| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-2A adrenergic receptor |
|---|
| Ligand | BDBM50474632 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_33207 (CHEMBL643232) |
|---|
| Ki | 23±n/a nM |
|---|
| Citation |  Altenbach, RJ; Khilevich, A; Kolasa, T; Rohde, JJ; Bhatia, PA; Patel, MV; Searle, XB; Yang, F; Bunnelle, WH; Tietje, K; Bayburt, EK; Carroll, WA; Meyer, MD; Henry, R; Buckner, SA; Kuk, J; Daza, AV; Milicic, IV; Cain, JC; Kang, CH; Ireland, LM; Carr, TL; Miller, TR; Hancock, AA; Nakane, M; Esbenshade, TA; Brune, ME; O'Neill, AB; Gauvin, DM; Katwala, SP; Holladay, MW; Brioni, JD; Sullivan, JP Synthesis and structure-activity studies on N-[5-(1H-imidazol-4-yl)-5,6,7,8-tetrahydro-1-naphthalenyl]methanesulfonamide, an imidazole-containing alpha(1A)-adrenoceptor agonist. J Med Chem47:3220-35 (2004) [PubMed] Article Altenbach, RJ; Khilevich, A; Kolasa, T; Rohde, JJ; Bhatia, PA; Patel, MV; Searle, XB; Yang, F; Bunnelle, WH; Tietje, K; Bayburt, EK; Carroll, WA; Meyer, MD; Henry, R; Buckner, SA; Kuk, J; Daza, AV; Milicic, IV; Cain, JC; Kang, CH; Ireland, LM; Carr, TL; Miller, TR; Hancock, AA; Nakane, M; Esbenshade, TA; Brune, ME; O'Neill, AB; Gauvin, DM; Katwala, SP; Holladay, MW; Brioni, JD; Sullivan, JP Synthesis and structure-activity studies on N-[5-(1H-imidazol-4-yl)-5,6,7,8-tetrahydro-1-naphthalenyl]methanesulfonamide, an imidazole-containing alpha(1A)-adrenoceptor agonist. J Med Chem47:3220-35 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-2A adrenergic receptor |
|---|
| Name: | Alpha-2A adrenergic receptor |
|---|
| Synonyms: | ADA2A_HUMAN | ADRA2A | ADRA2R | ADRAR | Adrenergic alpha2A | Adrenergic receptor alpha | Alpha-2 adrenergic receptor subtype C10 | Alpha-2A adrenoceptor | Alpha-2A adrenoreceptor | Alpha-2AAR | alpha-2A adrenergic receptor [Homo sapiens] |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48979.91 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P08913 |
|---|
| Residue: | 465 |
|---|
| Sequence: | MFRQEQPLAEGSFAPMGSLQPDAGNASWNGTEAPGGGARATPYSLQVTLTLVCLAGLLML
LTVFGNVLVIIAVFTSRALKAPQNLFLVSLASADILVATLVIPFSLANEVMGYWYFGKAW
CEIYLALDVLFCTSSIVHLCAISLDRYWSITQAIEYNLKRTPRRIKAIIITVWVISAVIS
FPPLISIEKKGGGGGPQPAEPRCEINDQKWYVISSCIGSFFAPCLIMILVYVRIYQIAKR
RTRVPPSRRGPDAVAAPPGGTERRPNGLGPERSAGPGGAEAEPLPTQLNGAPGEPAPAGP
RDTDALDLEESSSSDHAERPPGPRRPERGPRGKGKARASQVKPGDSLPRRGPGATGIGTP
AAGPGEERVGAAKASRWRGRQNREKRFTFVLAVVIGVFVVCWFPFFFTYTLTAVGCSVPR
TLFKFFFWFGYCNSSLNPVIYTIFNHDFRRAFKKILCRGDRKRIV
|
|
|
|---|
| BDBM50474632 |
|---|
| n/a |
|---|
| Name | BDBM50474632 |
|---|
| Synonyms: | CHEMBL327116 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H19N3O2S |
|---|
| Mol. Mass. | 305.395 |
|---|
| SMILES | CCS(=O)(=O)Nc1ccc2CCCC(c3c[nH]cn3)c2c1 |
|---|
| Structure | 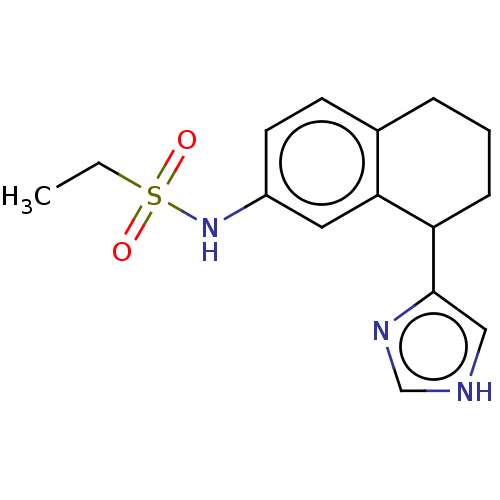
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Altenbach, RJ; Khilevich, A; Kolasa, T; Rohde, JJ; Bhatia, PA; Patel, MV; Searle, XB; Yang, F; Bunnelle, WH; Tietje, K; Bayburt, EK; Carroll, WA; Meyer, MD; Henry, R; Buckner, SA; Kuk, J; Daza, AV; Milicic, IV; Cain, JC; Kang, CH; Ireland, LM; Carr, TL; Miller, TR; Hancock, AA; Nakane, M; Esbenshade, TA; Brune, ME; O'Neill, AB; Gauvin, DM; Katwala, SP; Holladay, MW; Brioni, JD; Sullivan, JP Synthesis and structure-activity studies on N-[5-(1H-imidazol-4-yl)-5,6,7,8-tetrahydro-1-naphthalenyl]methanesulfonamide, an imidazole-containing alpha(1A)-adrenoceptor agonist. J Med Chem47:3220-35 (2004) [PubMed] Article
Altenbach, RJ; Khilevich, A; Kolasa, T; Rohde, JJ; Bhatia, PA; Patel, MV; Searle, XB; Yang, F; Bunnelle, WH; Tietje, K; Bayburt, EK; Carroll, WA; Meyer, MD; Henry, R; Buckner, SA; Kuk, J; Daza, AV; Milicic, IV; Cain, JC; Kang, CH; Ireland, LM; Carr, TL; Miller, TR; Hancock, AA; Nakane, M; Esbenshade, TA; Brune, ME; O'Neill, AB; Gauvin, DM; Katwala, SP; Holladay, MW; Brioni, JD; Sullivan, JP Synthesis and structure-activity studies on N-[5-(1H-imidazol-4-yl)-5,6,7,8-tetrahydro-1-naphthalenyl]methanesulfonamide, an imidazole-containing alpha(1A)-adrenoceptor agonist. J Med Chem47:3220-35 (2004) [PubMed] Article