| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sphingosine 1-phosphate receptor 5 |
|---|
| Ligand | BDBM22226 |
|---|
| Substrate/Competitor | BDBM10852 |
|---|
| Meas. Tech. | Ligand-Induced Uptake of [35S]-GTP-gamma-S |
|---|
| pH | 7.4±n/a |
|---|
| Temperature | 295.15±n/a K |
|---|
| EC50 | 28.6±n/a nM |
|---|
| Citation |  Yan, L; Huo, P; Hale, JJ; Mills, SG; Hajdu, R; Keohane, CA; Rosenbach, MJ; Milligan, JA; Shei, GJ; Chrebet, G; Bergstrom, J; Card, D; Mandala, SM SAR studies of 3-arylpropionic acids as potent and selective agonists of sphingosine-1-phosphate receptor-1 (S1P1) with enhanced pharmacokinetic properties. Bioorg Med Chem Lett17:828-31 (2007) [PubMed] Article Yan, L; Huo, P; Hale, JJ; Mills, SG; Hajdu, R; Keohane, CA; Rosenbach, MJ; Milligan, JA; Shei, GJ; Chrebet, G; Bergstrom, J; Card, D; Mandala, SM SAR studies of 3-arylpropionic acids as potent and selective agonists of sphingosine-1-phosphate receptor-1 (S1P1) with enhanced pharmacokinetic properties. Bioorg Med Chem Lett17:828-31 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Sphingosine 1-phosphate receptor 5 |
|---|
| Name: | Sphingosine 1-phosphate receptor 5 |
|---|
| Synonyms: | EDG8 | Endothelial differentiation sphingolipid G-protein-coupled receptor 8 | S1P5 | S1PR5 | S1PR5_HUMAN | Sphingosine 1-phosphate receptor | Sphingosine 1-phosphate receptor Edg-8 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 41796.42 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 398 |
|---|
| Sequence: | MESGLLRPAPVSEVIVLHYNYTGKLRGARYQPGAGLRADAVVCLAVCAFIVLENLAVLLV
LGRHPRFHAPMFLLLGSLTLSDLLAGAAYAANILLSGPLTLKLSPALWFAREGGVFVALT
ASVLSLLAIALERSLTMARRGPAPVSSRGRTLAMAAAAWGVSLLLGLLPALGWNCLGRLD
ACSTVLPLYAKAYVLFCVLAFVGILAAICALYARIYCQVRANARRLPARPGTAGTTSTRA
RRKPRSLALLRTLSVVLLAFVACWGPLFLLLLLDVACPARTCPVLLQADPFLGLAMANSL
LNPIIYTLTNRDLRHALLRLVCCGRHSCGRDPSGSQQSASAAEASGGLRRCLPPGLDGSF
SGSERSSPQRDGLDTSGSTGSPGAPTAARTLVSEPAAD
|
|
|
|---|
| BDBM22226 |
|---|
| BDBM10852 |
|---|
| Name | BDBM22226 |
|---|
| Synonyms: | 3-(5-{5-[3-cyano-4-(propan-2-yloxy)phenyl]-1,2,4-oxadiazol-3-yl}-6-methylpyridin-2-yl)propanoic acid | 3-arylpropionic acid derivative, 14 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H20N4O4 |
|---|
| Mol. Mass. | 392.4079 |
|---|
| SMILES | CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc(CCC(O)=O)nc1C |
|---|
| Structure | 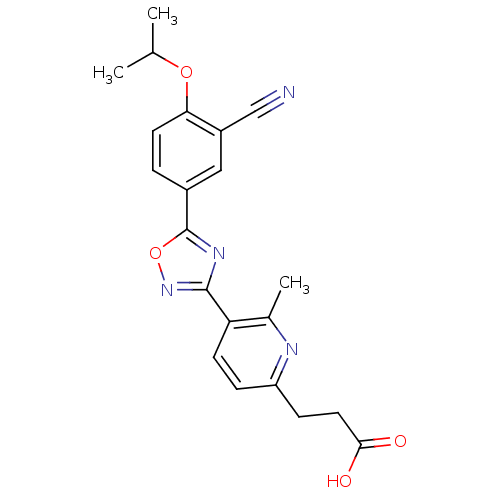
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yan, L; Huo, P; Hale, JJ; Mills, SG; Hajdu, R; Keohane, CA; Rosenbach, MJ; Milligan, JA; Shei, GJ; Chrebet, G; Bergstrom, J; Card, D; Mandala, SM SAR studies of 3-arylpropionic acids as potent and selective agonists of sphingosine-1-phosphate receptor-1 (S1P1) with enhanced pharmacokinetic properties. Bioorg Med Chem Lett17:828-31 (2007) [PubMed] Article
Yan, L; Huo, P; Hale, JJ; Mills, SG; Hajdu, R; Keohane, CA; Rosenbach, MJ; Milligan, JA; Shei, GJ; Chrebet, G; Bergstrom, J; Card, D; Mandala, SM SAR studies of 3-arylpropionic acids as potent and selective agonists of sphingosine-1-phosphate receptor-1 (S1P1) with enhanced pharmacokinetic properties. Bioorg Med Chem Lett17:828-31 (2007) [PubMed] Article