 Found 485 hits with Last Name = 'weiske' and Initial = 'j'
Found 485 hits with Last Name = 'weiske' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM50180955
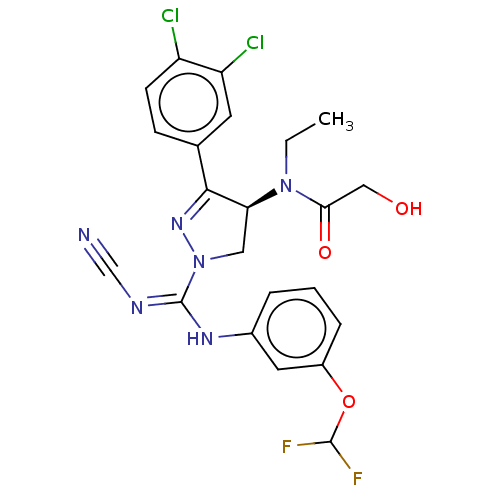
(CHEMBL3818617)Show SMILES CCN([C@H]1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cccc(OC(F)F)c1)=N/C#N)C(=O)CO |r,c:6| Show InChI InChI=1S/C22H20Cl2F2N6O3/c1-2-31(19(34)11-33)18-10-32(30-20(18)13-6-7-16(23)17(24)8-13)22(28-12-27)29-14-4-3-5-15(9-14)35-21(25)26/h3-9,18,21,33H,2,10-11H2,1H3,(H,28,29)/t18-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
BAYER Pharma AG
Curated by ChEMBL
| Assay Description
Competitive inhibition of full length 6xHis-tagged SMYD2 (unknown origin) expressed in Escherichia coli using varying levels of Btn-Ahx-GSRAHSSHLKSKK... |
J Med Chem 59: 4578-600 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01890
BindingDB Entry DOI: 10.7270/Q2RN39S5 |
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM50180955

(CHEMBL3818617)Show SMILES CCN([C@H]1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cccc(OC(F)F)c1)=N/C#N)C(=O)CO |r,c:6| Show InChI InChI=1S/C22H20Cl2F2N6O3/c1-2-31(19(34)11-33)18-10-32(30-20(18)13-6-7-16(23)17(24)8-13)22(28-12-27)29-14-4-3-5-15(9-14)35-21(25)26/h3-9,18,21,33H,2,10-11H2,1H3,(H,28,29)/t18-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 28 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
BAYER Pharma AG
Curated by ChEMBL
| Assay Description
Uncompetitive inhibition of full length 6xHis-tagged SMYD2 (unknown origin) expressed in Escherichia coli using fixed levels of Btn-Ahx-GSRAHSSHLKSKK... |
J Med Chem 59: 4578-600 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01890
BindingDB Entry DOI: 10.7270/Q2RN39S5 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50611048

(BAY-091) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PDB
UniChem
| PDB
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Ubiquitin carboxyl-terminal hydrolase 21
(Homo sapiens (Human)) | BDBM50612991

(CHEMBL5279102) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50611049

(CHEMBL5280127) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283143
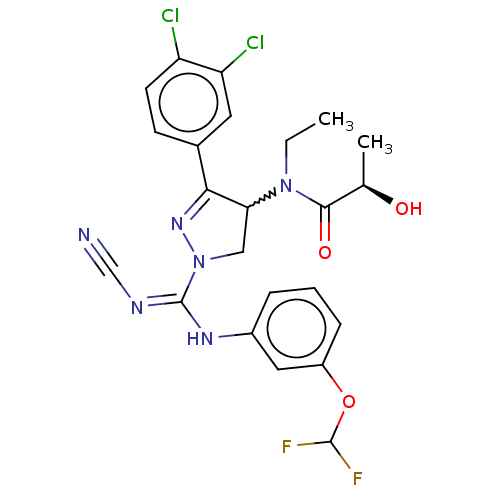
((2R)—N-[1-{N′-cyano-N-[3-(difluorometho...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cccc(OC(F)F)c1)=N/C#N)C(=O)[C@@H](C)O |r,w:3.2,c:6| Show InChI InChI=1S/C23H22Cl2F2N6O3/c1-3-32(21(35)13(2)34)19-11-33(31-20(19)14-7-8-17(24)18(25)9-14)23(29-12-28)30-15-5-4-6-16(10-15)36-22(26)27/h4-10,13,19,22,34H,3,11H2,1-2H3,(H,29,30)/t13-,19?/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.64 | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
For the detection of SMYD2 cellular methylation activity an In Cell Western (ICW) assay was established. This assay allows rapid processing of multip... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50611049

(CHEMBL5280127) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50611048

(BAY-091) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PDB
UniChem
| PDB
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50610997
(CHEMBL5276719) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50610997

