| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM145466 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2303337 |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Sun, S; Jia, Q; Zenova, AY; Wilson, MS; Chowdhury, S; Focken, T; Li, J; Decker, S; Grimwood, ME; Andrez, JC; Hemeon, I; Sheng, T; Chen, CA; White, A; Hackos, DH; Deng, L; Bankar, G; Khakh, K; Chang, E; Kwan, R; Lin, S; Nelkenbrecher, K; Sellers, BD; DiPasquale, AG; Chang, J; Pang, J; Sojo, L; Lindgren, A; Waldbrook, M; Xie, Z; Young, C; Johnson, JP; Robinette, CL; Cohen, CJ; Safina, BS; Sutherlin, DP; Ortwine, DF; Dehnhardt, CM Identification of Selective Acyl Sulfonamide-Cycloalkylether Inhibitors of the Voltage-Gated Sodium Channel (Na J Med Chem62:908-927 (2019) [PubMed] Sun, S; Jia, Q; Zenova, AY; Wilson, MS; Chowdhury, S; Focken, T; Li, J; Decker, S; Grimwood, ME; Andrez, JC; Hemeon, I; Sheng, T; Chen, CA; White, A; Hackos, DH; Deng, L; Bankar, G; Khakh, K; Chang, E; Kwan, R; Lin, S; Nelkenbrecher, K; Sellers, BD; DiPasquale, AG; Chang, J; Pang, J; Sojo, L; Lindgren, A; Waldbrook, M; Xie, Z; Young, C; Johnson, JP; Robinette, CL; Cohen, CJ; Safina, BS; Sutherlin, DP; Ortwine, DF; Dehnhardt, CM Identification of Selective Acyl Sulfonamide-Cycloalkylether Inhibitors of the Voltage-Gated Sodium Channel (Na J Med Chem62:908-927 (2019) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM145466 |
|---|
| n/a |
|---|
| Name | BDBM145466 |
|---|
| Synonyms: | US8952169, 245 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H26F3NO4S |
|---|
| Mol. Mass. | 445.496 |
|---|
| SMILES | CC1(COc2cc(F)c(cc2C2CC2)C(=O)NS(=O)(=O)C2CC2)CCC(F)(F)CC1 |
|---|
| Structure | 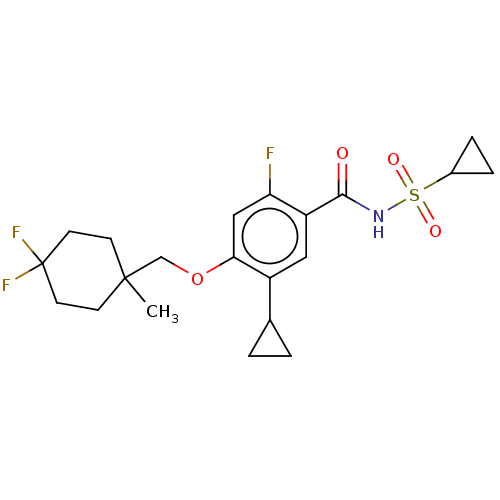
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sun, S; Jia, Q; Zenova, AY; Wilson, MS; Chowdhury, S; Focken, T; Li, J; Decker, S; Grimwood, ME; Andrez, JC; Hemeon, I; Sheng, T; Chen, CA; White, A; Hackos, DH; Deng, L; Bankar, G; Khakh, K; Chang, E; Kwan, R; Lin, S; Nelkenbrecher, K; Sellers, BD; DiPasquale, AG; Chang, J; Pang, J; Sojo, L; Lindgren, A; Waldbrook, M; Xie, Z; Young, C; Johnson, JP; Robinette, CL; Cohen, CJ; Safina, BS; Sutherlin, DP; Ortwine, DF; Dehnhardt, CM Identification of Selective Acyl Sulfonamide-Cycloalkylether Inhibitors of the Voltage-Gated Sodium Channel (Na J Med Chem62:908-927 (2019) [PubMed]
Sun, S; Jia, Q; Zenova, AY; Wilson, MS; Chowdhury, S; Focken, T; Li, J; Decker, S; Grimwood, ME; Andrez, JC; Hemeon, I; Sheng, T; Chen, CA; White, A; Hackos, DH; Deng, L; Bankar, G; Khakh, K; Chang, E; Kwan, R; Lin, S; Nelkenbrecher, K; Sellers, BD; DiPasquale, AG; Chang, J; Pang, J; Sojo, L; Lindgren, A; Waldbrook, M; Xie, Z; Young, C; Johnson, JP; Robinette, CL; Cohen, CJ; Safina, BS; Sutherlin, DP; Ortwine, DF; Dehnhardt, CM Identification of Selective Acyl Sulfonamide-Cycloalkylether Inhibitors of the Voltage-Gated Sodium Channel (Na J Med Chem62:908-927 (2019) [PubMed]