| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM10439 |
|---|
| Substrate/Competitor | BDBM8959 |
|---|
| Meas. Tech. | Enzyme Inhibition Assay |
|---|
| pH | 7±n/a |
|---|
| Temperature | 295.15±n/a K |
|---|
| IC50 | 151±36 nM |
|---|
| Citation |  Wong, DM; Greenblatt, HM; Dvir, H; Carlier, PR; Han, YF; Pang, YP; Silman, I; Sussman, JL Acetylcholinesterase complexed with bivalent ligands related to huperzine a: experimental evidence for species-dependent protein-ligand complementarity. J Am Chem Soc125:363-73 (2003) [PubMed] Article Wong, DM; Greenblatt, HM; Dvir, H; Carlier, PR; Han, YF; Pang, YP; Silman, I; Sussman, JL Acetylcholinesterase complexed with bivalent ligands related to huperzine a: experimental evidence for species-dependent protein-ligand complementarity. J Am Chem Soc125:363-73 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_RAT | Acetylcholinesterase (AChE) | Acetylcholinesterase and butyrylcholinesterase (AChE and BChE) | Acetylcholinesterase precursor | Acetylcholinesterase, AChE | Ache |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 68193.62 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | P37136 |
|---|
| Residue: | 614 |
|---|
| Sequence: | MRPPWYPLHTPSLASPLLFLLLSLLGGGARAEGREDPQLLVRVRGGQLRGIRLKAPGGPV
SAFLGIPFAEPPVGSRRFMPPEPKRPWSGILDATTFQNVCYQYVDTLYPGFEGTEMWNPN
RELSEDCLYLNVWTPYPRPTSPTPVLIWIYGGGFYSGASSLDVYDGRFLAQVEGTVLVSM
NYRVGTFGFLALPGSREAPGNVGLLDQRLALQWVQENIAAFGGDPMSVTLFGESAGAASV
GMHILSLPSRSLFHRAVLQSGTPNGPWATVSAGEARRRATLLARLVGCPPGGAGGNDTEL
ISCLRTRPAQDLVDHEWHVLPQESIFRFSFVPVVDGDFLSDTPDALINTGDFQDLQVLVG
VVKDEGSYFLVYGVPGFSKDNESLISRAQFLAGVRIGVPQASDLAAEAVVLHYTDWLHPE
DPAHLRDAMSAVVGDHNVVCPVAQLAGRLAAQGARVYAYIFEHRASTLTWPLWMGVPHGY
EIEFIFGLPLDPSLNYTVEERIFAQRLMQYWTNFARTGDPNDPRDSKSPRWPPYTTAAQQ
YVSLNLKPLEVRRGLRAQTCAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKN
QFDHYSKQERCSDL
|
|
|
|---|
| BDBM10439 |
|---|
| BDBM8959 |
|---|
| Name | BDBM10439 |
|---|
| Synonyms: | (5S)-5-[(10-{[(5S)-2-oxo-1,2,5,6,7,8-hexahydroquinolin-5-yl]amino}decyl)amino]-1,2,5,6,7,8-hexahydroquinolin-2-one | (S,S)-(-)-bis(10)-hupyridone |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H42N4O2 |
|---|
| Mol. Mass. | 466.6587 |
|---|
| SMILES | O=c1ccc2[C@H](CCCc2[nH]1)NCCCCCCCCCCN[C@H]1CCCc2[nH]c(=O)ccc12 |r| |
|---|
| Structure | 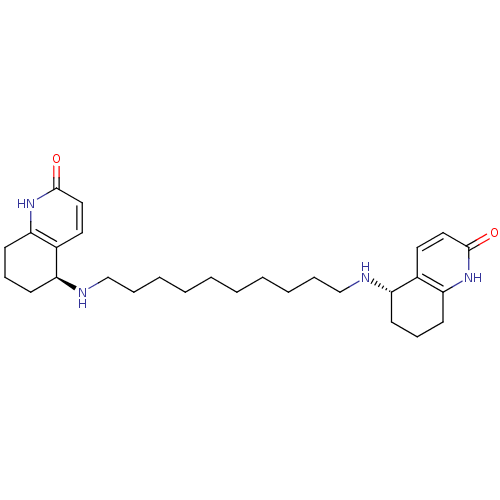
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wong, DM; Greenblatt, HM; Dvir, H; Carlier, PR; Han, YF; Pang, YP; Silman, I; Sussman, JL Acetylcholinesterase complexed with bivalent ligands related to huperzine a: experimental evidence for species-dependent protein-ligand complementarity. J Am Chem Soc125:363-73 (2003) [PubMed] Article
Wong, DM; Greenblatt, HM; Dvir, H; Carlier, PR; Han, YF; Pang, YP; Silman, I; Sussman, JL Acetylcholinesterase complexed with bivalent ligands related to huperzine a: experimental evidence for species-dependent protein-ligand complementarity. J Am Chem Soc125:363-73 (2003) [PubMed] Article