(CHEMBL5276719) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 5.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283194
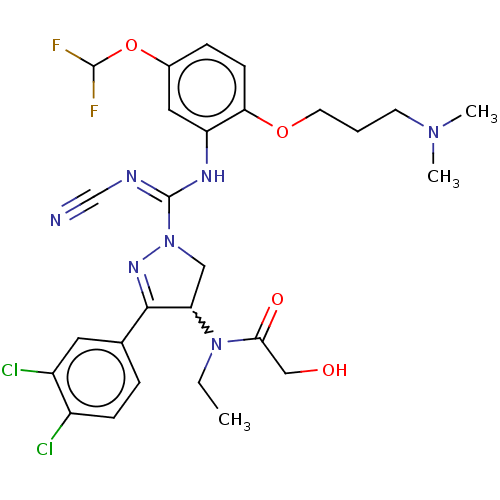
(Rac-N-[1-(N′-cyano-N-{5-(difluoromethoxy)-2-...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cc(OC(F)F)ccc1OCCCN(C)C)=N/C#N)C(=O)CO |w:3.2,c:6| Show InChI InChI=1S/C27H31Cl2F2N7O4/c1-4-37(24(40)15-39)22-14-38(35-25(22)17-6-8-19(28)20(29)12-17)27(33-16-32)34-21-13-18(42-26(30)31)7-9-23(21)41-11-5-10-36(2)3/h6-9,12-13,22,26,39H,4-5,10-11,14-15H2,1-3H3,(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 5.55 | n/a | n/a | n/a | n/a | 9.0 | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
SMYD2 inhibitory activities of the compounds described in the present invention were quantified using a scintillation proximity assay (SPA) which mea... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
Ubiquitin carboxyl-terminal hydrolase 21
(Homo sapiens (Human)) | BDBM50612991

(CHEMBL5279102) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283173
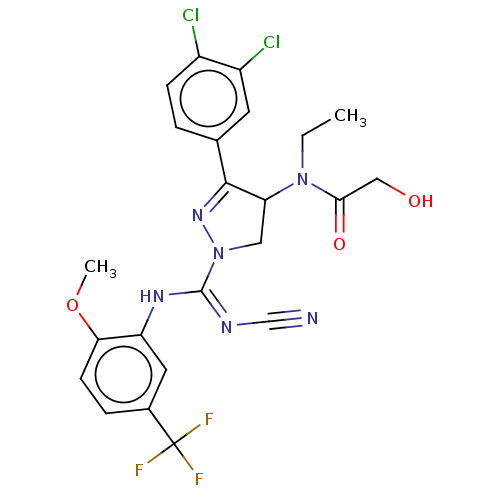
(N-[1-{N′-cyano-N-[2-methoxy-5-(trifluorometh...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cc(ccc1OC)C(F)(F)F)=N/C#N)C(=O)CO |c:6| Show InChI InChI=1S/C23H21Cl2F3N6O3/c1-3-33(20(36)11-35)18-10-34(32-21(18)13-4-6-15(24)16(25)8-13)22(30-12-29)31-17-9-14(23(26,27)28)5-7-19(17)37-2/h4-9,18,35H,3,10-11H2,1-2H3,(H,30,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 6.65 | n/a | n/a | n/a | n/a | 9.0 | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
SMYD2 inhibitory activities of the compounds described in the present invention were quantified using a scintillation proximity assay (SPA) which mea... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50611050

(CHEMBL5270765) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Ubiquitin carboxyl-terminal hydrolase 21
(Homo sapiens (Human)) | BDBM50612990

