| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Nitric oxide synthase, brain |
|---|
| Ligand | BDBM50164780 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_305408 (CHEMBL833057) |
|---|
| IC50 | 240±n/a nM |
|---|
| Citation |  Guthikonda, RN; Shah, SK; Pacholok, SG; Humes, JL; Mumford, RA; Grant, SK; Chabin, RM; Green, BG; Tsou, N; Ball, R; Fletcher, DS; Luell, S; Euan MacIntyre, D; Maccoss, M Bicyclic amidine inhibitors of nitric oxide synthase: discovery of perhydro-iminopyrindine and perhydro-iminoquinoline as potent, orally active inhibitors of inducible nitric oxide synthase. Bioorg Med Chem Lett15:1997-2001 (2005) [PubMed] Article Guthikonda, RN; Shah, SK; Pacholok, SG; Humes, JL; Mumford, RA; Grant, SK; Chabin, RM; Green, BG; Tsou, N; Ball, R; Fletcher, DS; Luell, S; Euan MacIntyre, D; Maccoss, M Bicyclic amidine inhibitors of nitric oxide synthase: discovery of perhydro-iminopyrindine and perhydro-iminoquinoline as potent, orally active inhibitors of inducible nitric oxide synthase. Bioorg Med Chem Lett15:1997-2001 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Nitric oxide synthase, brain |
|---|
| Name: | Nitric oxide synthase, brain |
|---|
| Synonyms: | Constitutive NOS | N-NOS | NC-NOS | NOS type I | NOS type I nNOS | NOS1 | NOS1_HUMAN | Neuronal NOS | Neuronal nitric oxide synthase | Nitric oxide synthase, brain (nNOS) | Nitric oxide synthase, neuronal (nNOS) | Peptidyl-cysteine S-nitrosylase NOS1 | bNOS | nNOS |
|---|
| Type: | Homodimer |
|---|
| Mol. Mass.: | 160985.98 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P29475 |
|---|
| Residue: | 1434 |
|---|
| Sequence: | MEDHMFGVQQIQPNVISVRLFKRKVGGLGFLVKERVSKPPVIISDLIRGGAAEQSGLIQA
GDIILAVNGRPLVDLSYDSALEVLRGIASETHVVLILRGPEGFTTHLETTFTGDGTPKTI
RVTQPLGPPTKAVDLSHQPPAGKEQPLAVDGASGPGNGPQHAYDDGQEAGSLPHANGLAP
RPPGQDPAKKATRVSLQGRGENNELLKEIEPVLSLLTSGSRGVKGGAPAKAEMKDMGIQV
DRDLDGKSHKPLPLGVENDRVFNDLWGKGNVPVVLNNPYSEKEQPPTSGKQSPTKNGSPS
KCPRFLKVKNWETEVVLTDTLHLKSTLETGCTEYICMGSIMHPSQHARRPEDVRTKGQLF
PLAKEFIDQYYSSIKRFGSKAHMERLEEVNKEIDTTSTYQLKDTELIYGAKHAWRNASRC
VGRIQWSKLQVFDARDCTTAHGMFNYICNHVKYATNKGNLRSAITIFPQRTDGKHDFRVW
NSQLIRYAGYKQPDGSTLGDPANVQFTEICIQQGWKPPRGRFDVLPLLLQANGNDPELFQ
