| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor IX |
|---|
| Ligand | BDBM12860 |
|---|
| Substrate/Competitor | BDBM12774 |
|---|
| Meas. Tech. | Enzyme Assay and Determination of the Inhibition Constants. |
|---|
| pH | 7.4±n/a |
|---|
| Temperature | 298.15±n/a K |
|---|
| Ki | >41000±n/a nM |
|---|
| Citation |  Li, YL; Fevig, JM; Cacciola, J; Buriak, J; Rossi, KA; Jona, J; Knabb, RM; Luettgen, JM; Wong, PC; Bai, SA; Wexler, RR; Lam, PY Preparation of 1-(3-aminobenzo[d]isoxazol-5-yl)-1H-pyrazolo[4,3-d]pyrimidin-7(6H)-ones as potent, selective, and efficacious inhibitors of coagulation factor Xa. Bioorg Med Chem Lett16:5176-82 (2006) [PubMed] Article Li, YL; Fevig, JM; Cacciola, J; Buriak, J; Rossi, KA; Jona, J; Knabb, RM; Luettgen, JM; Wong, PC; Bai, SA; Wexler, RR; Lam, PY Preparation of 1-(3-aminobenzo[d]isoxazol-5-yl)-1H-pyrazolo[4,3-d]pyrimidin-7(6H)-ones as potent, selective, and efficacious inhibitors of coagulation factor Xa. Bioorg Med Chem Lett16:5176-82 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Coagulation factor IX |
|---|
| Name: | Coagulation factor IX |
|---|
| Synonyms: | Christmas factor | Coagulation factor IX precursor | Coagulation factor IX/VIII | Coagulation factor IXa | Coagulation factor IXa heavy chain | Coagulation factor IXa light chain | F9 | FA9_HUMAN | Factor IX | Factor IXa | Factor IXa (fIXa) | PTC | Plasma thromboplastin component |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 51768.83 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00740 |
|---|
| Residue: | 461 |
|---|
| Sequence: | MQRVNMIMAESPGLITICLLGYLLSAECTVFLDHENANKILNRPKRYNSGKLEEFVQGNL
ERECMEEKCSFEEAREVFENTERTTEFWKQYVDGDQCESNPCLNGGSCKDDINSYECWCP
FGFEGKNCELDVTCNIKNGRCEQFCKNSADNKVVCSCTEGYRLAENQKSCEPAVPFPCGR
VSVSQTSKLTRAETVFPDVDYVNSTEAETILDNITQSTQSFNDFTRVVGGEDAKPGQFPW
QVVLNGKVDAFCGGSIVNEKWIVTAAHCVETGVKITVVAGEHNIEETEHTEQKRNVIRII
PHHNYNAAINKYNHDIALLELDEPLVLNSYVTPICIADKEYTNIFLKFGSGYVSGWGRVF
HKGRSALVLQYLRVPLVDRATCLRSTKFTIYNNMFCAGFHEGGRDSCQGDSGGPHVTEVE
GTSFLTGIISWGEECAMKGKYGIYTKVSRYVNWIKEKTKLT
|
|
|
|---|
| BDBM12860 |
|---|
| BDBM12774 |
|---|
| Name | BDBM12860 |
|---|
| Synonyms: | 1-(3-amino-1,2-benzoxazol-5-yl)-6-(4-{2-[(dimethylamino)methyl]phenyl}phenyl)-3,5-dimethyl-1H,6H,7H-pyrazolo[4,3-d]pyrimidin-7-one | Pyrazolo[4,3-d]pyrimidinone 19f |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H27N7O2 |
|---|
| Mol. Mass. | 505.5704 |
|---|
| SMILES | CN(C)Cc1ccccc1-c1ccc(cc1)-n1c(C)nc2c(C)nn(-c3ccc4onc(N)c4c3)c2c1=O |
|---|
| Structure | 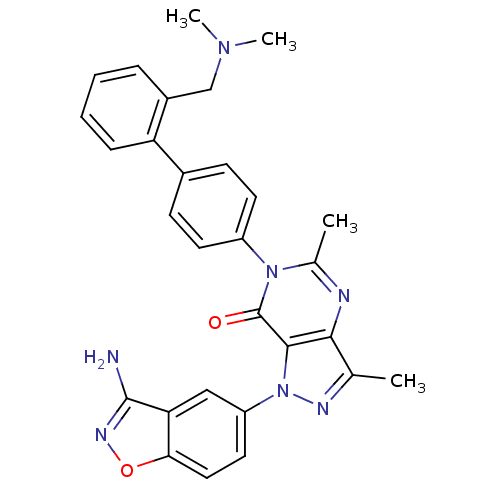
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Li, YL; Fevig, JM; Cacciola, J; Buriak, J; Rossi, KA; Jona, J; Knabb, RM; Luettgen, JM; Wong, PC; Bai, SA; Wexler, RR; Lam, PY Preparation of 1-(3-aminobenzo[d]isoxazol-5-yl)-1H-pyrazolo[4,3-d]pyrimidin-7(6H)-ones as potent, selective, and efficacious inhibitors of coagulation factor Xa. Bioorg Med Chem Lett16:5176-82 (2006) [PubMed] Article
Li, YL; Fevig, JM; Cacciola, J; Buriak, J; Rossi, KA; Jona, J; Knabb, RM; Luettgen, JM; Wong, PC; Bai, SA; Wexler, RR; Lam, PY Preparation of 1-(3-aminobenzo[d]isoxazol-5-yl)-1H-pyrazolo[4,3-d]pyrimidin-7(6H)-ones as potent, selective, and efficacious inhibitors of coagulation factor Xa. Bioorg Med Chem Lett16:5176-82 (2006) [PubMed] Article