

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Reverse transcriptase | ||
| Ligand | BDBM50492151 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_968173 (CHEMBL2399276) | ||
| IC50 | 5.5±n/a nM | ||
| Citation |  Sturino, CF; Bousquet, Y; James, CA; DeRoy, P; Duplessis, M; Edwards, PJ; Halmos, T; Minville, J; Morency, L; Morin, S; Thavonekham, B; Tremblay, M; Duan, J; Ribadeneira, M; Garneau, M; Pelletier, A; Tremblay, S; Lamorte, L; Bethell, R; Cordingley, MG; Rajotte, D; Simoneau, B Identification of potent and orally bioavailable nucleotide competing reverse transcriptase inhibitors: in vitro and in vivo optimization of a series of benzofurano[3,2-d]pyrimidin-2-one derived inhibitors. Bioorg Med Chem Lett23:3967-75 (2013) [PubMed] Article Sturino, CF; Bousquet, Y; James, CA; DeRoy, P; Duplessis, M; Edwards, PJ; Halmos, T; Minville, J; Morency, L; Morin, S; Thavonekham, B; Tremblay, M; Duan, J; Ribadeneira, M; Garneau, M; Pelletier, A; Tremblay, S; Lamorte, L; Bethell, R; Cordingley, MG; Rajotte, D; Simoneau, B Identification of potent and orally bioavailable nucleotide competing reverse transcriptase inhibitors: in vitro and in vivo optimization of a series of benzofurano[3,2-d]pyrimidin-2-one derived inhibitors. Bioorg Med Chem Lett23:3967-75 (2013) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Reverse transcriptase | |||
| Name: | Reverse transcriptase | ||
| Synonyms: | n/a | ||
| Type: | Protein | ||
| Mol. Mass.: | 29598.37 | ||
| Organism: | Human immunodeficiency virus 1 | ||
| Description: | Q9WKE8 | ||
| Residue: | 254 | ||
| Sequence: |
| ||
| BDBM50492151 | |||
| n/a | |||
| Name | BDBM50492151 | ||
| Synonyms: | CHEMBL2397588 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C26H27N7O3 | ||
| Mol. Mass. | 485.5377 | ||
| SMILES | CCC1(CC(C1)n1c2c3cc(cnc3oc2c(nc1=O)-c1ccc(NC)nc1)-c1cnn(C)c1)OC |(52.36,-18.14,;51.27,-19.22,;51.67,-20.72,;53.19,-20.95,;52.96,-22.47,;51.44,-22.24,;53.87,-23.71,;53.25,-25.13,;51.78,-25.61,;50.44,-24.85,;49.11,-25.62,;49.11,-27.16,;50.44,-27.93,;51.78,-27.16,;53.25,-27.64,;54.17,-26.38,;55.71,-26.22,;56.33,-24.8,;55.42,-23.54,;56.04,-22.13,;56.61,-27.45,;55.99,-28.86,;56.9,-30.1,;58.43,-29.94,;59.34,-31.18,;60.87,-31.01,;59.05,-28.52,;58.14,-27.28,;47.79,-24.84,;46.38,-25.47,;45.35,-24.32,;46.12,-22.99,;45.49,-21.58,;47.62,-23.31,;50.17,-20.31,;49.78,-18.83,)| | ||
| Structure | 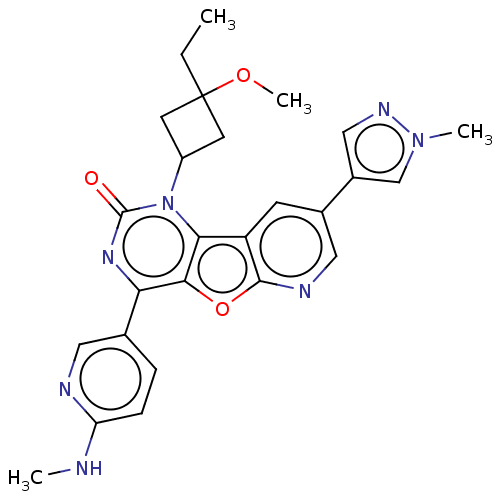
| ||