(CHEMBL5268828) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50611048

(BAY-091) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PDB
UniChem
| PDB
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283196
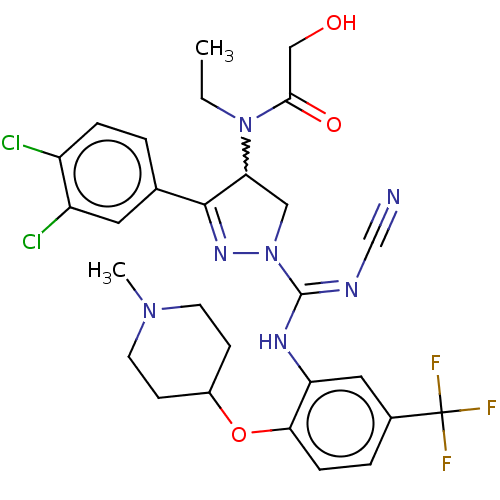
(Rac-N-[1-(N′-cyano-N-{2-[(1-methylpiperidin-...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cc(ccc1OC1CCN(C)CC1)C(F)(F)F)=N/C#N)C(=O)CO |w:3.2,c:6| Show InChI InChI=1S/C28H30Cl2F3N7O3/c1-3-39(25(42)15-41)23-14-40(37-26(23)17-4-6-20(29)21(30)12-17)27(35-16-34)36-22-13-18(28(31,32)33)5-7-24(22)43-19-8-10-38(2)11-9-19/h4-7,12-13,19,23,41H,3,8-11,14-15H2,1-2H3,(H,35,36) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 9.09 | n/a | n/a | n/a | n/a | 9.0 | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
SMYD2 inhibitory activities of the compounds described in the present invention were quantified using a scintillation proximity assay (SPA) which mea... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283172
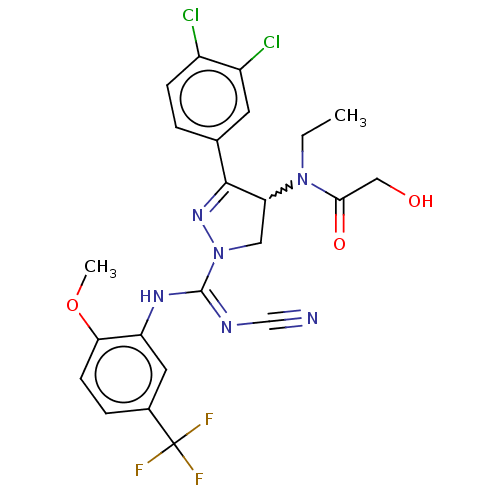
(Rac-N-[1-{N′-cyano-N-[2-methoxy-5-(trifluoro...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cc(ccc1OC)C(F)(F)F)=N/C#N)C(=O)CO |w:3.2,c:6| Show InChI InChI=1S/C23H21Cl2F3N6O3/c1-3-33(20(36)11-35)18-10-34(32-21(18)13-4-6-15(24)16(25)8-13)22(30-12-29)31-17-9-14(23(26,27)28)5-7-19(17)37-2/h4-9,18,35H,3,10-11H2,1-2H3,(H,30,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 9.98 | n/a | n/a | n/a | n/a | 9.0 | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
SMYD2 inhibitory activities of the compounds described in the present invention were quantified using a scintillation proximity assay (SPA) which mea... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
Proteinase-activated receptor 1
(Homo sapiens (Human)) | BDBM50181506
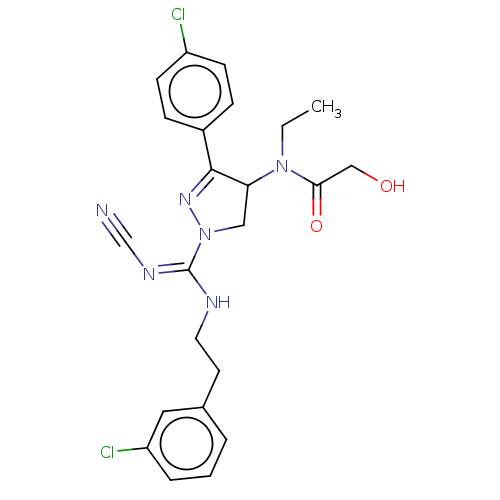
(CHEMBL3819038)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)cc1)C(\NCCc1cccc(Cl)c1)=N/C#N)C(=O)CO |c:6| Show InChI InChI=1S/C23H24Cl2N6O2/c1-2-30(21(33)14-32)20-13-31(29-22(20)17-6-8-18(24)9-7-17)23(28-15-26)27-11-10-16-4-3-5-19(25)12-16/h3-9,12,20,32H,2,10-11,13-14H2,1H3,(H,27,28) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
BAYER Pharma AG
Curated by ChEMBL
| Assay Description
Antagonist activity at PAR1 (unknown origin) |
J Med Chem 59: 4578-600 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01890
BindingDB Entry DOI: 10.7270/Q2RN39S5 |
More data for this
Ligand-Target Pair | |
Ubiquitin carboxyl-terminal hydrolase 21
(Homo sapiens (Human)) | BDBM50612988

(CHEMBL5286335) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Ubiquitin carboxyl-terminal hydrolase 21
(Homo sapiens (Human)) | BDBM50612990

(CHEMBL5268828) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283169

(Rac-N-[1-{N′-cyano-N-[5-(difluoromethoxy)-2-...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cc(OC(F)F)ccc1OC)=N/C#N)C(=O)CO |w:3.2,c:6| Show InChI InChI=1S/C23H22Cl2F2N6O4/c1-3-32(20(35)11-34)18-10-33(31-21(18)13-4-6-15(24)16(25)8-13)23(29-12-28)30-17-9-14(37-22(26)27)5-7-19(17)36-2/h4-9,18,22,34H,3,10-11H2,1-2H3,(H,29,30) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 12.5 | n/a | n/a | n/a | n/a | 9.0 | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
SMYD2 inhibitory activities of the compounds described in the present invention were quantified using a scintillation proximity assay (SPA) which mea... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283143

