| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50507989 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1832016 (CHEMBL4332024) |
|---|
| IC50 | >20000±n/a nM |
|---|
| Citation |  Nguyen, D; Lemos, C; Wortmann, L; Eis, K; Holton, SJ; Boemer, U; Moosmayer, D; Eberspaecher, U; Weiske, J; Lechner, C; Prechtl, S; Suelzle, D; Siegel, F; Prinz, F; Lesche, R; Nicke, B; Nowak-Reppel, K; Himmel, H; Mumberg, D; von Nussbaum, F; Nising, CF; Bauser, M; Haegebarth, A Discovery and Characterization of the Potent and Highly Selective (Piperidin-4-yl)pyrido[3,2- d]pyrimidine Based in Vitro Probe BAY-885 for the Kinase ERK5. J Med Chem62:928-940 (2019) [PubMed] Article Nguyen, D; Lemos, C; Wortmann, L; Eis, K; Holton, SJ; Boemer, U; Moosmayer, D; Eberspaecher, U; Weiske, J; Lechner, C; Prechtl, S; Suelzle, D; Siegel, F; Prinz, F; Lesche, R; Nicke, B; Nowak-Reppel, K; Himmel, H; Mumberg, D; von Nussbaum, F; Nising, CF; Bauser, M; Haegebarth, A Discovery and Characterization of the Potent and Highly Selective (Piperidin-4-yl)pyrido[3,2- d]pyrimidine Based in Vitro Probe BAY-885 for the Kinase ERK5. J Med Chem62:928-940 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50507989 |
|---|
| n/a |
|---|
| Name | BDBM50507989 |
|---|
| Synonyms: | CHEMBL4445670 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H28F3N7O2 |
|---|
| Mol. Mass. | 515.5307 |
|---|
| SMILES | CN1CCN(CC1)c1cnc2c(ncnc2c1)C1CCN(CC1)C(=O)c1ccc(OC(F)(F)F)cc1N |
|---|
| Structure | 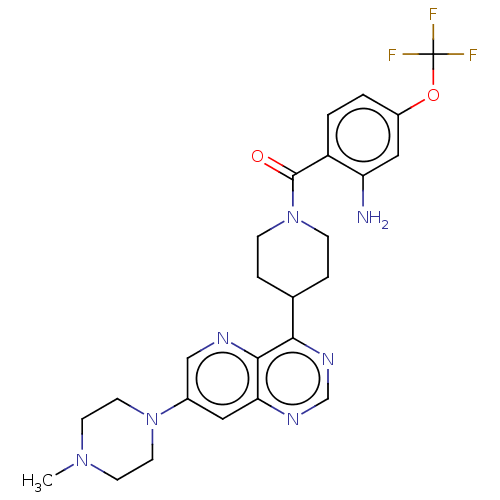
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nguyen, D; Lemos, C; Wortmann, L; Eis, K; Holton, SJ; Boemer, U; Moosmayer, D; Eberspaecher, U; Weiske, J; Lechner, C; Prechtl, S; Suelzle, D; Siegel, F; Prinz, F; Lesche, R; Nicke, B; Nowak-Reppel, K; Himmel, H; Mumberg, D; von Nussbaum, F; Nising, CF; Bauser, M; Haegebarth, A Discovery and Characterization of the Potent and Highly Selective (Piperidin-4-yl)pyrido[3,2- d]pyrimidine Based in Vitro Probe BAY-885 for the Kinase ERK5. J Med Chem62:928-940 (2019) [PubMed] Article
Nguyen, D; Lemos, C; Wortmann, L; Eis, K; Holton, SJ; Boemer, U; Moosmayer, D; Eberspaecher, U; Weiske, J; Lechner, C; Prechtl, S; Suelzle, D; Siegel, F; Prinz, F; Lesche, R; Nicke, B; Nowak-Reppel, K; Himmel, H; Mumberg, D; von Nussbaum, F; Nising, CF; Bauser, M; Haegebarth, A Discovery and Characterization of the Potent and Highly Selective (Piperidin-4-yl)pyrido[3,2- d]pyrimidine Based in Vitro Probe BAY-885 for the Kinase ERK5. J Med Chem62:928-940 (2019) [PubMed] Article