| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Polyunsaturated fatty acid 5-lipoxygenase |
|---|
| Ligand | BDBM31132 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1800826 (CHEMBL4273118) |
|---|
| IC50 | 2120±n/a nM |
|---|
| Citation |  Bruno, F; Errico, S; Pace, S; Nawrozkij, MB; Mkrtchyan, AS; Guida, F; Maisto, R; Olga�, A; D'Amico, M; Maione, S; De Rosa, M; Banoglu, E; Werz, O; Fiorentino, A; Filosa, R Structural insight into the optimization of ethyl 5-hydroxybenzo[g]indol-3-carboxylates and their bioisosteric analogues as 5-LO/m-PGES-1 dual inhibitors able to suppress inflammation. Eur J Med Chem155:946-960 (2018) [PubMed] Article Bruno, F; Errico, S; Pace, S; Nawrozkij, MB; Mkrtchyan, AS; Guida, F; Maisto, R; Olga�, A; D'Amico, M; Maione, S; De Rosa, M; Banoglu, E; Werz, O; Fiorentino, A; Filosa, R Structural insight into the optimization of ethyl 5-hydroxybenzo[g]indol-3-carboxylates and their bioisosteric analogues as 5-LO/m-PGES-1 dual inhibitors able to suppress inflammation. Eur J Med Chem155:946-960 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Polyunsaturated fatty acid 5-lipoxygenase |
|---|
| Name: | Polyunsaturated fatty acid 5-lipoxygenase |
|---|
| Synonyms: | 5-LO | 5-Lipo-oxygenase (5-LOX) | 5-Lipoxygenase (5-LO) | 5-Lipoxygenase (LOX) | 5-Lipoygenase | 5-lipoxygenase/FLAP | ALOX5 | Arachidonate 5-lipoxygenase | LOG5 | LOX5_HUMAN |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 77972.74 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Recombinant protein was purified from E. coli lysate. After ammonium sulfate precipitation and subsequent steps, the supernatant (S100) was used for 5-LO activity assay.
|
|---|
| Residue: | 674 |
|---|
| Sequence: | MPSYTVTVATGSQWFAGTDDYIYLSLVGSAGCSEKHLLDKPFYNDFERGAVDSYDVTVDE
ELGEIQLVRIEKRKYWLNDDWYLKYITLKTPHGDYIEFPCYRWITGDVEVVLRDGRAKLA
RDDQIHILKQHRRKELETRQKQYRWMEWNPGFPLSIDAKCHKDLPRDIQFDSEKGVDFVL
NYSKAMENLFINRFMHMFQSSWNDFADFEKIFVKISNTISERVMNHWQEDLMFGYQFLNG
CNPVLIRRCTELPEKLPVTTEMVECSLERQLSLEQEVQQGNIFIVDFELLDGIDANKTDP
CTLQFLAAPICLLYKNLANKIVPIAIQLNQIPGDENPIFLPSDAKYDWLLAKIWVRSSDF
HVHQTITHLLRTHLVSEVFGIAMYRQLPAVHPIFKLLVAHVRFTIAINTKAREQLICECG
LFDKANATGGGGHVQMVQRAMKDLTYASLCFPEAIKARGMESKEDIPYYFYRDDGLLVWE
AIRTFTAEVVDIYYEGDQVVEEDPELQDFVNDVYVYGMRGRKSSGFPKSVKSREQLSEYL
TVVIFTASAQHAAVNFGQYDWCSWIPNAPPTMRAPPPTAKGVVTIEQIVDTLPDRGRSCW
HLGAVWALSQFQENELFLGMYPEEHFIEKPVKEAMARFRKNLEAIVSVIAERNKKKQLPY
YYLSPDRIPNSVAI
|
|
|
|---|
| BDBM31132 |
|---|
| n/a |
|---|
| Name | BDBM31132 |
|---|
| Synonyms: | 5-hydroxy-1H-benzo[g]indole-3-carboxylate, 11k |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H16ClNO3 |
|---|
| Mol. Mass. | 365.81 |
|---|
| SMILES | CCOC(=O)c1c([nH]c2c1cc(O)c1ccccc21)-c1cccc(Cl)c1 |
|---|
| Structure | 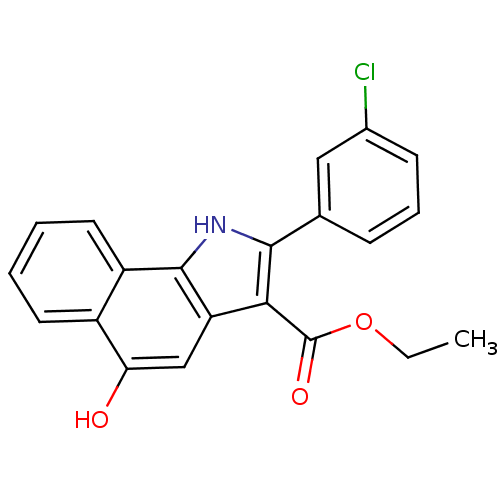
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bruno, F; Errico, S; Pace, S; Nawrozkij, MB; Mkrtchyan, AS; Guida, F; Maisto, R; Olga�, A; D'Amico, M; Maione, S; De Rosa, M; Banoglu, E; Werz, O; Fiorentino, A; Filosa, R Structural insight into the optimization of ethyl 5-hydroxybenzo[g]indol-3-carboxylates and their bioisosteric analogues as 5-LO/m-PGES-1 dual inhibitors able to suppress inflammation. Eur J Med Chem155:946-960 (2018) [PubMed] Article
Bruno, F; Errico, S; Pace, S; Nawrozkij, MB; Mkrtchyan, AS; Guida, F; Maisto, R; Olga�, A; D'Amico, M; Maione, S; De Rosa, M; Banoglu, E; Werz, O; Fiorentino, A; Filosa, R Structural insight into the optimization of ethyl 5-hydroxybenzo[g]indol-3-carboxylates and their bioisosteric analogues as 5-LO/m-PGES-1 dual inhibitors able to suppress inflammation. Eur J Med Chem155:946-960 (2018) [PubMed] Article