| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Aromatase |
|---|
| Ligand | BDBM10055 |
|---|
| Substrate/Competitor | BDBM8592 |
|---|
| Meas. Tech. | Aromatase Assay |
|---|
| pH | 7.2±n/a |
|---|
| Temperature | 310.15±n/a K |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Lesuisse, D; Gourvest, JF; Benslimane, O; Canu, F; Delaisi, C; Doucet, B; Hartmann, C; Lefrancois, JM; Tric, B; Mansuy, D; Philibert, D; Teutsch, G Structure-activity relationships of a new family of steroidal aromatase inhibitors. 1. Synthesis and evaluation of a series of analogs related to 19-[(methylthio)methyl]androstenedione (RU54115). J Med Chem39:757-72 (1996) [PubMed] Article Lesuisse, D; Gourvest, JF; Benslimane, O; Canu, F; Delaisi, C; Doucet, B; Hartmann, C; Lefrancois, JM; Tric, B; Mansuy, D; Philibert, D; Teutsch, G Structure-activity relationships of a new family of steroidal aromatase inhibitors. 1. Synthesis and evaluation of a series of analogs related to 19-[(methylthio)methyl]androstenedione (RU54115). J Med Chem39:757-72 (1996) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Aromatase |
|---|
| Name: | Aromatase |
|---|
| Synonyms: | ARO1 | Aromatase | CP19A_HUMAN | CYAR | CYP19 | CYP19A1 | CYPXIX | Cytochrome P-450AROM | Cytochrome P450 19A1 | Cytochrome P450 2C19 | Cytochrome P450-C19 (CYP19) | Estrogen synthetase | FL cytokine receptor precursor | P-450AROM |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57888.92 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11511 |
|---|
| Residue: | 503 |
|---|
| Sequence: | MVLEMLNPIHYNITSIVPEAMPAATMPVLLLTGLFLLVWNYEGTSSIPGPGYCMGIGPLI
SHGRFLWMGIGSACNYYNRVYGEFMRVWISGEETLIISKSSSMFHIMKHNHYSSRFGSKL
GLQCIGMHEKGIIFNNNPELWKTTRPFFMKALSGPGLVRMVTVCAESLKTHLDRLEEVTN
ESGYVDVLTLLRRVMLDTSNTLFLRIPLDESAIVVKIQGYFDAWQALLIKPDIFFKISWL
YKKYEKSVKDLKDAIEVLIAEKRRRISTEEKLEECMDFATELILAEKRGDLTRENVNQCI
LEMLIAAPDTMSVSLFFMLFLIAKHPNVEEAIIKEIQTVIGERDIKIDDIQKLKVMENFI
YESMRYQPVVDLVMRKALEDDVIDGYPVKKGTNIILNIGRMHRLEFFPKPNEFTLENFAK
NVPYRYFQPFGFGPRGCAGKYIAMVMMKAILVTLLRRFHVKTLQGQCVESIQKIHDLSLH
PDETKNMLEMIFTPRNSDRCLEH
|
|
|
|---|
| BDBM10055 |
|---|
| BDBM8592 |
|---|
| Name | BDBM10055 |
|---|
| Synonyms: | (2R,15S)-2-[2-(acetylsulfanyl)ethyl]-15-methyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadeca-1(17),6-diene-5,14-dione | 3,17-Dioxo-19-[(acetylthio)methyl]androsta-4,9(11)-diene | RU54115 Analog 13 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H28O3S |
|---|
| Mol. Mass. | 372.521 |
|---|
| SMILES | CC(=O)SCC[C@]12CCC(=O)C=C1CCC1C3CCC(=O)[C@@]3(C)CC=C21 |r,c:11,t:26| |
|---|
| Structure | 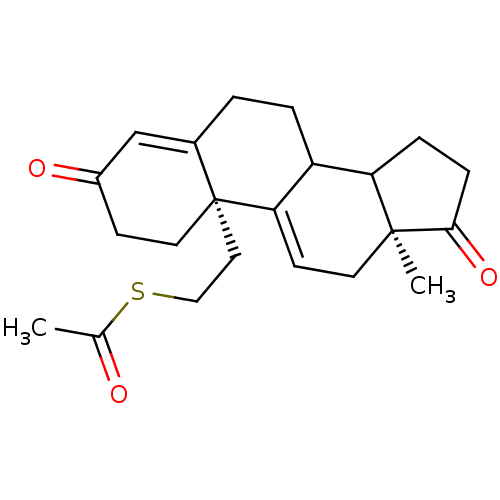
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lesuisse, D; Gourvest, JF; Benslimane, O; Canu, F; Delaisi, C; Doucet, B; Hartmann, C; Lefrancois, JM; Tric, B; Mansuy, D; Philibert, D; Teutsch, G Structure-activity relationships of a new family of steroidal aromatase inhibitors. 1. Synthesis and evaluation of a series of analogs related to 19-[(methylthio)methyl]androstenedione (RU54115). J Med Chem39:757-72 (1996) [PubMed] Article
Lesuisse, D; Gourvest, JF; Benslimane, O; Canu, F; Delaisi, C; Doucet, B; Hartmann, C; Lefrancois, JM; Tric, B; Mansuy, D; Philibert, D; Teutsch, G Structure-activity relationships of a new family of steroidal aromatase inhibitors. 1. Synthesis and evaluation of a series of analogs related to 19-[(methylthio)methyl]androstenedione (RU54115). J Med Chem39:757-72 (1996) [PubMed] Article