((2R)—N-[1-{N′-cyano-N-[3-(difluorometho...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cccc(OC(F)F)c1)=N/C#N)C(=O)[C@@H](C)O |r,w:3.2,c:6| Show InChI InChI=1S/C23H22Cl2F2N6O3/c1-3-32(21(35)13(2)34)19-11-33(31-20(19)14-7-8-17(24)18(25)9-14)23(29-12-28)30-15-5-4-6-16(10-15)36-22(26)27/h4-10,13,19,22,34H,3,11H2,1-2H3,(H,29,30)/t13-,19?/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | 9.0 | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
SMYD2 inhibitory activities of the compounds described in the present invention were quantified using a scintillation proximity assay (SPA) which mea... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50611046
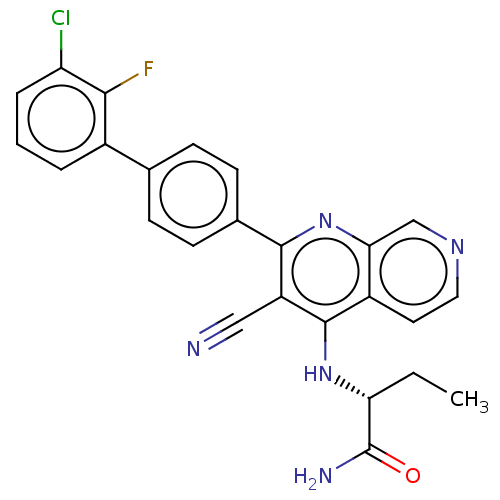
(CHEMBL5290005) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PDB
UniChem
| PDB
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283139
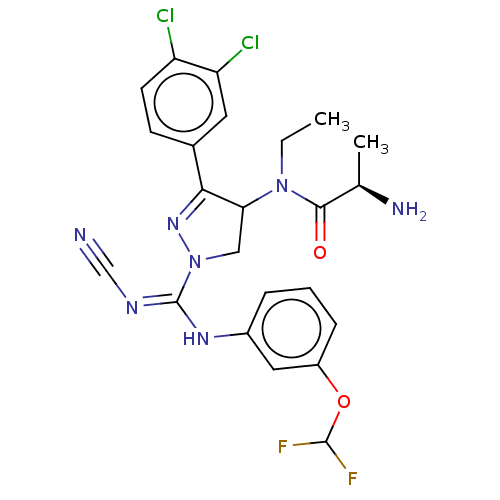
(N-[1-{N′-cyano-N-[3-(difluoromethoxy)phenyl]...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cccc(OC(F)F)c1)=N/C#N)C(=O)[C@@H](C)N |r,c:6| Show InChI InChI=1S/C23H23Cl2F2N7O2/c1-3-33(21(35)13(2)29)19-11-34(32-20(19)14-7-8-17(24)18(25)9-14)23(30-12-28)31-15-5-4-6-16(10-15)36-22(26)27/h4-10,13,19,22H,3,11,29H2,1-2H3,(H,30,31)/t13-,19?/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 13.2 | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
For the detection of SMYD2 cellular methylation activity an In Cell Western (ICW) assay was established. This assay allows rapid processing of multip... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283139

(N-[1-{N′-cyano-N-[3-(difluoromethoxy)phenyl]...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cccc(OC(F)F)c1)=N/C#N)C(=O)[C@@H](C)N |r,c:6| Show InChI InChI=1S/C23H23Cl2F2N7O2/c1-3-33(21(35)13(2)29)19-11-34(32-20(19)14-7-8-17(24)18(25)9-14)23(30-12-28)31-15-5-4-6-16(10-15)36-22(26)27/h4-10,13,19,22H,3,11,29H2,1-2H3,(H,30,31)/t13-,19?/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 13.5 | n/a | n/a | n/a | n/a | 9.0 | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
SMYD2 inhibitory activities of the compounds described in the present invention were quantified using a scintillation proximity assay (SPA) which mea... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50611049

