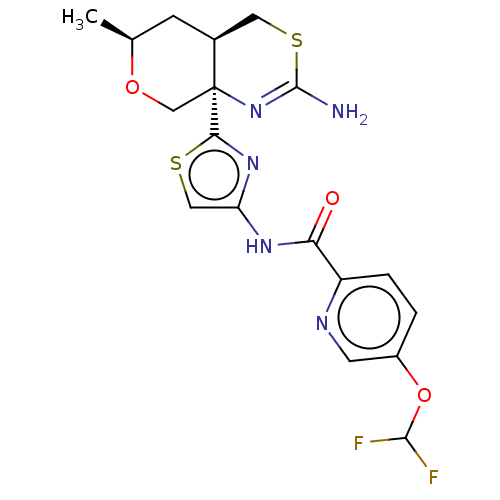
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 O'Neill, BT; Beck, EM; Butler, CR; Nolan, CE; Gonzales, C; Zhang, L; Doran, SD; Lapham, K; Buzon, LM; Dutra, JK; Barreiro, G; Hou, X; Martinez-Alsina, LA; Rogers, BN; Villalobos, A; Murray, JC; Ogilvie, K; LaChapelle, EA; Chang, C; Lanyon, LF; Steppan, CM; Robshaw, A; Hales, K; Boucher, GG; Pandher, K; Houle, C; Ambroise, CW; Karanian, D; Riddell, D; Bales, KR; Brodney, MA Design and Synthesis of Clinical Candidate PF-06751979: A Potent, Brain Penetrant, ?-Site Amyloid Precursor Protein Cleaving Enzyme 1 (BACE1) Inhibitor Lacking Hypopigmentation. J Med Chem61:4476-4504 (2018) [PubMed] Article
O'Neill, BT; Beck, EM; Butler, CR; Nolan, CE; Gonzales, C; Zhang, L; Doran, SD; Lapham, K; Buzon, LM; Dutra, JK; Barreiro, G; Hou, X; Martinez-Alsina, LA; Rogers, BN; Villalobos, A; Murray, JC; Ogilvie, K; LaChapelle, EA; Chang, C; Lanyon, LF; Steppan, CM; Robshaw, A; Hales, K; Boucher, GG; Pandher, K; Houle, C; Ambroise, CW; Karanian, D; Riddell, D; Bales, KR; Brodney, MA Design and Synthesis of Clinical Candidate PF-06751979: A Potent, Brain Penetrant, ?-Site Amyloid Precursor Protein Cleaving Enzyme 1 (BACE1) Inhibitor Lacking Hypopigmentation. J Med Chem61:4476-4504 (2018) [PubMed] Article