| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2E1 |
|---|
| Ligand | BDBM50341416 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1734947 (CHEMBL4150483) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Lin, S; Wang, C; Ji, M; Wu, D; Lv, Y; Zhang, K; Dong, Y; Jin, J; Chen, J; Zhang, J; Sheng, L; Li, Y; Chen, X; Xu, H Discovery and Optimization of 2-Amino-4-methylquinazoline Derivatives as Highly Potent Phosphatidylinositol 3-Kinase Inhibitors for Cancer Treatment. J Med Chem61:6087-6109 (2018) [PubMed] Article Lin, S; Wang, C; Ji, M; Wu, D; Lv, Y; Zhang, K; Dong, Y; Jin, J; Chen, J; Zhang, J; Sheng, L; Li, Y; Chen, X; Xu, H Discovery and Optimization of 2-Amino-4-methylquinazoline Derivatives as Highly Potent Phosphatidylinositol 3-Kinase Inhibitors for Cancer Treatment. J Med Chem61:6087-6109 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2E1 |
|---|
| Name: | Cytochrome P450 2E1 |
|---|
| Synonyms: | CP2E1_HUMAN | CYP2E | CYP2E1 | Cytochrome P450 2E1 (CYP2E1) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 56860.34 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05181 |
|---|
| Residue: | 493 |
|---|
| Sequence: | MSALGVTVALLVWAAFLLLVSMWRQVHSSWNLPPGPFPLPIIGNLFQLELKNIPKSFTRL
AQRFGPVFTLYVGSQRMVVMHGYKAVKEALLDYKDEFSGRGDLPAFHAHRDRGIIFNNGP
TWKDIRRFSLTTLRNYGMGKQGNESRIQREAHFLLEALRKTQGQPFDPTFLIGCAPCNVI
ADILFRKHFDYNDEKFLRLMYLFNENFHLLSTPWLQLYNNFPSFLHYLPGSHRKVIKNVA
EVKEYVSERVKEHHQSLDPNCPRDLTDCLLVEMEKEKHSAERLYTMDGITVTVADLFFAG
TETTSTTLRYGLLILMKYPEIEEKLHEEIDRVIGPSRIPAIKDRQEMPYMDAVVHEIQRF
ITLVPSNLPHEATRDTIFRGYLIPKGTVVVPTLDSVLYDNQEFPDPEKFKPEHFLNENGK
FKYSDYFKPFSTGKRVCAGEGLARMELFLLLCAILQHFNLKPLVDPKDIDLSPIHIGFGC
IPPRYKLCVIPRS
|
|
|
|---|
| BDBM50341416 |
|---|
| n/a |
|---|
| Name | BDBM50341416 |
|---|
| Synonyms: | CHEMBL4174909 | US11534443, Example 32 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H25F2N5O5S |
|---|
| Mol. Mass. | 557.569 |
|---|
| SMILES | CNc1nc(C)c2cc(cc(O[C@@H]3CCOC3)c2n1)-c1cnc(OC)c(NS(=O)(=O)c2ccc(F)cc2F)c1 |r| |
|---|
| Structure | 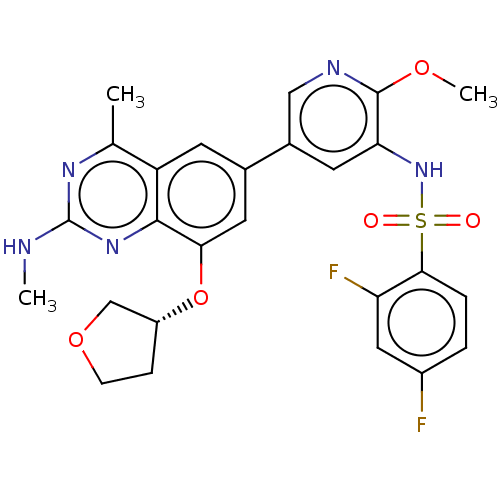
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lin, S; Wang, C; Ji, M; Wu, D; Lv, Y; Zhang, K; Dong, Y; Jin, J; Chen, J; Zhang, J; Sheng, L; Li, Y; Chen, X; Xu, H Discovery and Optimization of 2-Amino-4-methylquinazoline Derivatives as Highly Potent Phosphatidylinositol 3-Kinase Inhibitors for Cancer Treatment. J Med Chem61:6087-6109 (2018) [PubMed] Article
Lin, S; Wang, C; Ji, M; Wu, D; Lv, Y; Zhang, K; Dong, Y; Jin, J; Chen, J; Zhang, J; Sheng, L; Li, Y; Chen, X; Xu, H Discovery and Optimization of 2-Amino-4-methylquinazoline Derivatives as Highly Potent Phosphatidylinositol 3-Kinase Inhibitors for Cancer Treatment. J Med Chem61:6087-6109 (2018) [PubMed] Article