(CHEMBL5280127) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50610996

(CHEMBL5271652) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283132
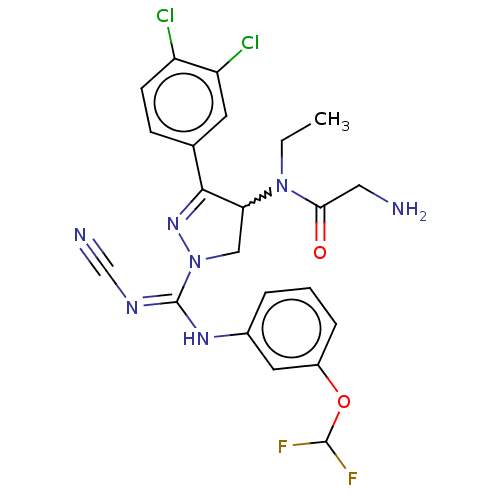
(Rac-N-[1-{N′-cyano-N-[3-(difluoromethoxy)phe...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cccc(OC(F)F)c1)=N/C#N)C(=O)CN |w:3.2,c:6| Show InChI InChI=1S/C22H21Cl2F2N7O2/c1-2-32(19(34)10-27)18-11-33(31-20(18)13-6-7-16(23)17(24)8-13)22(29-12-28)30-14-4-3-5-15(9-14)35-21(25)26/h3-9,18,21H,2,10-11,27H2,1H3,(H,29,30) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | 9.0 | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
SMYD2 inhibitory activities of the compounds described in the present invention were quantified using a scintillation proximity assay (SPA) which mea... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM50075102
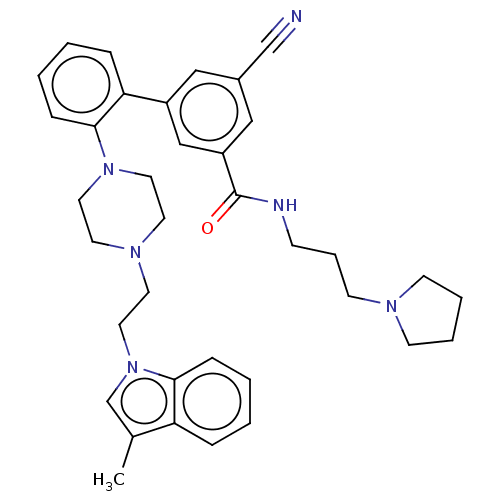
(CHEMBL3414623)Show SMILES Cc1cn(CCN2CCN(CC2)c2ccccc2-c2cc(cc(c2)C(=O)NCCCN2CCCC2)C#N)c2ccccc12 Show InChI InChI=1S/C36H42N6O/c1-28-27-42(34-11-4-2-9-32(28)34)22-19-40-17-20-41(21-18-40)35-12-5-3-10-33(35)30-23-29(26-37)24-31(25-30)36(43)38-13-8-16-39-14-6-7-15-39/h2-5,9-12,23-25,27H,6-8,13-22H2,1H3,(H,38,43) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | <15 | n/a | n/a | n/a | n/a | n/a | n/a |
BAYER Pharma AG
Curated by ChEMBL
| Assay Description
Inhibition of SMYD2 (unknown origin) expressed in Escherichia coli BL21 (DE3) using Biotinaminohexanoyl-GSRAHSSHLKSKKGQSTSRH as substrate after 75 mi... |
J Med Chem 59: 4578-600 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01890
BindingDB Entry DOI: 10.7270/Q2RN39S5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283195
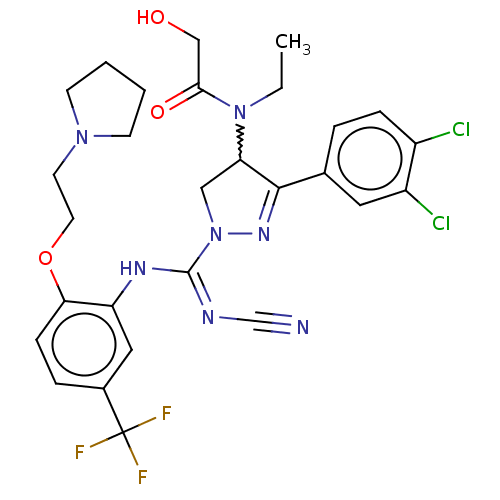
(Rac-N-[r-(N′-cyano-N-[2-[2-(pyrrolidin-1-yl)...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cc(ccc1OCCN1CCCC1)C(F)(F)F)=N/C#N)C(=O)CO |w:3.2,c:6| Show InChI InChI=1S/C28H30Cl2F3N7O3/c1-2-39(25(42)16-41)23-15-40(37-26(23)18-5-7-20(29)21(30)13-18)27(35-17-34)36-22-14-19(28(31,32)33)6-8-24(22)43-12-11-38-9-3-4-10-38/h5-8,13-14,23,41H,2-4,9-12,15-16H2,1H3,(H,35,36) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 15.8 | n/a | n/a | n/a | n/a | 9.0 | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
SMYD2 inhibitory activities of the compounds described in the present invention were quantified using a scintillation proximity assay (SPA) which mea... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50611048

(BAY-091) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PDB
UniChem
| PDB
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mitogen-activated protein kinase 7
(Homo sapiens (Human)) | BDBM50508008

(CHEMBL4438379)Show SMILES FC(F)(F)Oc1ccc(cc1)C(=O)N1CCC(CC1)c1ncnc2cc(cnc12)N1CCOCC1 Show InChI InChI=1S/C24H24F3N5O3/c25-24(26,27)35-19-3-1-17(2-4-19)23(33)32-7-5-16(6-8-32)21-22-20(29-15-30-21)13-18(14-28-22)31-9-11-34-12-10-31/h1-4,13-16H,5-12H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer AG
Curated by ChEMBL
| Assay Description
Inhibition of His-tagged MAP2K5 activated N-terminal GST-tagged recombinant human ERK5 (1 to 398 residues) expressed in Escherichia coli using biotin... |
J Med Chem 62: 928-940 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01606
BindingDB Entry DOI: 10.7270/Q2FN19H1 |
More data for this
Ligand-Target Pair | |
Ubiquitin carboxyl-terminal hydrolase 21
(Homo sapiens (Human)) | BDBM50612988

