| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histamine H4 receptor |
|---|
| Ligand | BDBM26226 |
|---|
| Substrate/Competitor | BDBM7966 |
|---|
| Meas. Tech. | Radioligand Binding Assay |
|---|
| pH | 7.4±n/a |
|---|
| Temperature | 298.15±n/a K |
|---|
| Ki | 5.8±2.2 nM |
|---|
| Citation |  Liu, H; Altenbach, RJ; Carr, TL; Chandran, P; Hsieh, GC; Lewis, LG; Manelli, AM; Milicic, I; Marsh, KC; Miller, TR; Strakhova, MI; Vortherms, TA; Wakefield, BD; Wetter, JM; Witte, DG; Honore, P; Esbenshade, TA; Brioni, JD; Cowart, MD cis-4-(Piperazin-1-yl)-5,6,7a,8,9,10,11,11a-octahydrobenzofuro[2,3-h]quinazolin-2-amine (A-987306), A New Histamine H4R Antagonist that Blocks Pain Responses against Carrageenan-Induced Hyperalgesia. J Med Chem51:7094-8 (2008) [PubMed] Article Liu, H; Altenbach, RJ; Carr, TL; Chandran, P; Hsieh, GC; Lewis, LG; Manelli, AM; Milicic, I; Marsh, KC; Miller, TR; Strakhova, MI; Vortherms, TA; Wakefield, BD; Wetter, JM; Witte, DG; Honore, P; Esbenshade, TA; Brioni, JD; Cowart, MD cis-4-(Piperazin-1-yl)-5,6,7a,8,9,10,11,11a-octahydrobenzofuro[2,3-h]quinazolin-2-amine (A-987306), A New Histamine H4R Antagonist that Blocks Pain Responses against Carrageenan-Induced Hyperalgesia. J Med Chem51:7094-8 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Histamine H4 receptor |
|---|
| Name: | Histamine H4 receptor |
|---|
| Synonyms: | AXOR35 | G-protein coupled receptor 105 | GPCR105 | GPRv53 | HH4R | HISTAMINE H4 | HRH4 | HRH4_HUMAN | Histamine H4 receptor | Histamine H4 receptor (H4R) | Histamine receptor (H3 and H4) | Pfi-013 | SP9144 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44517.02 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Binding assays were using CHO cells stably expressing hH4R receptors. |
|---|
| Residue: | 390 |
|---|
| Sequence: | MPDTNSTINLSLSTRVTLAFFMSLVAFAIMLGNALVILAFVVDKNLRHRSSYFFLNLAIS
DFFVGVISIPLYIPHTLFEWDFGKEICVFWLTTDYLLCTASVYNIVLISYDRYLSVSNAV
SYRTQHTGVLKIVTLMVAVWVLAFLVNGPMILVSESWKDEGSECEPGFFSEWYILAITSF
LEFVIPVILVAYFNMNIYWSLWKRDHLSRCQSHPGLTAVSSNICGHSFRGRLSSRRSLSA
STEVPASFHSERQRRKSSLMFSSRTKMNSNTIASKMGSFSQSDSVALHQREHVELLRARR
LAKSLAILLGVFAVCWAPYSLFTIVLSFYSSATGPKSVWYRIAFWLQWFNSFVNPLLYPL
CHKRFQKAFLKIFCIKKQPLPSQHSRSVSS
|
|
|
|---|
| BDBM26226 |
|---|
| BDBM7966 |
|---|
| Name | BDBM26226 |
|---|
| Synonyms: | (12S,17S)-6-(piperazin-1-yl)-11-oxa-3,5-diazatetracyclo[8.7.0.0^{2,7}.0^{12,17}]heptadeca-1(10),2(7),3,5-tetraen-4-amine | A-987306 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H25N5O |
|---|
| Mol. Mass. | 327.424 |
|---|
| SMILES | [H][C@]12CCCC[C@@]1([H])C1=C(CCc3c(nc(N)nc13)N1CCNCC1)O2 |r,t:9| |
|---|
| Structure | 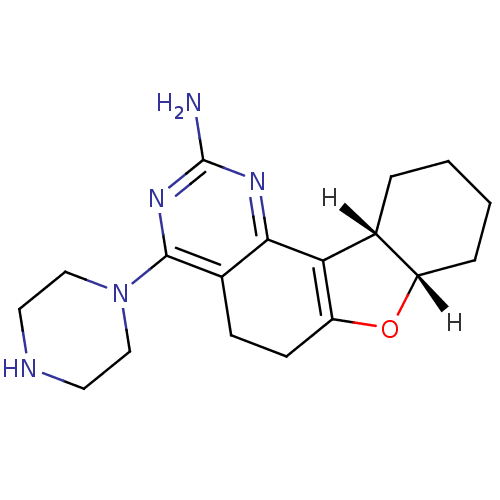
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Liu, H; Altenbach, RJ; Carr, TL; Chandran, P; Hsieh, GC; Lewis, LG; Manelli, AM; Milicic, I; Marsh, KC; Miller, TR; Strakhova, MI; Vortherms, TA; Wakefield, BD; Wetter, JM; Witte, DG; Honore, P; Esbenshade, TA; Brioni, JD; Cowart, MD cis-4-(Piperazin-1-yl)-5,6,7a,8,9,10,11,11a-octahydrobenzofuro[2,3-h]quinazolin-2-amine (A-987306), A New Histamine H4R Antagonist that Blocks Pain Responses against Carrageenan-Induced Hyperalgesia. J Med Chem51:7094-8 (2008) [PubMed] Article
Liu, H; Altenbach, RJ; Carr, TL; Chandran, P; Hsieh, GC; Lewis, LG; Manelli, AM; Milicic, I; Marsh, KC; Miller, TR; Strakhova, MI; Vortherms, TA; Wakefield, BD; Wetter, JM; Witte, DG; Honore, P; Esbenshade, TA; Brioni, JD; Cowart, MD cis-4-(Piperazin-1-yl)-5,6,7a,8,9,10,11,11a-octahydrobenzofuro[2,3-h]quinazolin-2-amine (A-987306), A New Histamine H4R Antagonist that Blocks Pain Responses against Carrageenan-Induced Hyperalgesia. J Med Chem51:7094-8 (2008) [PubMed] Article