IPPELVLEVPIRHPKFEWFKDLGLKWYGLPAVSNMLLEIGGLEFSACPFSGWYMGTEIGV
RDYCDNSRYNILEEVAKKMNLDMRKTSSLWKDQALVEINIAVLYSFQSDKVTIVDHHSAT
ESFIKHMENEYRCRGGCPADWVWIVPPMSGSITPVFHQEMLNYRLTPSFEYQPDPWNTHV
WKGTNGTPTKRRAIGFKKLAEAVKFSAKLMGQAMAKRVKATILYATETGKSQAYAKTLCE
IFKHAFDAKVMSMEEYDIVHLEHETLVLVVTSTFGNGDPPENGEKFGCALMEMRHPNSVQ
EERKSYKVRFNSVSSYSDSQKSSGDGPDLRDNFESAGPLANVRFSVFGLGSRAYPHFCAF
GHAVDTLLEELGGERILKMREGDELCGQEEAFRTWAKKVFKAACDVFCVGDDVNIEKANN
SLISNDRSWKRNKFRLTFVAEAPELTQGLSNVHKKRVSAARLLSRQNLQSPKSSRSTIFV
RLHTNGSQELQYQPGDHLGVFPGNHEDLVNALIERLEDAPPVNQMVKVELLEERNTALGV
ISNWTDELRLPPCTIFQAFKYYLDITTPPTPLQLQQFASLATSEKEKQRLLVLSKGLQEY
EEWKWGKNPTIVEVLEEFPSIQMPATLLLTQLSLLQPRYYSISSSPDMYPDEVHLTVAIV
SYRTRDGEGPIHHGVCSSWLNRIQADELVPCFVRGAPSFHLPRNPQVPCILVGPGTGIAP
FRSFWQQRQFDIQHKGMNPCPMVLVFGCRQSKIDHIYREETLQAKNKGVFRELYTAYSRE
PDKPKKYVQDILQEQLAESVYRALKEQGGHIYVCGDVTMAADVLKAIQRIMTQQGKLSAE
DAGVFISRMRDDNRYHEDIFGVTLRTYEVTNRLRSESIAFIEESKKDTDEVFSS
|
|
|
|---|
| BDBM50164780 |
|---|
| n/a |
|---|
| Name | BDBM50164780 |
|---|
| Synonyms: | 4-Methyl-3,4-dihydro-1H-quinolin-(2E)-ylideneamine | CHEMBL192033 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C10H12N2 |
|---|
| Mol. Mass. | 160.2157 |
|---|
| SMILES | CC1CC(N)=Nc2ccccc12 |c:4| |
|---|
| Structure | 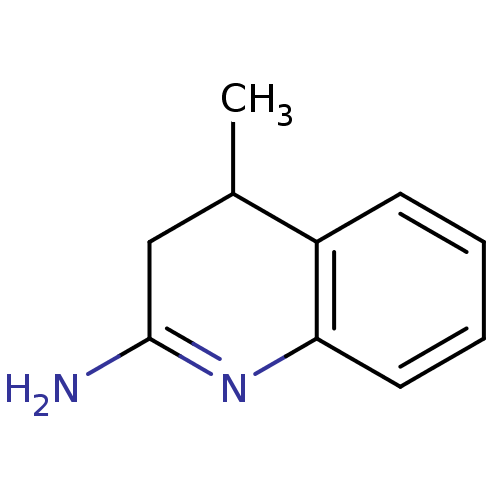
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Guthikonda, RN; Shah, SK; Pacholok, SG; Humes, JL; Mumford, RA; Grant, SK; Chabin, RM; Green, BG; Tsou, N; Ball, R; Fletcher, DS; Luell, S; Euan MacIntyre, D; Maccoss, M Bicyclic amidine inhibitors of nitric oxide synthase: discovery of perhydro-iminopyrindine and perhydro-iminoquinoline as potent, orally active inhibitors of inducible nitric oxide synthase. Bioorg Med Chem Lett15:1997-2001 (2005) [PubMed] Article
Guthikonda, RN; Shah, SK; Pacholok, SG; Humes, JL; Mumford, RA; Grant, SK; Chabin, RM; Green, BG; Tsou, N; Ball, R; Fletcher, DS; Luell, S; Euan MacIntyre, D; Maccoss, M Bicyclic amidine inhibitors of nitric oxide synthase: discovery of perhydro-iminopyrindine and perhydro-iminoquinoline as potent, orally active inhibitors of inducible nitric oxide synthase. Bioorg Med Chem Lett15:1997-2001 (2005) [PubMed] Article