(CHEMBL5286335) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM50180967

(CHEMBL3818487)Show SMILES CCN([C@H]1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cc(F)cc(OC(F)F)c1)=N/C#N)C(=O)CO |r,c:6| Show InChI InChI=1S/C22H19Cl2F3N6O3/c1-2-32(19(35)10-34)18-9-33(31-20(18)12-3-4-16(23)17(24)5-12)22(29-11-28)30-14-6-13(25)7-15(8-14)36-21(26)27/h3-8,18,21,34H,2,9-10H2,1H3,(H,29,30)/t18-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
BAYER Pharma AG
Curated by ChEMBL
| Assay Description
Inhibition of SMYD2 (unknown origin) using H4 as substrate after 2hrs in presence 3H-SAM of by scintillation proximity assay |
J Med Chem 59: 4578-600 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01890
BindingDB Entry DOI: 10.7270/Q2RN39S5 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50611047

(CHEMBL5277824) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283184
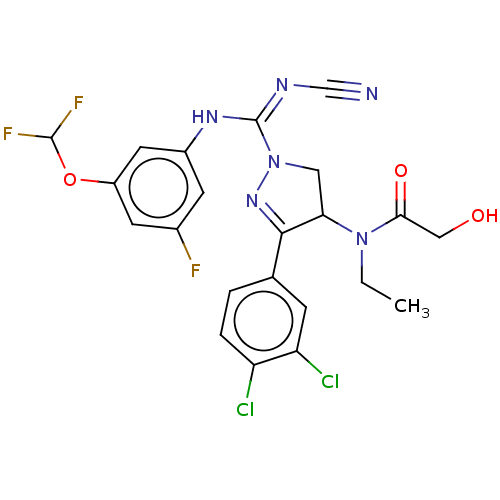
(N-[1-{N′-cyano-N-[3-(difluoromethoxy)-5-fluo...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cc(F)cc(OC(F)F)c1)=N/C#N)C(=O)CO |c:6| Show InChI InChI=1S/C22H19Cl2F3N6O3/c1-2-32(19(35)10-34)18-9-33(31-20(18)12-3-4-16(23)17(24)5-12)22(29-11-28)30-14-6-13(25)7-15(8-14)36-21(26)27/h3-8,18,21,34H,2,9-10H2,1H3,(H,29,30) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 19.2 | n/a | n/a | n/a | n/a | 9.0 | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
SMYD2 inhibitory activities of the compounds described in the present invention were quantified using a scintillation proximity assay (SPA) which mea... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50611050

(CHEMBL5270765) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 7
(Homo sapiens (Human)) | BDBM50508013
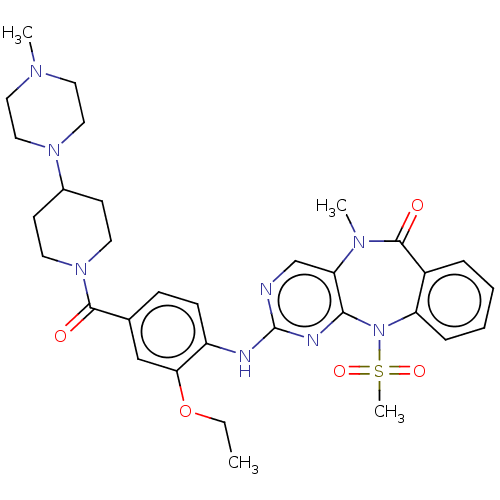
(CHEMBL4541479)Show SMILES CCOc1cc(ccc1Nc1ncc2N(C)C(=O)c3ccccc3N(c2n1)S(C)(=O)=O)C(=O)N1CCC(CC1)N1CCN(C)CC1 Show InChI InChI=1S/C32H40N8O5S/c1-5-45-28-20-22(30(41)39-14-12-23(13-15-39)38-18-16-36(2)17-19-38)10-11-25(28)34-32-33-21-27-29(35-32)40(46(4,43)44)26-9-7-6-8-24(26)31(42)37(27)3/h6-11,20-21,23H,5,12-19H2,1-4H3,(H,33,34,35) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer AG
Curated by ChEMBL
| Assay Description
Inhibition of His-tagged MAP2K5 activated N-terminal GST-tagged recombinant human ERK5 (1 to 398 residues) expressed in Escherichia coli using biotin... |
J Med Chem 62: 928-940 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01606
BindingDB Entry DOI: 10.7270/Q2FN19H1 |
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283172

(Rac-N-[1-{N′-cyano-N-[2-methoxy-5-(trifluoro...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cc(ccc1OC)C(F)(F)F)=N/C#N)C(=O)CO |w:3.2,c:6| Show InChI InChI=1S/C23H21Cl2F3N6O3/c1-3-33(20(36)11-35)18-10-34(32-21(18)13-4-6-15(24)16(25)8-13)22(30-12-29)31-17-9-14(23(26,27)28)5-7-19(17)37-2/h4-9,18,35H,3,10-11H2,1-2H3,(H,30,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 21.3 | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
For the detection of SMYD2 cellular methylation activity an In Cell Western (ICW) assay was established. This assay allows rapid processing of multip... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283205
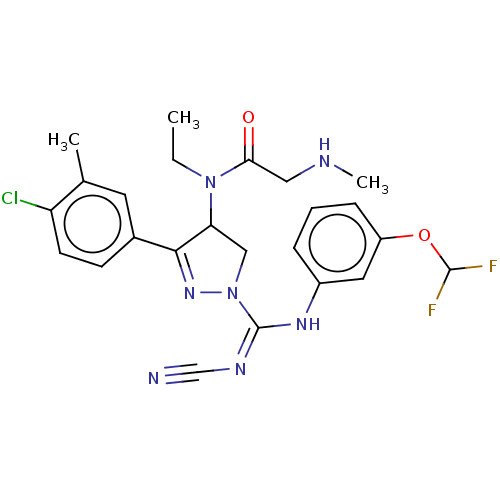
(N-[3-(4-Chloro-3-methylphenyl)-1-{N′-cyano-N...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(C)c1)C(\Nc1cccc(OC(F)F)c1)=N/C#N)C(=O)CNC |c:6| Show InChI InChI=1S/C24H26ClF2N7O2/c1-4-33(21(35)12-29-3)20-13-34(32-22(20)16-8-9-19(25)15(2)10-16)24(30-14-28)31-17-6-5-7-18(11-17)36-23(26)27/h5-11,20,23,29H,4,12-13H2,1-3H3,(H,30,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 22.9 | n/a | n/a | n/a | n/a | 9.0 | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
SMYD2 inhibitory activities of the compounds described in the present invention were quantified using a scintillation proximity assay (SPA) which mea... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283173

(N-[1-{N′-cyano-N-[2-methoxy-5-(trifluorometh...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cc(ccc1OC)C(F)(F)F)=N/C#N)C(=O)CO |c:6| Show InChI InChI=1S/C23H21Cl2F3N6O3/c1-3-33(20(36)11-35)18-10-34(32-21(18)13-4-6-15(24)16(25)8-13)22(30-12-29)31-17-9-14(23(26,27)28)5-7-19(17)37-2/h4-9,18,35H,3,10-11H2,1-2H3,(H,30,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 23.6 | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
For the detection of SMYD2 cellular methylation activity an In Cell Western (ICW) assay was established. This assay allows rapid processing of multip... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283197

(Rac-N-[1-(N′-cyano-N-{2-[(1-methylpiperidin-...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cc(ccc1OCCN1CCOCC1)C(F)(F)F)=N/C#N)C(=O)CO |w:3.2,c:6| Show InChI InChI=1S/C28H30Cl2F3N7O4/c1-2-39(25(42)16-41)23-15-40(37-26(23)18-3-5-20(29)21(30)13-18)27(35-17-34)36-22-14-19(28(31,32)33)4-6-24(22)44-12-9-38-7-10-43-11-8-38/h3-6,13-14,23,41H,2,7-12,15-16H2,1H3,(H,35,36) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 26.1 | n/a | n/a | n/a | n/a | 9.0 | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
SMYD2 inhibitory activities of the compounds described in the present invention were quantified using a scintillation proximity assay (SPA) which mea... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 5-phosphate 4-kinase type-2 alpha
(Homo sapiens) | BDBM50610997

(CHEMBL5276719) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM50180955

(CHEMBL3818617)Show SMILES CCN([C@H]1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cccc(OC(F)F)c1)=N/C#N)C(=O)CO |r,c:6| Show InChI InChI=1S/C22H20Cl2F2N6O3/c1-2-31(19(34)11-33)18-10-32(30-20(18)13-6-7-16(23)17(24)8-13)22(28-12-27)29-14-4-3-5-15(9-14)35-21(25)26/h3-9,18,21,33H,2,10-11H2,1H3,(H,28,29)/t18-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
BAYER Pharma AG
Curated by ChEMBL
| Assay Description
Inhibition of full length 6xHis-tagged SMYD2 (unknown origin) expressed in Escherichia coli using Btn-Ahx GSRAHSSHLKSKKGQSTSRH-amide as substrate aft... |
J Med Chem 59: 4578-600 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01890
BindingDB Entry DOI: 10.7270/Q2RN39S5 |
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283169

(Rac-N-[1-{N′-cyano-N-[5-(difluoromethoxy)-2-...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cc(OC(F)F)ccc1OC)=N/C#N)C(=O)CO |w:3.2,c:6| Show InChI InChI=1S/C23H22Cl2F2N6O4/c1-3-32(20(35)11-34)18-10-33(31-21(18)13-4-6-15(24)16(25)8-13)23(29-12-28)30-17-9-14(37-22(26)27)5-7-19(17)36-2/h4-9,18,22,34H,3,10-11H2,1-2H3,(H,29,30) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 27.7 | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
For the detection of SMYD2 cellular methylation activity an In Cell Western (ICW) assay was established. This assay allows rapid processing of multip... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
Ubiquitin carboxyl-terminal hydrolase 21
(Homo sapiens (Human)) | BDBM50612984
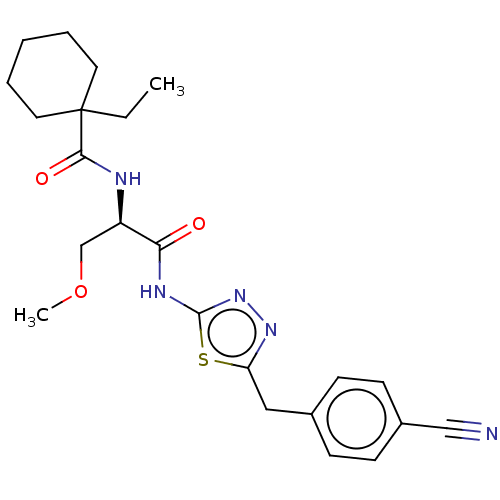
(CHEMBL5277351) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM50181508
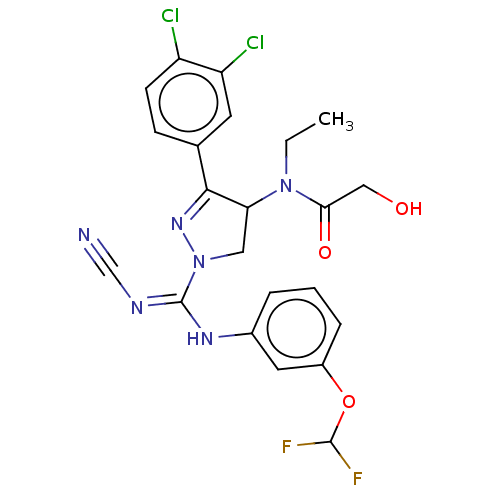
(CHEMBL3818083 | US10023539, Example 4.2)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cccc(OC(F)F)c1)=N/C#N)C(=O)CO |c:6| Show InChI InChI=1S/C22H20Cl2F2N6O3/c1-2-31(19(34)11-33)18-10-32(30-20(18)13-6-7-16(23)17(24)8-13)22(28-12-27)29-14-4-3-5-15(9-14)35-21(25)26/h3-9,18,21,33H,2,10-11H2,1H3,(H,28,29) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 28.2 | n/a | n/a | n/a | n/a | 9.0 | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
SMYD2 inhibitory activities of the compounds described in the present invention were quantified using a scintillation proximity assay (SPA) which mea... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM50181508

(CHEMBL3818083 | US10023539, Example 4.2)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cccc(OC(F)F)c1)=N/C#N)C(=O)CO |c:6| Show InChI InChI=1S/C22H20Cl2F2N6O3/c1-2-31(19(34)11-33)18-10-32(30-20(18)13-6-7-16(23)17(24)8-13)22(28-12-27)29-14-4-3-5-15(9-14)35-21(25)26/h3-9,18,21,33H,2,10-11H2,1H3,(H,28,29) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 28.2 | n/a | n/a | n/a | n/a | 9.0 | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
SMYD2 inhibitory activities of the compounds described in the present invention were quantified using a scintillation proximity assay (SPA) which mea... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |
N-lysine methyltransferase SMYD2
(Homo sapiens (Human)) | BDBM283170
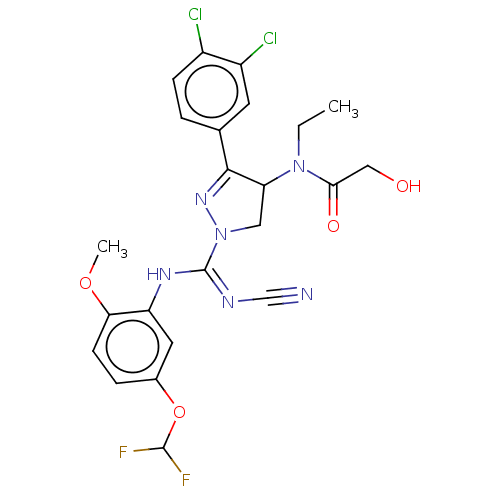
(N-[1-{N′-cyano-N-[5-(difluoromethoxy)-2-meth...)Show SMILES CCN(C1CN(N=C1c1ccc(Cl)c(Cl)c1)C(\Nc1cc(OC(F)F)ccc1OC)=N/C#N)C(=O)CO |c:6| Show InChI InChI=1S/C23H22Cl2F2N6O4/c1-3-32(20(35)11-34)18-10-33(31-21(18)13-4-6-15(24)16(25)8-13)23(29-12-28)30-17-9-14(37-22(26)27)5-7-19(17)36-2/h4-9,18,22,34H,3,10-11H2,1-2H3,(H,29,30) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 28.7 | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer Pharma Aktiengesellschaft
US Patent
| Assay Description
For the detection of SMYD2 cellular methylation activity an In Cell Western (ICW) assay was established. This assay allows rapid processing of multip... |
US Patent US10023539 (2018)
BindingDB Entry DOI: 10.7270/Q2MG7RKD |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data